Age-related macular degeneration (AMD) stands as a primary cause of irreversible vision loss in individuals aged 65 and older within industrialized nations. Globally, in 2020, approximately 200 million people were affected by AMD, contributing to about 9% of all blindness cases worldwide. Alarmingly, the prevalence of AMD has significantly increased in recent years. In Germany alone, the number of individuals with early AMD, often asymptomatic, rose from 5.7 million in 2002 to around 7 million in 2017, marking a roughly 23% increase in just 15 years. The more advanced stages of AMD, which frequently present with noticeable symptoms and pose a threat to permanent vision impairment, also became more prevalent in Germany during the same period, increasing from approximately 360,000 to 490,000 individuals—a rise of about 36%. It’s estimated that late-stage AMD is responsible for half of all cases of blindness and severe visual impairment in Germany. In patients with late-stage AMD, neovascular AMD is observed to be 1.4 times more frequent than geographic atrophy, the end-stage of dry AMD. This increase in prevalence may be attributed not only to the aging population but also to enhanced detection through improved diagnostic methods. The substantial impact of demographic trends on AMD prevalence is evident in the age-adjusted prevalence rates, increasing from 24% in those aged 65 to 74 to over 44% in individuals aged 70 to 95.
Understanding AMD Prevalence
Age is the most significant risk factor for AMD, and as populations in industrialized countries age, the prevalence of this condition continues to rise.
German Statistics on AMD
Currently, approximately 7 million individuals in Germany are living with AMD.
The strong correlation between age and AMD development is further highlighted by the observation that individuals under 50 typically exhibit minimal to no typical AMD-related changes. Conversely, the Gutenberg Health Study conducted in Mainz, Germany, revealed that 24% of individuals aged 65 to 74 already show characteristic AMD changes, although many of these cases are still asymptomatic. The majority of early AMD-related changes observed were funduscopically visible deposits of metabolic byproducts beneath or above the retinal pigment epithelium of the macula, known as drusen and pseudodrusen. The prevalence of such changes increases with age. For instance, the AugUR cohort study in Regensburg, Germany, found intermediate-stage AMD in 44% of subjects aged 70 to 95 and late AMD in 19% of the same age group. Other studies estimate the prevalence of AMD in individuals over 85 to be around 30%.
Stages of Age-Related Macular Degeneration
AMD is clinically categorized into early, intermediate, and late stages. Late AMD is further divided into two subtypes: dry (atrophic) and neovascular (wet, exudative) late AMD. It is not uncommon to find a combination of both dry and wet AMD in the same eye.
Learning Objectives for AMD Diagnosis
Upon reading this guide, you will be able to:
- Understand the prevalence and risk factors associated with AMD.
- Recognize the typical clinical manifestations of AMD and the appropriate diagnostic evaluation process.
- Know the treatment strategies for each stage of AMD and be aware of potential treatment complications.
Diagnostic Methodology for AMD
This review is grounded in relevant publications identified through a focused PubMed search for original research and review articles, supplemented by current guidelines from pertinent specialist societies.
AMD Progression and Its Impact
In a significant number of AMD patients, 64.5% to be precise, the disease stage is consistent in both eyes. For those with asymmetrical involvement, the progression of AMD in the eye with better vision is of paramount concern. Large-scale population-based studies indicate that when AMD is initially diagnosed in only one eye, the other eye becomes affected within 5 years in 19% to 28% of cases.
Late-stage AMD has a far greater impact on vision compared to early AMD, which is often asymptomatic, or intermediate AMD, which is usually only mildly symptomatic. The progression rate from intermediate to late AMD in the natural course of the disease is typically reported as 28% within five years. Initial symptoms often include distorted vision or central visual field defects, frequently described as fixed, centrally located gray spots. Late AMD manifests in two primary forms: dry or atrophic, and wet or exudative (neovascular). Atrophic AMD is characterized by the gradual loss of retinal pigment epithelium, photoreceptors, and choroidal capillaries in the macula, the area responsible for sharp central vision. This form typically progresses slowly over several years and, in advanced stages, can lead to complete central vision loss, resulting in a central scotoma. Natural history studies of AMD have shown that atrophic areas develop in 19% of eyes with intermediate AMD within 5 years. Importantly, peripheral and orienting vision are generally preserved even in late AMD, as the degenerative process is confined to the macular region, sparing the rest of the retina. However, because the macula is the central part of the retina with the highest spatial resolution, patients with AMD often experience increasing difficulty with reading and recognizing objects and faces, while spatial orientation remains intact due to preserved peripheral vision.
