CONTENTS
Diagnosis of Metabolic Acid-Base Disorders
Anion Gap (AG)
Elevated Lactate (Hyperlactatemia)
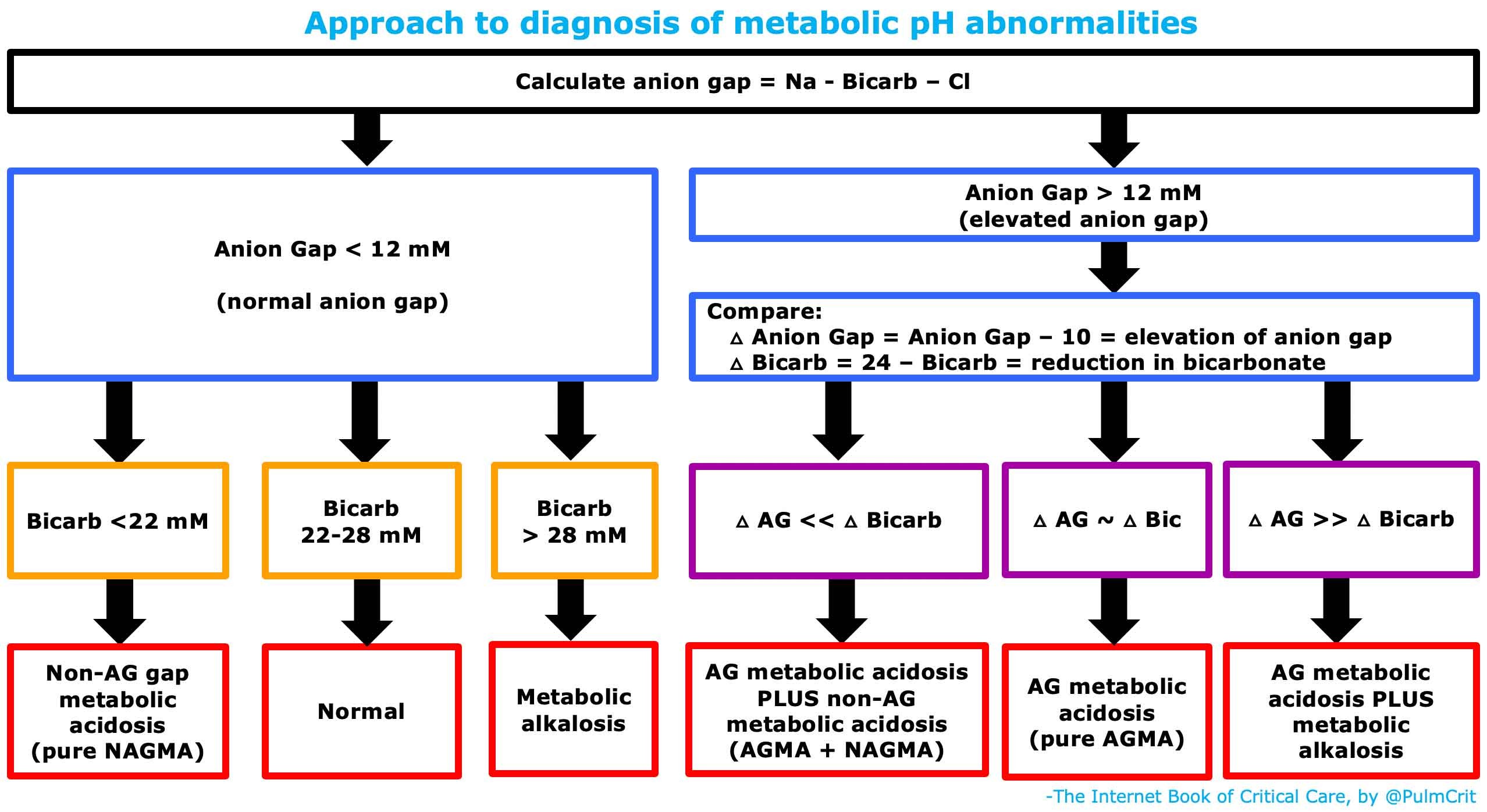
### Diagnostic Approach to Metabolic pH Abnormalities
Metabolic disorders are characterized by a primary shift in serum bicarbonate and/or the anion gap. These parameters should be evaluated whenever electrolytes are measured.
