Cardiac masses, frequently detected via echocardiography, present diagnostic challenges for cardiologists. These masses can be primary (benign or malignant) or secondary (metastatic), necessitating a comprehensive differential diagnosis to determine the optimal therapeutic strategy. While malignant cardiac tumors often carry a poor prognosis, timely and appropriate interventions can significantly improve patient outcomes. This article delves into the echocardiographic characteristics of both benign and malignant cardiac tumors, aiming to guide differential diagnosis and inform subsequent diagnostic and therapeutic decisions, with a specific focus on atrial masses.
1. Introduction
The occurrence of secondary cardiac tumors is rare, and primary cardiac tumors are even rarer. Malignant tumors within the heart are associated with a grim prognosis if left untreated. However, advancements in surgical and medical treatments have led to improved prognoses for certain primary malignant cardiac tumors in recent years.
Secondary cardiac tumors commonly manifest as pericardial metastases, often originating from carcinomas such as lung or breast cancer, or melanoma. Myocardial metastases can arise from solid or hematological malignancies. Intracavitary metastases or extensions may stem from gynecologic, urological, or pulmonary tumors. Primary cardiac tumors are classified as benign or malignant, with some histotypes capable of presenting as either benign masses or malignant differentiations (refer to Table 1). Certain tumors, including inflammatory myofibroblastic tumors, paragangliomas, and specific germ cell tumors, exhibit intermediate or unpredictable clinical behaviors. The classification is further complicated by the potential for diverse histotypes within a single mass.
TABLE 1. Simplified Classification of Common Benign and Malignant Cardiac Tumors
| Benign | Malignant |
|---|---|
| Myxoma | Myxosarcoma |
| Rhabdomyoma | Rhabdomyosarcoma |
| Angiomas | Angiosarcomas |
| Fibromas | Fibrosarcoma |
| Lipoma | Liposarcoma |
| Schwannoma | Malignant peripheral nerve sheath tumor (MPNST) |
| Cystic tumor of the AV node | Other sarcomas (myxofibro, leiomyo, undifferentiated pleomorphic, synovial, etc.) |
| Papillary fibroelastoma | Lymphomas |
| Metastatic tumors | |
| Mesothelioma | |
| Intermediate Behavior | |
| Paraganglioma | |
| Inflammatory myofibroblastic tumor | |
| Germ cell tumors | Immature teratoma, Yolk sac tumor |






Benign cardiac tumors may be managed surgically or through observation if they do not cause hemodynamic compromise or pose embolic risks. Some, like rhabdomyomas, may regress spontaneously, warranting a follow-up approach. However, even benign tumors can induce symptoms such as hemodynamic impairment, arrhythmias, or embolism, necessitating medical or surgical intervention. Rhabdomyomas, notably, have been linked to arrhythmias in a significant proportion of cases, with tumor cells resembling Purkinje cells and potentially causing pre-excitation syndromes. Hematological neoplasms, whether primary cardiac lymphomas or metastases, are primarily treated with chemotherapy, while metastatic melanomas are addressed with targeted therapy or immunotherapy. In contrast, primary cardiac sarcomas typically require surgical resection, often combined with chemotherapy or radiotherapy, to improve survival.
Differentiating between a thrombus and a metastasis is critical when a cardiac mass is found in a patient with known systemic cancer, as this distinction guides treatment strategy and has prognostic implications. Accurate differential diagnosis is therefore paramount for appropriate therapy planning, often necessitating a multimodality imaging approach.
2. General Diagnostic Approach to Atrial Masses
When evaluating an atrial mass, the initial step is to distinguish it from other cardiac masses such as thrombi or vegetations. A structured approach is essential for accurate diagnosis.