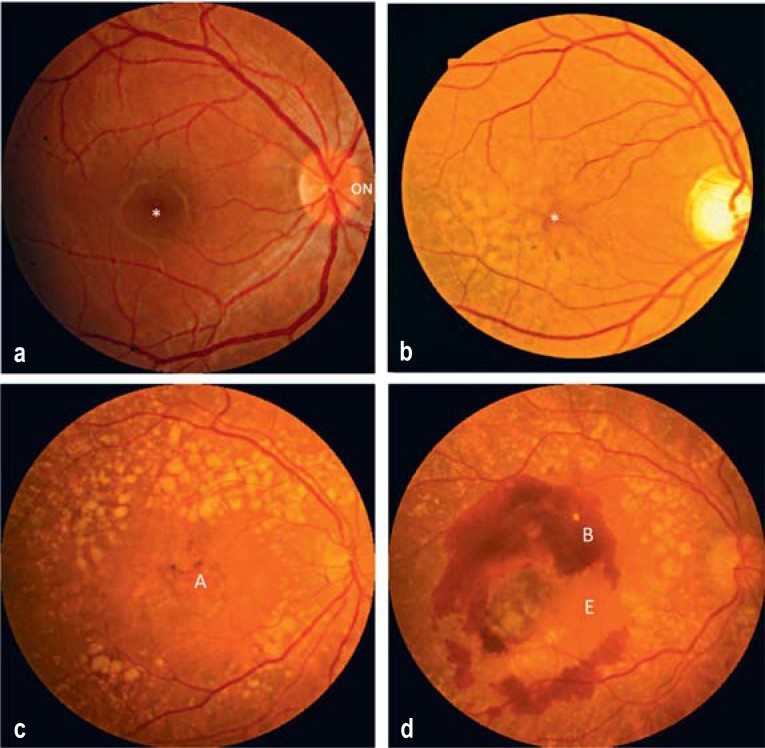
Figure 1. Stages of Age-Related Macular Degeneration (AMD)
Figure 1: Illustrating the progression of age-related macular degeneration from a normal juvenile macula to advanced stages, highlighting drusen formation, atrophic areas, and neovascularization.
- Normal juvenile macular image: Demonstrates the optic nerve (ON) on the right, where it enters the eye along with retinal arteries and veins. Blood vessels form an arc around the macula, which is free of larger vessels. The fovea (*), the center of the macula, still shows a physiological juvenile macular reflex.
- Early to intermediate AMD: Shows numerous, partly confluent drusen, mainly temporal to the fovea (*). Drusen are typically yellow deposits of non-degraded metabolic products under the retina.
- Dry (atrophic) form of late AMD: Displays many drusen along the vascular arcades and a punched-out area of atrophy (A) in the center.
- Wet form of late AMD (exudative or neovascular AMD): Illustrates abnormal blood vessel growth in the choroidal plexus below the retina, leading to exudation (E) and bleeding (B) into the macula.
Figure 2. Clinical Manifestations of Age-Related Macular Degeneration (AMD)
Figure 2: Amsler grid test illustrating normal vision versus vision affected by late-stage AMD with central scotoma and metamorphopsia.
- Amsler test with normal retinal findings: Shows straight lines and a central fixation point.
- Typical Amsler test findings in late AMD: Demonstrates a central fixation point obscured by a gray spot (central scotoma) and distorted lines (metamorphopsia).
Functional Impairment Due to AMD
AMD primarily affects the macula, leading to impaired central vision. This significantly impacts essential daily activities such as reading, driving, and facial recognition.
The exudative form of late AMD is typically associated with a more rapid and severe loss of vision compared to the atrophic form. Reading ability can deteriorate in just a few days. Untreated patients with exudative AMD may lose an average of three lines (15 letters) of visual acuity within two years. This visual loss in exudative AMD is caused by the development of choroidal neovascularization (CNV) in the macular area. These newly formed vessels are fragile and can rupture, causing hemorrhage into the macula followed by scarring. Pathological neovascularization is thought to be a misguided attempt by damaged retinal tissues to repair themselves, complicated by exudation or rupture of abnormal vessels and/or the retinal pigment epithelium, leading to rapid vision decline. Despite sharing an initial stage of retinal pigment epithelium degeneration, dry and exudative AMD follow distinct clinical courses. Transitional or mixed forms between these two main types are also observed.
eFigure 1. Normal Findings and Tear of Retinal Pigment Epithelium in Exudative Late AMD
eFigure 1: Comparison of normal retinal structure versus retinal pigment epithelium tear in exudative late AMD, showing hemorrhage and disrupted retinal layers.
Normal findings on funduscopy and optical coherence tomography (OCT) are shown in images (a, c) on the left.