[1] Determination of the Anion Gap for Anion Gap Metabolic Acidosis (AGMA) Evaluation
- The anion gap should be a routine calculation in every electrolyte panel. Ideally, this process is automated within the laboratory system. If not, it must be manually calculated.
- The anion gap is calculated using the formula: (Na – Cl – Bicarb). This is a straightforward calculation without adjustments for glucose, albumin, potassium, or other factors.
- An elevated anion gap is a significant clinical concern because it is frequently associated with life-threatening conditions. This contrasts with non-anion gap metabolic acidosis, where the underlying causes are often less immediately critical.
[2] Normal Anion Gap: Assess Bicarbonate Levels
- Bicarbonate Levels
- A bicarbonate level exceeding 28 mM in the context of a normal anion gap indicates pure metabolic alkalosis.
- A bicarbonate level between 22-28 mM with a normal anion gap is generally considered to represent a normal metabolic pH status.
[3] Elevated Anion Gap: Delta Gap and Delta Bicarbonate Analysis
- Understanding Delta Delta
- Delta Anion Gap = (Anion Gap) – 10. This value approximates the degree of elevation above the normal anion gap.
- Delta Bicarbonate = 24 – bicarbonate. This value represents the degree of reduction from the normal serum bicarbonate level.
- Comparing the delta anion gap and delta bicarbonate can help identify the presence of additional acid-base disturbances alongside anion gap metabolic acidosis. Specifically:
- (1) If the delta anion gap is roughly equal to the delta bicarbonate, it suggests a pure anion gap metabolic acidosis without other concurrent metabolic processes. This is the expected finding in isolated AGMA.
- (2) If the delta anion gap is significantly higher than the delta bicarbonate, it indicates a combined anion gap metabolic acidosis and metabolic alkalosis. The additional metabolic alkalosis is elevating the bicarbonate level, masking the full extent of bicarbonate reduction from AGMA alone.
- (3) If the delta anion gap is significantly lower than the delta bicarbonate, it suggests a combined anion gap metabolic acidosis and non-anion gap metabolic acidosis. The additional non-anion gap acidosis is further decreasing the bicarbonate level beyond what is expected from AGMA alone.
### Respiratory pH Analysis: Clinical Relevance
Arterial blood gas (ABG) or venous blood gas (VBG) analysis provides information about respiratory pH abnormalities. While traditionally considered a mandatory component of pH analysis, the clinical utility of respiratory analysis can vary. Blood gas analysis primarily addresses two key questions:
[#1] Adequacy of Respiratory Compensation
- In patients with metabolic pH disorders, respiratory compensation is expected. This physiological response is demonstrated below:
- Metabolic alkalosis is typically compensated by mild hypoventilation (secondary respiratory acidosis).
- Metabolic acidosis is usually compensated by hyperventilation (secondary respiratory alkalosis).
- By assessing whether a patient’s respiratory response aligns with these expected compensatory mechanisms, clinicians can evaluate the integrity of their ventilatory function. Discrepancies may indicate proportionally inadequate or excessive respiratory compensation.
- However, in critically ill patients, ventilation is often influenced by a multitude of factors, including anxiety, opioid use, and mechanical ventilation settings. Compensation equations are historically derived from studies of relatively stable patients without complex medical issues. Their accuracy in the context of modern critically ill patients, who often present with multiple active medical problems, is uncertain.
- Consequently, the direct impact of respiratory compensation information on the management of critically ill patients is often ambiguous. Decisions concerning intubation or respiratory support strategies are primarily guided by clinical assessment and underlying diagnosis, rather than solely by blood gas values.
[#2] Detection of Primary Respiratory Disorders
- When metabolic pH abnormalities are detected, it is crucial to consider whether they might be secondary compensatory responses to an underlying primary respiratory disorder. The table above illustrates these relationships on the right side. Specifically:
- (a) Metabolic acidosis could be a compensatory response to chronic respiratory alkalosis.