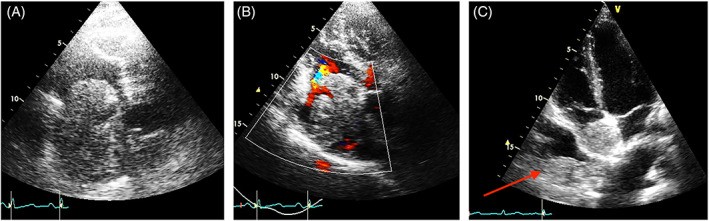
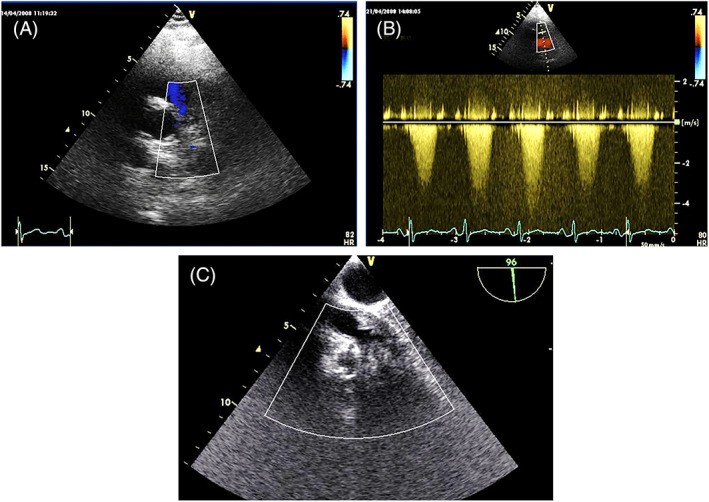
FIGURE 1. Tumor Thrombus vs. Metastatic Sarcoma
Figure 1. Echocardiographic differentiation of tumor thrombus and metastasis. (A) TTE-2D apical four-chamber view showing tumor thrombus extending from the inferior vena cava to the right atrium in liver carcinoma. (B) Color Doppler enhances the irregular shape of the tumor thrombus. (C) Right atrial mass, distinct from the inferior vena cava, in a patient with metastatic sarcoma, displaying similar echogenicity to intrapleural metastasis (red arrow), indicative of metastasis.
TABLE 2. Clinicopathologic Features of Primary Heart Tumors
| Histologic Type | Age | Site in Heart | Multiplicity | Syndromic Association |
|---|---|---|---|---|
| Fetuses/Infants | Children | Adults | Layer | |
| Benign Congenital Tumors | ||||
| Rhabdomyoma | ++ | + | Myocardium | |
| Fibroma | + | ++ | + | Myocardium |
| Histiocytoid cardiomyopathy | ++ | +/1 | Endo/Myocardium | |
| Benign Acquired Tumors | ||||
| Myxoma | +/− | ++ | Endocardium | |
| Papillary fibroelastoma | ++ | Endocardium | ||
| Hemangioma | + | + | + | Myo/Endocardium |
| Lipomatous hypertrophy | ++ | Myocardium (Septum) | ||
| Lipoma | ++ | Myo/Epi/Endo | ||
| Inflammatory myofibroblastic tumor | ++ | + | +/− | |
| Germ Cell Tumors | ||||
| Teratoma | ++ | + | +/− | |
| Yolk sac tumor | ++ | + | ||
| Malignant Tumors | ||||
| Angiosarcoma | +/− | ++ | All layers | |
| UPS/Myxofibrosarcoma | +/− | +/− | ++ | |
| Rhabdomyosarcoma | +/− | ++ | + | |
| Leiomyosarcoma | +/− | ++ | ||
| Lymphoma | +/− | ++ |
Abbreviations: AV, atrioventricular; LA, left atrium; RA, right atrium; UPS, undifferentiated pleomorphic sarcoma; SA, sinoatrial.
The diagnostic process involves evaluating:
- Clinical context: Patient history, presence of systemic malignancy, symptoms.
- Location and anatomical relationships: Chamber of origin, attachment site, involvement of adjacent structures.
- Morphological and functional characteristics: Size, shape, mobility, tissue characterization, vascularity, metabolic activity.
Echocardiography, CT, MRI, and PET/CT are employed to investigate points 3 and 4, often in combination due to their complementary strengths and weaknesses in cost, availability, and diagnostic accuracy. Transthoracic echocardiography (TTE) and transesophageal echocardiography (TOE) are typically the initial diagnostic modalities for suspected cardiac tumors due to their accessibility and cost-effectiveness.