Exudative late AMD findings are depicted in images (b, d) on the right, illustrating hemorrhage from a choroidal neovascularization membrane (CNV) along with a tear in the retinal pigment epithelium (RPE). The regular structure of retinal layers is disrupted, and central vision is significantly impaired due to hemorrhage and atrophy.
Risk Factors for Age-Related Macular Degeneration
The primary risk factor for AMD is age. Pathogenetically, it is hypothesized that the high metabolic activity of the macula places a significant cumulative burden on the retinal pigment epithelium throughout life to process and remove metabolic waste. AMD develops when retinal pigment epithelium cells can no longer meet this demand in older age. However, AMD is not necessarily linked to other typical aging-related diseases like osteoporosis. Besides age, other risk factors modulate both the onset and progression of AMD. The most significant modifiable risk factor is smoking. Smokers have a 2.6 to 4.8 times higher odds ratio of developing AMD compared to non-smokers. Former smokers also have an elevated odds ratio of 1.7 for AMD development.
Exudative Late AMD: An Aggressive Subtype
Exudative late AMD is the most aggressive form of the disease, often causing rapid central visual function deterioration due to pathological blood vessel growth, exudation, bleeding, fibrosis, and sometimes retinal pigment epithelium tears.
In addition to smoking, numerous genetic risk alleles for AMD have been identified in recent years. The two most prominent are polymorphisms in CFH (complement factor H) and ARMS2 (age-related maculopathy susceptibility 2). These two alleles together account for up to 45% of the risk of developing AMD. Furthermore, studies have shown associations between AMD and body mass index, cardiovascular disease, and arterial hypertension. Dyslipidemia and metabolic dysfunction have also been linked to AMD in some studies, but definitive causal relationships remain unclear. Drusen found in AMD resemble atherosclerotic deposits in vascular walls, commonly seen in cardiovascular high-risk patients. However, research into a potential association between AMD and atherosclerosis has yielded conflicting results to date.
Diagnostic Evaluation of AMD: A Comprehensive Approach
Patient history is crucial in identifying potential AMD. Patients with AMD often report sudden or gradual vision worsening in one or both eyes, which may become more pronounced in low light conditions. Inquiring about distorted vision (metamorphopsia) is important, as its presence strongly suggests macular disease. Metamorphopsia can become apparent when patients look at straight lines like road stripes, windowpanes, or tiles. Many patients also report facial distortion or size differences in images between their eyes. However, patients may remain asymptomatic for extended periods, especially in early stages or if the fovea is not yet involved. Therefore, proper AMD diagnosis necessitates a comprehensive ophthalmological examination, including best-corrected visual acuity measurement, funduscopy with dilated pupils, macular layer imaging with optical coherence tomography (OCT), and, in certain cases, fluorescein angiography, particularly when exudative AMD treatment is considered.
Genetic Predisposition to AMD
Besides age and smoking, genetic risk alleles in complement system genes and the ARMS2 HTRA locus are major risk factors for AMD onset and progression.
OCT has become a pivotal tool in AMD diagnosis. (It received a reimbursable procedure code in Germany in 2019). OCT is non-invasive and easily performed on most patients. Maintaining minimal standards of OCT image quality is crucial to avoid missing subtle but clinically significant changes. It is important to note that neither OCT alone, nor OCT angiography, can completely replace traditional fluorescein angiography in differentiating between dry and exudative late AMD. Fluorescein angiography uniquely reveals active exudation from pathological blood vessels into the retinal parenchyma.
eFigure 2. Fluorescein Angiography for the Diagnosis of Exudative AMD
eFigure 2: Fluorescein angiography demonstrating normal macular findings versus a classic CNV membrane with leakage in exudative AMD.
a) Normal fluorescein-angiographic findings in a healthy macula.
b) A classic CNV membrane in exudative AMD, clearly showing pathological vessels.
c) Angiographic late phase documenting significant leakage from pathological blood vessels of the CNV membrane into the macular parenchyma.
CNV, choroidal neovascularization
Treatment Strategies for AMD
The appropriate AMD treatment is determined by the disease stage. Risk factor modification, especially smoking cessation, is crucial at all stages. Numerous prospective population-based studies have demonstrated that smokers face a higher risk of AMD progression even after diagnosis. A Korean study further indicated that visual acuity improvement with anti-VEGF treatment is less pronounced in smokers with exudative AMD compared to non-smokers. Early AMD detection can be a strong motivator for patients to adopt healthier lifestyle habits to slow disease progression.
