The advent of artificial intelligence (AI) has dramatically transformed various sectors, and healthcare is no exception. Within medical imaging, AI is proving to be an invaluable asset, particularly in ophthalmology. Automated analysis of retinal images holds immense potential for early and accurate diagnosis of various eye diseases. This article delves into the critical datasets that underpin the development of RETFound, a significant advancement in AI-driven retinal diagnosis, exploring its architecture, implementation, and implications for the future of eye care.
The Foundation of RETFound: Extensive Datasets for AI Learning
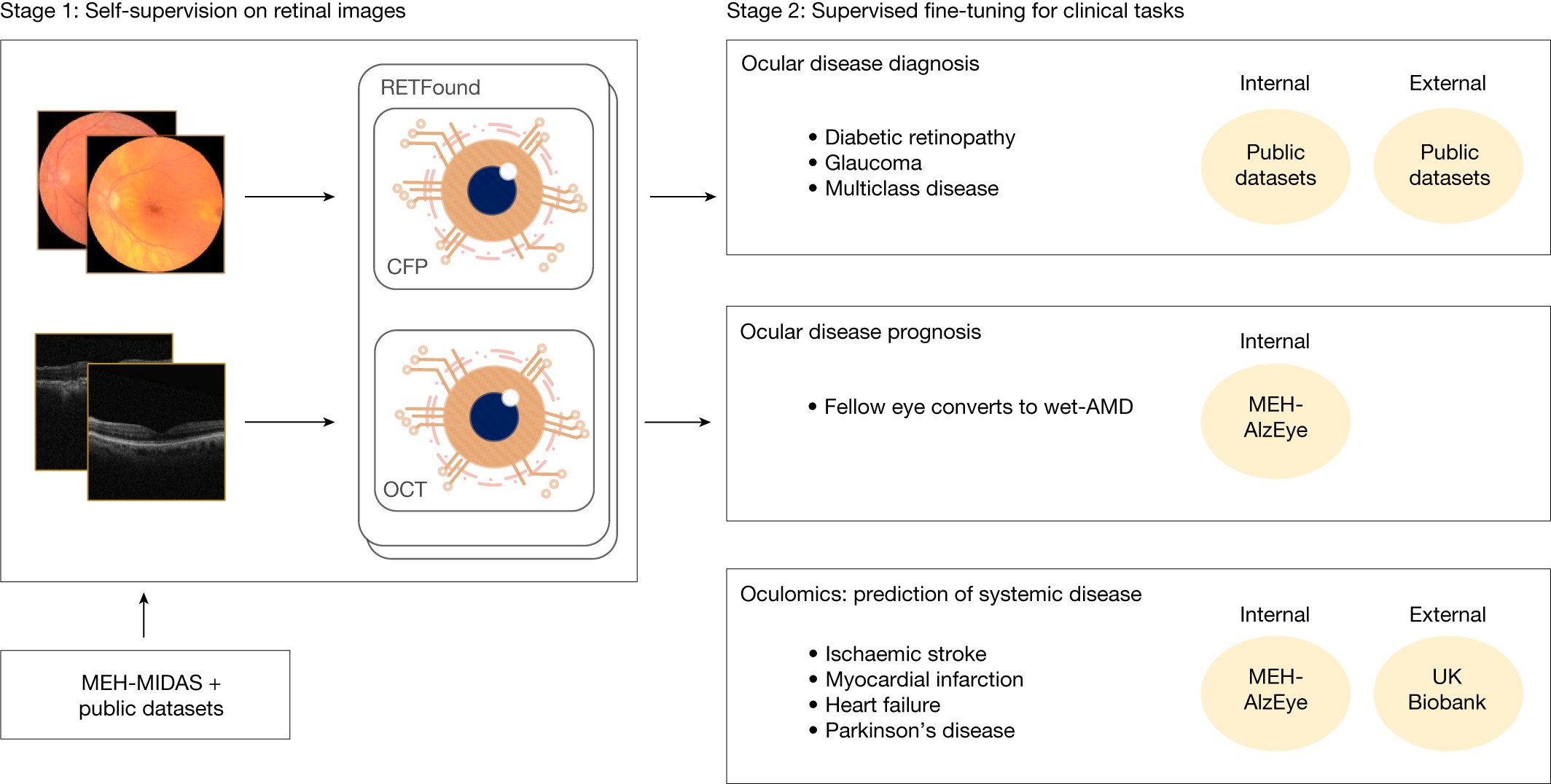
The power of AI models hinges on the quality and quantity of data they are trained on. RETFound, a groundbreaking model in the realm of Ai Retina Diagnosis, is built upon an enormous collection of unannotated retinal images. This vast dataset comprises 904,170 Color Fundus Photographs (CFPs) and 736,442 Optical Coherence Tomography (OCT) scans, establishing a robust foundation for self-supervised learning (SSL).