This article focuses on the echocardiographic features relevant to the differential diagnosis of atrial masses.
3. Echocardiographic Approach to Atrial Mass Differential Diagnosis
A comprehensive echocardiographic examination, utilizing TTE and TOE, is crucial for characterizing atrial masses. This includes 2D, 3D, Color Doppler Imaging (CDI), Spectral Doppler Imaging (SDI), and contrast echocardiography using agitated saline or Ultrasound Enhancement Agents (UEAs). TOE (2D and 3D) and intracardiac ultrasound also guide biopsy procedures for histological confirmation.
The inherent contrast between the mass and the blood pool within cardiac chambers facilitates visualization of intracavitary tumors. Color Doppler can delineate the borders of tumors with low echogenicity (Figure 5).
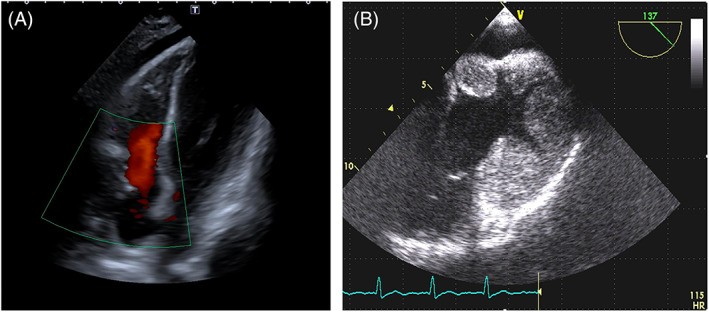
FIGURE 2. Papillary Fibroelastoma vs. Angiosarcoma
Figure 2. Echocardiographic comparison of benign and malignant atrial masses. (A) TTE-2D showing a papillary fibroelastoma on the aortic valve with a thin stalk (red arrow). (B) TTE-2D apical four-chamber view of a right atrial angiosarcoma with broad attachment to the lateral wall and infiltration of the tricuspid annulus (yellow arrow), exhibiting a lobulated surface.
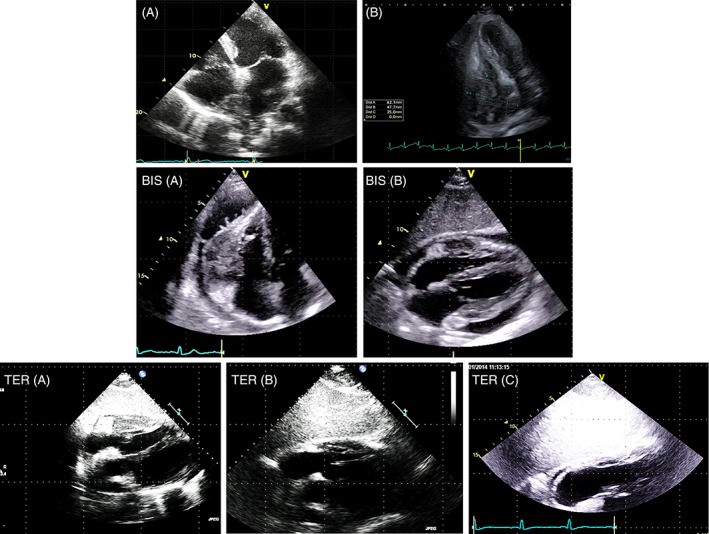
FIGURE 3. Angiosarcoma Cases Demonstrating Infiltration
Figure 3. Echocardiographic views of angiosarcoma infiltration. (A) Apical four-chamber view of a large angiosarcoma infiltrating the right atrium, right ventricle, and pericardium. (B) TOE-2D focused on right chambers, revealing the tumor infiltrating the interatrial septum and right atrial walls.