Key Indicators for AMD Diagnosis
Typical symptoms suggestive of AMD include distorted vision (metamorphopsia), reduced visual acuity, and central scotoma.
Dietary supplements for AMD are extensively discussed. The AREDS trials provide the most robust clinical trial data in this area. AREDS-1 and AREDS-2 were long-term randomized, controlled trials evaluating the potential impact of dietary supplements on AMD progression. AREDS-1, published in 2001, showed that high-dose supplementation with vitamin C and E, beta-carotene, and zinc had a beneficial effect in patients with intermediate-stage AMD. However, this must be considered alongside findings from other studies linking high-dose beta-carotene and vitamin supplementation in smokers (current or former) to an increased cancer risk. A combination of vitamin E and beta-carotene was reported to increase lung cancer risk by 18% (95% confidence interval [3; 36%], p = 0.01), while vitamin A and beta-carotene supplementation was associated with a relative risk of 1.28 ([95% CI 1.04; 1.57]; p = 0.02). This led to changes in AREDS-2 dietary supplements, replacing beta-carotene with lutein/zeaxanthin and omega-3 fatty acids. The efficacy of AREDS-2 supplements was only demonstrated in patients with intermediate or late-stage AMD, and the effect sizes were smaller than those of smoking cessation. The odds ratio for AMD progression in AREDS-1 was 0.72 (99% CI [0.52; 0.98]). The hazard ratio in AREDS-2 was 0.89 (98.7% CI: [0.75; 1.06]) for lutein + zeaxanthin + omega-3 fatty acids (DHA + EPA). Supplementation showed only a minor effect on intermediate-stage AMD and no effect on early or late stages. Therefore, routine dietary supplements are not generally recommended for preventing AMD before intermediate stage signs appear. German ophthalmological societies advocate a balanced diet for primary prophylaxis, aligning with the recommendations of the German Nutrition Society (Deutsche Gesellschaft für Ernährung).
Nanosecond laser therapy for drusen has been explored as a treatment for intermediate-stage AMD, where large drusen are present but without atrophy or exudation. The LEAD trial investigated whether laser treatment of drusen could slow AMD progression in intermediate-stage patients. The primary endpoint was not met, showing no protective effect of laser treatment. In patients with reticular pseudodrusen, the disease progression was actually faster. Thus, retinal laser therapy is not recommended for dry AMD patients outside of controlled clinical trials.
Dry (Atrophic) Late AMD: Current Challenges
Essential Clinical Evaluation Techniques
Key diagnostic techniques include visual acuity testing, bilateral funduscopy with dilated pupils, optical coherence tomography (OCT), and, in some cases, fluorescein angiography. Patients can use an Amsler grid to self-detect early metamorphopsia.
Currently, there is no proven effective treatment for atrophic late AMD. Clinical trials, including recent studies on complement system modulators, have yielded negative results. Common reasons cited for these failures mirror those in other degenerative central nervous system diseases: treatment may be initiated too late in a disease cascade that has already reached a point of no return. At a certain stage, neural tissue, specifically retinal photoreceptors, is irreversibly lost, and no method has yet been found to prevent further photoreceptor loss at the periphery of atrophic macular regions. Current research in atrophic late AMD focuses on deepening our understanding of disease progression pathogenesis to identify promising therapeutic targets and optimal intervention timing.
Wet (Exudative) Late AMD: Advances in Treatment
Despite the aging population and increased AMD prevalence, statistics from Germany and other countries show a stagnation or even decrease in blindness and severe visual impairment rates. This is largely attributed to the introduction of effective treatment for exudative late AMD in 2005. In 2006, Science journal recognized anti-VEGF therapy for exudative macular degeneration as one of the top ten scientific breakthroughs of the year, following successful phase 3 clinical trials. This treatment involves injecting anti-VEGF drugs directly into the eye’s vitreous body (intravitreal administration). Four such drugs are available: bevacizumab (off-label, since 2005) and three approved in Europe—ranibizumab (approved 2007), aflibercept (approved 2012), and brolucizumab (approved 2020). Approved drugs cost approximately 1000 euros per injection, while bevacizumab is significantly less expensive. Bevacizumab is not expected to receive approval for intraocular use, and more biosimilar drugs are likely to become available soon.
Lifestyle Modifications for AMD Management
Smoking cessation is recommended for all AMD stages to prevent onset or progression. Dietary supplements have limited efficacy and are only beneficial in specific AMD stages.