A significant portion of these images, specifically 815,468 CFPs and 627,133 OCTs (representing over 85-90%), are sourced from the Moorfields Diabetic imAge dataSet (MEH-MIDAS). MEH-MIDAS is a comprehensive retrospective dataset encompassing the complete ocular imaging records of 37,401 patients with diabetes, collected at Moorfields Eye Hospital in London between 2000 and 2022. This dataset is rich in diversity, reflecting a wide age range (mean 64.5 years, standard deviation 13.3) and varied ethnicities including British, Indian, Caribbean, and African. Furthermore, MEH-MIDAS incorporates images from diverse imaging devices such as Topcon 3DOCT-2000SA, CLARUS (ZEISS), and Triton (Topcon).
Alt Text: Data sources for RETFound, highlighting the Moorfields Eye Hospital Diabetic Retinopathy Dataset (MEH-MIDAS) and other datasets used in AI retina diagnosis research.
Complementing MEH-MIDAS, the dataset includes 88,702 CFPs from Kaggle EyePACS and 109,309 OCTs from a study by Kermany et al., further enriching the diversity of imaging devices to include Centervue DRS, Optovue iCam, Canon CR1/DGi/CR2, Topcon NW, and SPECTRALIS (Heidelberg). This extensive and diverse dataset is crucial for training a robust and generalizable AI model for ai retina diagnosis.
Validating RETFound: Datasets for Ocular Disease Diagnosis
To rigorously evaluate the performance of RETFound in ai retina diagnosis, the model was tested across three distinct categories of disease detection tasks. The first category focused on diagnostic classification of ocular diseases using publicly accessible ophthalmic data.
For diabetic retinopathy diagnosis, a critical application of ai retina diagnosis, datasets like Kaggle APTOS-2019 (India), IDRID (India), and MESSIDOR-2 (France) were utilized. These datasets are labeled according to the International Clinical Diabetic Retinopathy Severity scale, a five-stage system ranging from no diabetic retinopathy to proliferative diabetic retinopathy.
Glaucoma, another leading cause of blindness, was assessed using PAPILA (Spain) and Glaucoma Fundus (South Korea) datasets. These datasets categorize glaucoma into non-glaucoma, early glaucoma (suspected glaucoma), and advanced glaucoma, enabling the evaluation of RETFound’s capability in glaucoma detection and severity classification within ai retina diagnosis frameworks.
Alt Text: Overview of datasets used for evaluating RETFound’s performance in ocular disease diagnosis, including diabetic retinopathy and glaucoma detection within AI retina diagnosis.
Datasets encompassing multiple diseases, such as JSIEC (China), Retina, and OCTID (India), were also incorporated. JSIEC provides images with 39 categories of common referable fundus diseases and conditions, offering a broad spectrum for testing ai retina diagnosis models. Retina dataset includes labels for normal, glaucoma, cataract, and retina disease, while OCTID provides OCT scans labeled with normal, macular hole, AMD, central serous retinopathy, and diabetic retinopathy.
The grading protocols for these public datasets varied, ranging from consensus grades by multiple medical experts (IDRiD, MESSIDOR-2) to labeling by ophthalmologists and confirmation by senior retina specialists (JSIEC). This diverse set of diagnostic datasets ensures a comprehensive evaluation of RETFound’s efficacy in ai retina diagnosis across different disease types and severities.
Predicting the Future: Data for Disease Prognosis and Prediction with AI
Beyond diagnosis, ai retina diagnosis models are increasingly being explored for disease prognosis and prediction. RETFound’s capabilities in this domain were assessed using data from the Moorfields AlzEye study (MEH-AlzEye). This retrospective cohort study linked ophthalmic data of 353,157 patients with systemic health data, focusing on cardiovascular disease and dementia.
For predicting the prognosis of fellow eye conversion to wet-AMD within one year, MEH-AlzEye data was crucial. This dataset allowed researchers to identify patients diagnosed with wet-AMD and analyze retinal images taken within a year before fellow eye conversion, or in patients whose fellow eyes did not convert.