- (b) Metabolic alkalosis could be a compensatory response to chronic respiratory acidosis.
- Differentiating between primary metabolic disorders and metabolic compensation for respiratory disorders is essential for accurate diagnosis and appropriate management.
- From a clinical perspective:
- (a) Metabolic acidosis as a result of chronic respiratory alkalosis is exceptionally rare in the absence of iatrogenic mechanical ventilation mismanagement. It is an uncommon clinical scenario.
- (b) Metabolic alkalosis secondary to chronic respiratory acidosis is frequently observed in patients with hypercapnia from various causes, most commonly chronic obstructive pulmonary disease (COPD), obesity hypoventilation syndrome, or chronic opioid use. This diagnosis is typically supported by a compatible clinical history and review of previous laboratory results demonstrating chronic metabolic alkalosis.
Clinical Significance of Blood Gas Analysis in pH Assessment
- Blood gas analysis often does not fundamentally alter the diagnosis and management of a patient’s acid-base status, particularly when a thorough electrolyte panel evaluation and comprehensive clinical assessment have been performed.
- In some instances, blood gas analysis may reveal chronic respiratory acidosis as the etiology of metabolic alkalosis (2b). However, in many such cases, this would already be clinically suspected based on patient history or prior laboratory findings.
- Therefore, blood gas analysis is not universally mandatory for acid-base status assessment. It can be strategically employed on a selective basis, rather than routinely ordered for all patients with pH abnormalities. This approach is preferable for several reasons:
- (1) Acid-base disturbances are highly prevalent among critically ill patients, potentially affecting a majority. Indiscriminate blood gas analysis for every acid-base abnormality would lead to a substantial increase in testing, causing patient discomfort and increased healthcare costs.
- (2) Treatment decisions can often be effectively guided by electrolyte panel results alone (metabolic acid-base analysis). This streamlines clinical management and avoids delays associated with blood gas analysis.
- While French guidelines advocate for arterial blood gas measurement in all patients with reduced plasma bicarbonate levels, these recommendations lack robust, high-quality evidence. (31418093)
### Alternative Approaches to pH Analysis
- Numerous alternative methodologies exist for pH analysis, many of which are considerably more complex, such as Stewart’s acid-base approach. However, different analytical strategies generally converge on similar clinical conclusions.
- The most effective approach is one that is well-understood and consistently applied. The method described herein is straightforward and widely adopted by clinicians, facilitating interdisciplinary communication. If a clinician is already proficient with another method, there is no compelling need to change.
- The primary clinical pitfall is neglecting pH analysis altogether, for example, failing to recognize an elevated anion gap.
### Etiologies of Single-Digit Bicarbonate Levels
Specific etiologies associated with very low serum bicarbonate levels (36796238). This list is not exhaustive and should not replace a comprehensive differential diagnosis.
- Liver failure
- Post-cardiac arrest
- Severe ketoacidosis:
- Diabetic ketoacidosis (DKA)
- Alcoholic ketoacidosis (AKA)
- Toxicological causes:
- Toxic alcohol ingestion
- Metformin poisoning
- Salicylate intoxication
- Cyanide toxicity (36796238)
### Anion Gap in Detail
Introduction to the Anion Gap
Basic Calculation and Interpretation
- Anion Gap (AG) = Na – Bicarb – Chloride
- The normal anion gap range is approximately 4-12 mM.
- Historically, the normal range for the anion gap was often cited as higher, up to ~16 mM. However, with modern electrolyte analyzers, the upper limit of normal has decreased to ~11-12 mM. (24766940)
- Normal ranges may vary slightly between hospital laboratories.
- Comparison to a baseline anion gap, if available, is clinically valuable. For instance:
- [1] Patients with chronic renal insufficiency may have a chronically elevated anion gap. If the current anion gap is consistent with their chronic level, it is less concerning.
- [2] A rapidly increasing anion gap over time is worrisome and warrants thorough investigation.
Albumin Correction is Unnecessary 
- Albumin, a negatively charged protein, theoretically contributes to the anion gap.