Although anti-VEGF drugs vary in chemical structure, binding affinity, and specificity, they share a common mechanism: blocking vascular endothelial growth factor (VEGF). VEGF is a pro-angiogenic factor promoting pathological blood vessel formation in exudative AMD and a permeability factor facilitating blood plasma component extravasation into the retinal parenchyma. Sub- and/or intraretinal fluid accumulation from hyperpermeable choroidal vessels is a primary cause of vision worsening in exudative AMD. VEGF inhibitors largely work by reducing vessel permeability rather than inhibiting angiogenesis.
Anti-VEGF therapy typically requires repeated, long-term administration, especially in the initial treatment years. Patients must understand this and the associated logistical demands to ensure treatment compliance. AMD is a chronic condition where the underlying pathogenic cascade cannot be stopped early with causal treatment, even in treatable stages. Effective management requires intensive, consistent, long-term treatment.
Various established anti-VEGF therapy strategies exist for exudative AMD. Initial clinical trials tested regular monthly VEGF inhibitor administration via intravitreal injection, an ambulatory surgical procedure. Each injection carries a roughly 0.029% (1:3500) risk of severe intraocular infection (endophthalmitis). Endophthalmitis is treated with vitrectomy and intraocular antibiotics, with variable and often poor visual outcomes depending on the pathogen and other factors. Other rare but relevant risks of intravitreal therapy include sterile inflammatory reactions (0.09–2.9%) and, very rarely, retinal detachment (0.013%).
Intravitreal Anti-VEGF Therapy Options
Currently, four anti-VEGF drugs are available: bevacizumab (off-label), ranibizumab, aflibercept, and brolucizumab, with the latter three approved for use in Europe.
To personalize treatment, “pro re nata” (PRN) and “treat and extend” strategies have been developed and are now preferred over regular monthly injections at most AMD treatment centers. These strategies aim to provide patients with the optimal number of injections based on individual needs. Clinical trials show most patients need about 7–8 injections in the first treatment year to effectively control exudative AMD, with fewer injections often required in subsequent years. Real-world care studies in Germany indicate a greater risk of under-treatment than over-treatment. In practice, treatment intervals are often suboptimal due to comorbidities, transport difficulties, etc. Numerous studies show that undertreatment phases are often linked to irreversible vision loss. Ensuring uninterrupted treatment is a vital therapeutic goal. Proper management, based on disease activity, includes intravitreal injections and/or follow-up visits with visual acuity measurement, funduscopy, and OCT.
In conclusion, current exudative late AMD treatment, while not addressing the underlying etiology, effectively stabilizes visual acuity in many patients. Visual acuity remains stable in over 70% of treated eyes, with nearly 20% showing marked improvement after initial treatments. However, anti-VEGF therapy is ineffective in early, intermediate, and atrophic late AMD. Accurate AMD subtype diagnosis is crucial for timely treatment initiation in exudative late AMD, ideally before irreversible visual loss occurs.
Atrophic Late AMD: Unmet Treatment Needs
No evidence-based treatment is yet available for atrophic late AMD, but several approaches are under investigation in clinical trials.
Exudative Late AMD Treatment Summary
Exudative late AMD is treated with intravitreally injected anti-VEGF drugs. Most patients require multiple injections, typically 7 to 8, in the first year of treatment.
CME and Further Learning
- CME certification is available online at cme.aerzteblatt.de until 19 July 2021.
- New CME units are accessible for 12 months. Answers are available online four weeks after publication. Check website for submission deadlines.
- This article is certified by the North Rhine Academy for Continuing Medical Education. CME points can be managed with the 15-digit “uniform CME number” (EFN) found on the CME card. Register at www.aerzteblatt.de (“Mein DÄ”) or enter in “Meine Daten” and agree to result communication.
CME Credit Questions (Answer online at cme.aerzteblatt.de by 19 July 2021)
Question 1: AMD’s impact on vision loss and blindness?
Question 2: Main risk factors for AMD?
Question 3: Stages of AMD?
Question 4: Typical symptom of advanced AMD?
Question 5: Morphological correlate of AMD vision loss?
Question 6: Diagnostic technique for active leakage in exudative AMD?
Question 7: Recommended lifestyle change for AMD patients?
Question 8: Treatment for atrophic late AMD?
Question 9: Treatment for exudative late AMD?
Question 10: Typical signs of postoperative endophthalmitis?
Acknowledgements
Translated from German by Ethan Taub, M.D.
Grateful to Prof. Hansjürgen Agostini and Prof. Daniel Pauleikhoff for review and comments, and to Prof. Clemens Lange and Dr. Bastian Grundel for providing images.
Footnotes
Conflict of interest statement:
Prof. Stahl has consulted for Novartis and Bayer and received fees from Allergan, Bayer, and Novartis for meetings and CME sessions.
References
(References from original article would be listed here)