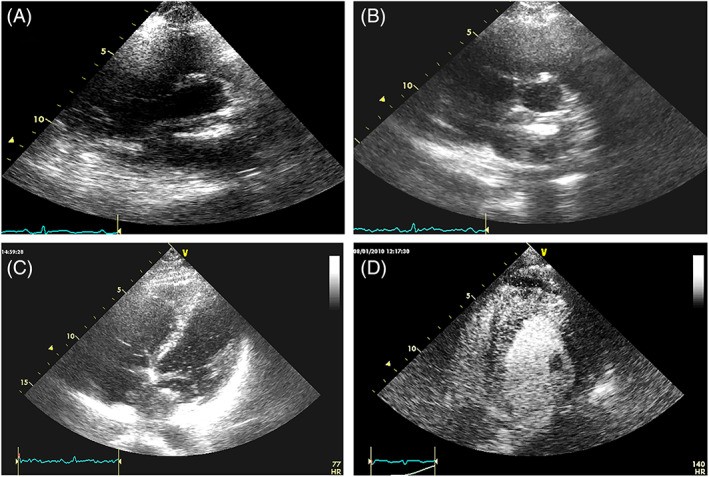
FIGURE 4. Cardiac Lymphoma Cases with Rapid Growth
Figure 4. Echocardiographic presentation of cardiac lymphoma. (A) TTE-2D apical four-chamber view showing lymphoma infiltrating the interatrial septum, right atrium, and superior vena cava. (B) Extensive lymphoma occupying the right atrium and infiltrating adjacent structures. (C-E) Monitoring chemotherapy efficacy in pericardial lymphoma using TTE-2D subcostal views over two months, showing tumor regression.
Systematic analysis of cardiac tumors, including atrial masses, involves defining morphology, dynamics, hemodynamic effects, and potential therapeutic strategies, regardless of their benign or malignant nature, primary or metastatic origin.
4. Size of Atrial Masses
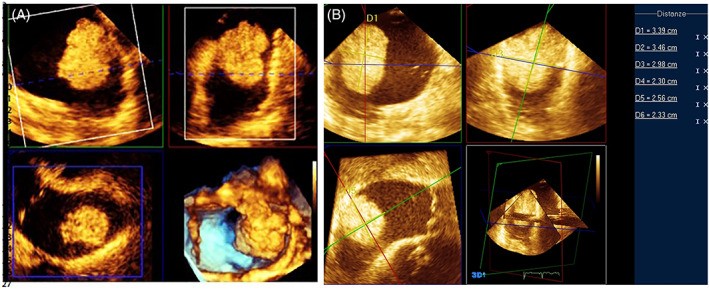
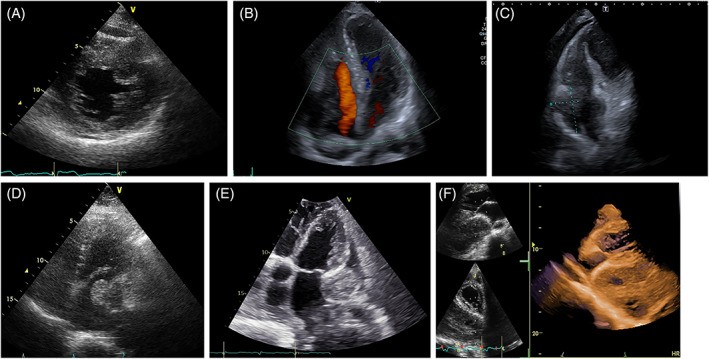
Atrial masses vary significantly in size, from millimeters to centimeters. TTE often accurately determines tumor size. Myxomas, the most common primary atrial tumors, can range from 2 to 9.5 cm in diameter, as documented in echocardiographic studies. However, soft, mobile tumors like myxomas can deform and elongate during prolapse, potentially altering their apparent size. Irregularly shaped tumors may also lead to size misestimation with 2D echocardiography. 3D echocardiography offers improved size assessment by imaging the entire tumor volume and enabling orthogonal measurements (Figure 7).
FIGURE 5. Left Atrial Sarcoma Post-Chemotherapy
Figure 5. Echocardiographic assessment of left atrial sarcoma response to chemotherapy. TTE-2D views (four-chamber and short-axis) before and after chemotherapy, showing significant reduction in tumor size and restoration of left atrial cavity.