Furthermore, RETFound was evaluated for its ability in 3-year prediction of systemic diseases, specifically cardiovascular and neurodegenerative dysfunctions, using MEH-AlzEye and UK Biobank datasets. The UK Biobank, encompassing 502,665 UK residents, provided CFP and OCT examinations for a subset of participants, offering a large-scale dataset for exploring the link between retinal health and systemic diseases in ai retina diagnosis and predictive modeling.
Alt Text: Data processing flowchart for disease prognosis and prediction tasks using AI retina diagnosis, illustrating the use of MEH-AlzEye and UK Biobank datasets.
These prognosis and prediction tasks highlight the expanded potential of ai retina diagnosis beyond immediate disease detection, venturing into the realm of preventative and personalized healthcare.
Data Preprocessing and Augmentation for Enhanced AI Learning
Effective ai retina diagnosis models require careful data preprocessing and augmentation. For CFP images in RETFound, AutoMorph, an automated retinal image analysis tool, was used to remove backgrounds and isolate the retinal area. All images were then resized to 256 × 256 pixels. For OCT scans, middle slices were extracted and similarly resized.
Data augmentation techniques, mirroring those used in masked autoencoders, were applied during model training. These included random cropping, resizing, horizontal flipping, and image normalization. These augmentations are essential for improving the model’s robustness and generalization capability in ai retina diagnosis, enabling it to perform effectively on diverse and real-world retinal images.
RETFound Architecture and Implementation: Powering AI Retina Diagnosis
RETFound’s architecture is based on a masked autoencoder framework, comprising an encoder and a decoder. The encoder utilizes a large vision Transformer (ViT-large), while the decoder employs a smaller vision Transformer (Vit-small). This architecture is detailed in Supplementary Figure 6 of the original article.
The encoder processes unmasked image patches and extracts high-level features. The decoder then reconstructs the original image from a highly masked version, with masking ratios of 0.75 for CFPs and 0.85 for OCTs. This masked autoencoder approach facilitates self-supervised learning, allowing the model to learn meaningful representations from vast amounts of unlabeled retinal images, a key advantage in the field of ai retina diagnosis where labeled data can be scarce.
Alt Text: Architecture diagram of RETFound, showcasing the encoder-decoder structure based on Vision Transformers for AI retina diagnosis image analysis.
Model training involved a large batch size and extensive epochs, leveraging powerful NVIDIA Tesla A100 GPUs. This computational resource underlines the intensive nature of training state-of-the-art ai retina diagnosis models.
Adaptation and Refinement for Downstream AI Tasks
For specific downstream tasks in ai retina diagnosis, such as disease classification, only the encoder (ViT-large) component of RETFound is utilized. A multilayer perceptron is added to the encoder to output disease category probabilities. Label smoothing is incorporated to prevent overfitting and enhance model generalization.
The training objective is to align the model’s categorical output with the ground-truth labels. Model performance is evaluated on a validation set, and the model weights with the highest AUROC are saved for final evaluation. This adaptation process demonstrates the versatility of RETFound as a foundation model for various ai retina diagnosis applications.
Exploring Contrastive SSL and Explainability in AI Retina Diagnosis
To further enhance RETFound and compare different self-supervised learning techniques for ai retina diagnosis, contrastive SSL approaches like SimCLR, SwAV, DINO, and MoCo-v3 were explored. These methods were implemented within the RETFound framework to generate model variants for comparative analysis.
Furthermore, to improve the interpretability of ai retina diagnosis models, RELPROP, a method specifically designed for Transformer-based networks, was employed. RELPROP visualizes the image areas that contribute most significantly to a particular classification, offering insights into the model’s decision-making process and enhancing trust in ai retina diagnosis systems.
Conclusion: The Future of Eye Care is Driven by AI Retina Diagnosis
RETFound represents a significant leap forward in ai retina diagnosis. By leveraging massive and diverse datasets and employing advanced self-supervised learning techniques, RETFound establishes a powerful foundation model for a wide range of retinal image analysis tasks. From disease diagnosis and prognosis to prediction of systemic conditions, RETFound holds immense promise for transforming eye care, enabling earlier detection, more accurate diagnoses, and personalized treatment strategies. As AI continues to evolve, models like RETFound will undoubtedly play a crucial role in shaping the future of ophthalmology and improving patient outcomes worldwide.