- Many acid-base textbooks and resources recommend correcting the anion gap for albumin levels. However, in clinical practice, this correction is often not performed due to the frequent unavailability of albumin measurements.
- While albumin correction has theoretical validity, it lacks supporting clinical evidence.
- Albumin correction typically increases the anion gap by approximately 4 mM. Consequently, if albumin correction is applied, a higher cutoff value is necessary to identify anion gap metabolic acidosis.
- Using an uncorrected anion gap with a cutoff of >10 mM demonstrates equivalent diagnostic performance for detecting anion gap metabolic acidosis compared to using an albumin-corrected anion gap with a higher cutoff of ~14 mM. (16858097, 18431828, 19087326) Different cutoff values may affect sensitivity and specificity, but the overall test performance, as measured by the area under the Receiver Operator Curve, remains unchanged. Therefore, albumin correction represents an additional step that does not enhance the clinical utility of the anion gap test.
- While albumin correction is permissible, a higher cutoff value must be used if implemented.
Performance of Anion Gap in Lactic Acidosis Detection
- The anion gap is not a reliable indicator for detecting mild lactic acidosis (e.g., lactate levels of 2-4 mM). The normal anion gap range spans approximately 10 mM. A patient with a baseline anion gap at the lower end of the normal range (e.g., 4 mM) could develop significant lactic acidosis while still maintaining an anion gap within the normal range. (17699401)
- Studies vary regarding the precise sensitivity and specificity of the anion gap for lactate detection, which likely also varies between laboratories. One study reported that an anion gap >10 mM detected a lactate level >2.5 mM with 63% sensitivity and 65% specificity. (19087326)
- If lactic acidosis is specifically suspected, direct lactate measurement is the optimal diagnostic strategy. The anion gap cannot reliably exclude elevated lactate. However, it remains a valuable surveillance tool for identifying marked lactic acidosis.
Causes of High Anion Gap
Ketoacidosis
- Diabetic Ketoacidosis (DKA)
- Alcoholic Ketoacidosis (AKA)
- Starvation Ketoacidosis
Uremic Acidosis
- Occurs with reduced glomerular filtration rate (GFR)
- Uncomplicated uremia typically does not cause bicarbonate levels to fall below ~12-15 mM or the anion gap to exceed >20 mM. If these more extreme values are observed, consider alternative or additional underlying conditions.
Hyperlactatemia
Medication and Substance-Related Causes
- Inhalants (carbon monoxide, cyanide, toluene)
- Iron
- Isoniazid
- NSAIDs (Non-Steroidal Anti-Inflammatory Drugs)
- Salicylates
- Sympathomimetics
- Toxic alcohols (methanol, ethylene glycol, diethylene glycol, propylene glycol)
- High-dose penicillins
- Colchicine
- 5-Oxoproline (pyroglutamic acidosis) associated with therapeutic acetaminophen use (35461626). Typically occurs in malnourished, ill women, leading to glutathione and cysteine depletion. Acetaminophen discontinuation usually results in rapid resolution. (34400023)
Other Causes of High Anion Gap
- Hyperphosphatemia
- Metabolic alkalosis (increases the negative charge on albumin)
- D-Hyperlactatemia: Resulting from small intestinal resection or malabsorption leading to carbohydrate delivery to the colon and fermentation by gut bacteria. Standard laboratory lactate tests do not detect D-lactate. D-hyperlactatemia can cause neurological symptoms, including confusion, ataxia, incontinence, and nystagmus. (34400023)
Evaluation of High Anion Gap
Standard Initial Evaluation
- Repeat laboratory blood draw with simultaneous measurement of:
- [1] Electrolytes
- [2] Lactate level
- [3] Beta-hydroxybutyrate
- Repeating electrolyte measurement helps exclude laboratory error or random variation.
- Simultaneous testing of these parameters facilitates rapid and definitive clinical management.
Additional Tests to Consider
- In suspected poisoning cases: Evaluate for salicylates, acetaminophen, carbon monoxide, and toxic alcohols.