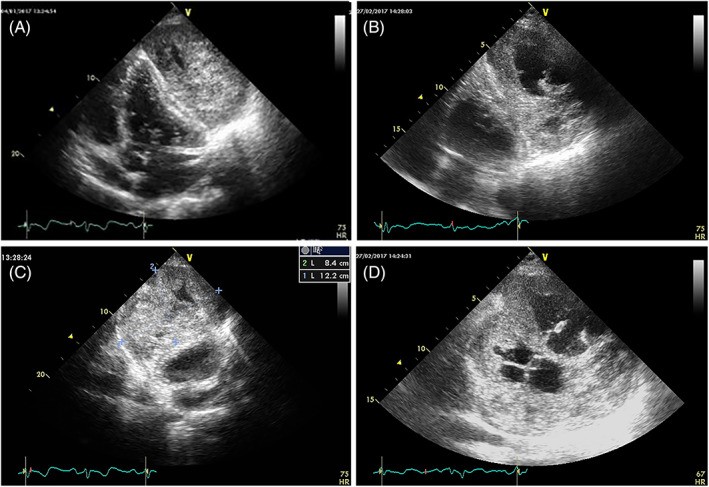
FIGURE 6. Variable Sizes of Left Atrial Myxomas
Figure 6. Echocardiographic demonstration of variable myxoma sizes. TTE-2D apical four-chamber views illustrating left atrial myxomas of varying dimensions across six patients.
FIGURE 7. 3D Echocardiography for Myxoma Measurement
Figure 7. 3D echocardiographic precision in myxoma assessment. TOE-3D multiplanar reconstruction (A) and orthogonal linear measurements (B) of an atrial myxoma, enhancing size and volume determination.
5. Site and Attachment of Atrial Masses
The location and mode of attachment are crucial in Atrial Mass Differential Diagnosis. Left atrial myxomas typically attach to the fossa ovalis region of the atrial septum (Figure 8), while right atrial angiosarcomas can arise from any part of the right atrial wall (Figures 2B, 3, 9). Atypical locations for myxomas include the inferior atrial septum or the lateral left atrial wall (Figure 10).
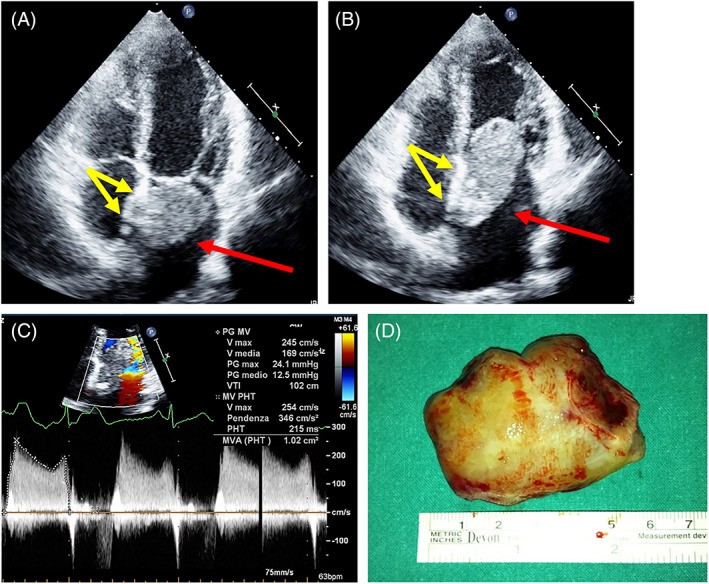
FIGURE 8. Typical Myxoma Attachment to Fossa Ovalis
Figure 8. Echocardiographic visualization of typical myxoma attachment. TTE-2D apical four-chamber views (systole and diastole) showing a left atrial myxoma attached to the fossa ovalis (red arrow: mass, yellow arrows: attachment site). Doppler study and surgical specimen confirm obstruction and myxoma.
FIGURE 9. Right Atrial Angiosarcoma with Extensive Infiltration
Figure 9. TOE-2D imaging of right atrial angiosarcoma. Views focused on right chambers showing a large, irregular mass infiltrating the atrial wall and tricuspid annulus.