Clinical Management of High Anion Gap
Treatment for an elevated anion gap is directed at the underlying cause. While diagnostic evaluation is underway, the following management strategies may be considered:
Bicarbonate Therapy
- Uremic AGMA:
- Bicarbonate administration has been traditionally used to support pH levels in uremic acidosis, potentially delaying the need for dialysis.
- The BICAR-ICU trial supported the use of bicarbonate in uremic metabolic acidosis, aiming to reduce dialysis requirements.
- Other causes of AGMA (e.g., hyperlactatemia or diabetic ketoacidosis):
- Bicarbonate therapy has no proven evidence-based role in these conditions, regardless of pH level.
Hemodialysis
- May be indicated in metabolic acidosis associated with renal failure, particularly when volume overload is present, which contraindicates intravenous bicarbonate administration.
- The optimal timing for dialysis initiation remains debated (early vs. late dialysis).
- French guidelines suggest considering dialysis for refractory acidemia (e.g., pH <7.15). (31418093) However, the overall clinical context is often more informative than any specific pH cutoff.
- Earlier dialysis may be warranted in specific intoxications (e.g., metformin, ethylene glycol, methanol, or salicylate).
Compensatory Hyperventilation
- For intubated, mechanically ventilated patients, targeting a lower pCO2 than usual may be reasonable.
- The objective is to mimic physiological compensatory respiratory alkalosis, a function assumed by the clinician in mechanically ventilated patients.
- The degree of hyperventilation should be balanced against other physiological considerations:
- In patients with ARDS (Acute Respiratory Distress Syndrome) or obstructive lung disease, achieving a low pCO2 may be challenging or detrimental.
- In patients with severe metabolic acidosis and hemodynamic instability, there might be a stronger rationale to improve pH by reducing pCO2. Conversely, if the acidosis is clinically well-tolerated, aggressive ventilator adjustments may be less necessary.
Low Anion Gap
A low anion gap is typically defined as an anion gap ≤3 mM. (37783490)
Causes of Low Anion Gap:
- Laboratory Error:
- Possibly the most common cause.
- May occur in conditions such as:
- (1) Severe hypernatremia
- (2) Pseudohyponatremia (e.g., due to hyperlipidemia)
- (3) Blood draw proximal to an intravenous infusion site.
- Increased Cation Levels:
- Hyperkalemia
- Hypercalcemia
- Hypermagnesemia (more commonly associated with MgCl2 or MgBicarb than MgSO4)
- Elevated Immunoglobulin Levels:
- Multiple myeloma
- Polyclonal gammopathy (variable effects on anion gap)
- Pseudohyperchloremia: Falsely elevated chloride levels may be caused by:
- Other Medications:
- Polymyxin B
- Ammonium chloride administration
- Lithium overdose/poisoning
- Low Albumin Level: (Mild effect on anion gap reduction)
Initial Management Considerations
- Consider the differential diagnoses of low anion gap within the patient’s clinical context.
- Compare current electrolytes with recent or baseline values:
- Is the low anion gap chronic? (Suggestive of chronic conditions like elevated immunoglobulins)
- Rapid shifts in sodium or chloride levels may indicate laboratory error during blood draw.
- Is the chloride level exceptionally high? Consider pseudohyperchloremia.
- Is the sodium level low? Consider pseudohyponatremia.
Laboratory Investigation Panel
- Initial investigation may include:
- Electrolytes (repeat measurement)
- Calcium, magnesium levels
- Salicylate level
- Lithium level (if lithium toxicity is possible)
- Albumin level
- Point-of-care electrolyte panel (for rapid assessment of pseudohyponatremia)
- Further investigations to consider:
- SPEP (Serum Protein Electrophoresis)
- Serum osmolality (for pseudohyponatremia evaluation)
Rising Anion Gap (Delta AG) in Hospitalized Patients
- Frequent electrolyte monitoring in hospitalized patients allows for tracking anion gap trends over time.
- A rapidly increasing anion gap can be an early indicator of ketoacidosis or lactic acidosis, even if the absolute anion gap value remains within the normal range. The normal range is relatively broad (e.g., 5-14 mM), so a patient with a low baseline anion gap may develop substantial lactic acidosis without exceeding the upper limit of normal.
Defining a Significant Rise in Anion Gap
- The threshold for a clinically significant rise in anion gap is not precisely defined.
- Some studies indirectly suggest that changes of ≥5 or ≥8 mM are clinically relevant. (35568886, 23190721)
- Additional factors should be considered in determining the need for further investigation:
- Presence of other subtle signs of clinical instability.
- Deviation of the current anion gap from the patient’s prior trends.
Causes of Rapidly Rising Anion Gap in Hospitalized Patients
- The differential diagnosis for a rapidly increasing anion gap in hospitalized patients is relatively narrow:
- [1] Laboratory artifact or inaccurate electrolyte measurement.
- [2] Lactic acidosis
- [3] Ketoacidosis (e.g., starvation or diabetic ketoacidosis)
- Most other causes of elevated anion gap develop more gradually (e.g., uremia) or are less likely to arise acutely in a hospital setting (e.g., overdose).
Investigation of Rapidly Rising Anion Gap
- Obtain repeat laboratory tests with simultaneous measurement of electrolytes, lactate, and beta-hydroxybutyrate levels.
- (Further discussion on evaluating elevated anion gap:
)
### Elevated L-Lactate (Hyperlactatemia)
Definition of Elevated Lactate
- Normal lactate levels are approximately 0.5-2 mM.
- Levels >2 mM are considered elevated.
- Levels >4 mM are increasingly concerning for occult shock or serious illness, although the differential diagnosis is broad.
Mechanisms of Increased Lactate Production
- Increased glycolysis and pyruvate production:
- Intravenous glucose administration
- Beta-2 adrenergic agonist activity (also inhibits pyruvate formation from lactate)
- Metabolic or respiratory alkalosis (increases phosphofructokinase activity). Clinically rare, sometimes termed “lactic alkalosis.”
- Increased NADH/NAD+ ratio (NAD+ deficiency):
- Mitochondrial dysfunction due to hypoxemia
- Mitochondrial dysfunction from other causes (e.g., medications)
- Low insulin levels or insulin resistance (e.g., ketoacidotic states)
Causes of Hyperlactatemia
Type A: Generalized or Regional Tissue Hypoxia
- Shock of any etiology, including:
- Septic shock
- Cardiogenic shock
- Obstructive shock (e.g., pulmonary embolism)
- Hypovolemic shock
- (Note: This is an oversimplification; systemic oxygen delivery is often preserved. Further discussion here).
- Regional hypoperfusion:
- Ischemic limb
- Mesenteric ischemia
- Muscle hyperactivity:
- Generalized seizure
- Extreme exertion
- Extreme anemia (e.g., (Reddi 2020))
- Systemic hypoxemia
Type B1: Systemic Diseases
- Liver failure
- Malignancy (typically leukemia/lymphoma)
- Thiamine deficiency (e.g., chronic critical illness, poor nutritional status, gastric bypass surgery)
- Alcoholic ketoacidosis
- Lactic alkalosis
Type B2: Drugs
- Beta-agonist excess:
- Albuterol
- Terbutaline
- Epinephrine
- Linezolid
- Metformin
- Propofol (Propofol Infusion Syndrome)
- Propylene glycol intoxication:
Type B2: Toxins
- Acetaminophen poisoning (massive)
- Alcohols:
- Carbon monoxide
- Cyanide
- Salicylate
- Sympathomimetics (cocaine, amphetamine, cathinones)
- Toluene
- Iron
Type B3: Inherited
- MELAS (Mitochondrial Encephalomyopathy, Lactic Acidosis, and Stroke-like episodes)
Evaluation of Hyperlactatemia to Determine Cause
[1] Medication Review
- Review the patient’s medication list for potentially problematic medications listed above.
- Discontinue any medications suspected of contributing to acidosis, especially propofol, nitroprusside, or metformin, if clinically appropriate.
[2] Patient Examination for Shock Features
- Assess for signs of shock:
(tachycardia, oliguria, hypotension, altered mental status, impaired peripheral perfusion).
- If shock signs are present, initiate resuscitation immediately without delay.
- When uncertain, assume hyperlactatemia is clinically significant and requires prompt attention.
- POCUS (Point-of-Care Ultrasound): Evaluate for less common shock etiologies (e.g., massive pulmonary embolism, cardiac tamponade).
- Assess for focal ischemia: (abdominal pain, cold extremity).
[3] Laboratory Panel
- Repeat lactate level and electrolytes: (if there is any doubt about the accuracy of initial results).
- Note: Occasionally, falsely elevated lactate measurements may occur due to blood draws upstream from lactated Ringer’s infusions.
- Venous lactate measurement is clinically acceptable. (Arterial lactate is the technical reference standard, but venous values are highly correlated, and minor lactate differences should not dictate clinical decisions).
- Complete Blood Count (CBC): (if not recently available):
- Elevated white blood cell count or neutrophil-to-lymphocyte ratio may indicate systemic inflammation, such as sepsis.
- Severe anemia can contribute to hyperlactatemia (but typically requires extreme degrees of anemia).
- Liver Function Tests (LFTs): Hepatic insufficiency can directly cause or exacerbate hyperlactatemia.
- Sepsis Evaluation: If sepsis is considered, obtain:
- Blood cultures
- Imaging studies (e.g., chest radiograph +/- CT scans)
- Procalcitonin and/or C-reactive protein levels
- (Further discussion of sepsis management:
)
- Suspected Poisoning: Consider testing for:
- Acetaminophen
- Carbon monoxide
- Salicylates
- Toxic alcohols
Treatment of Hyperlactatemia
- Treat underlying causes of hyperlactatemia.
- Review and discontinue any potentially causative medications.
- Empiric intravenous thiamine repletion if deficiency is possible:
- Thiamine deficiency may be underdiagnosed, particularly in critically ill patients with prolonged ICU stays.
- Thiamine deficiency testing is impractical. Empiric thiamine administration is safe and reasonable when deficiency is suspected.
- Insulin for insulin-deficient states:
- Insulin enhances pyruvate dehydrogenase complex (PDC) activity, theoretically reducing lactate levels.
- Insulin administration may be considered for hyperglycemic patients with borderline ketoacidosis. (Reddi 2020)
- Diuresis or exogenous alkali administration:
- Dialysis is generally ineffective for hyperlactatemia management. In vivo, lactic acid is rapidly produced and metabolized. Dialysis cannot remove lactate quickly enough to significantly alter this balance. However, dialysis might be considered as a temporizing measure in severe acidemia.
- Bicarbonate therapy has no evidence-based role in treating elevated lactate levels.
### Podcast
Follow us on iTunes
The Podcast Episode
Want to Download the Episode?Right Click Here and Choose Save-As
### Questions & Discussion
For questions and discussions related to this content, please visit here.
- Always calculate the anion gap when evaluating electrolyte panels, even if your laboratory system does not automatically provide it.
- An elevated anion gap in a critically ill patient should be presumed to be lactic acidosis and indicative of a life-threatening condition until proven otherwise.
- An elevated anion gap should be treated as a marker of a serious underlying abnormality until a definitive diagnosis is established. In acutely ill patients, strongly consider hyperlactatemia as a potential cause of an elevated anion gap and initiate evaluation for hyperlactatemia promptly, even before lactate results are available.
- In cases of elevated lactate with unclear etiology, consider empiric intravenous thiamine. Thiamine deficiency is common in critical illness, and thiamine administration is safe.
- Do not be overly concerned about elevated lactate following a generalized seizure. This is typically benign and should resolve within approximately one hour. Monitor closely to ensure clinical improvement.
- Do not equate lactic acidosis solely with septic shock. Lactic acidosis has a broad differential diagnosis encompassing numerous conditions, including all types of shock.
Guide to Emoji Hyperlinks
- = Link to online calculator.
- = Link to Medscape monograph about a drug.
- = Link to IBCC section about a drug.
- = Link to IBCC section covering that topic.
- = Link to FOAMed site with related information.
- = Link to supplemental media.