FIGURE 10. Atypical Myxoma Location Below Fossa Ovalis
Figure 10. Echocardiographic detection of atypically located myxoma. Tee-2D apical four-chamber views (systole and diastole) of a left atrial myxoma attached to the atrial septum below the fossa ovalis (red arrow: mass, yellow arrows: stalk).
Tumors can be pedunculated (stalk attachment) or sessile (broad-based). Papillary fibroelastomas typically have a narrow stalk (Figure 11), while myxoma stalks vary in width. Sessile tumors, especially malignant ones, often infiltrate surrounding structures (Figures 2B, 3, 4, 5, 9). Detecting the stalk can be challenging with TTE, often necessitating TOE. TOE-3D precisely locates the stalk and its relationship to adjacent structures (Figure 7).
FIGURE 11. Papillary Fibroelastoma with Narrow Stalk
Figure 11. Echocardiographic and surgical correlation of papillary fibroelastoma. Top: TEE long-axis view showing a papillary fibroelastoma on the aortic valve with a narrow stalk (yellow arrow). Bottom: surgical specimen confirming the pedunculated nature.
6. Shape and Surface Characteristics of Atrial Masses
Tumor shape on echocardiography is influenced by size, consistency, mobility, and chamber size. Benign primary tumors are usually regular and rounded. Atrial myxomas exhibit the most shape variability. Encapsulated myxomas are round/ovoid with smooth surfaces. Papillary and gelatinous myxomas have irregular, multilobated surfaces, correlating with higher embolization risk due to surface fragmentation. Ovoid myxomas elongate upon ventricular prolapse. Papillary fibroelastomas are club-shaped with smooth surfaces (Figure 11). Intramural tumors are typically ovoid. Malignant primary tumors are broad-based with multilobated shapes. Metastatic tumor shapes depend on invasion mechanism and site.
7. Mobility of Atrial Masses
Mobility depends on location, attachment, stalk characteristics, size, and tissue properties. Intramyocardial and intrapericardial tumors move with the heart but lack intrinsic mobility, potentially restricting cardiac motion (Figure 12). Intracavitary tumors are mobile if pedunculated or prolapsing. Papillary fibroelastomas are highly mobile, exhibiting fluttering. Myxoma mobility varies from immobile to hypermobile, depending on type. Broad-based, solid myxomas may be immobile, while gelatinous, polypoid myxomas can be hypermobile during prolapse. TOE, especially 2D and 3D, is essential for detailed assessment of mass attachment, type, surrounding relationships, and mobility, crucial for embolism risk assessment and therapeutic decisions.
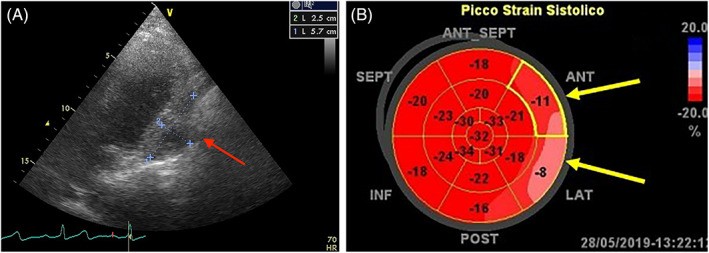
FIGURE 12. Sarcoma Infiltration and Reduced Cardiac Kinetics
Figure 12. Echocardiographic assessment of sarcoma impact on cardiac function. (A) Sarcoma infiltrating the pericardium and left ventricular lateral wall (red arrow). (B) Global longitudinal strain imaging showing reduced strain values at infiltrated wall levels (yellow arrow), indicating impaired kinetics.
8. Tissue Characterization of Atrial Masses by Echogenicity
Echogenicity on 2D-3D echocardiography provides preliminary tissue characterization. Homogeneous echogenicity is seen in rhabdomyomas, rhabdomyosarcomas, and leiomyosarcomas, with heterogeneity suggesting necrosis or calcification. Lipomas are typically homogeneous but vary in echogenicity. Fibromas are usually hyperechoic. Hemangiomas, angiosarcomas, and lymphomas are typically inhomogeneous with echolucent areas (Figure 13).
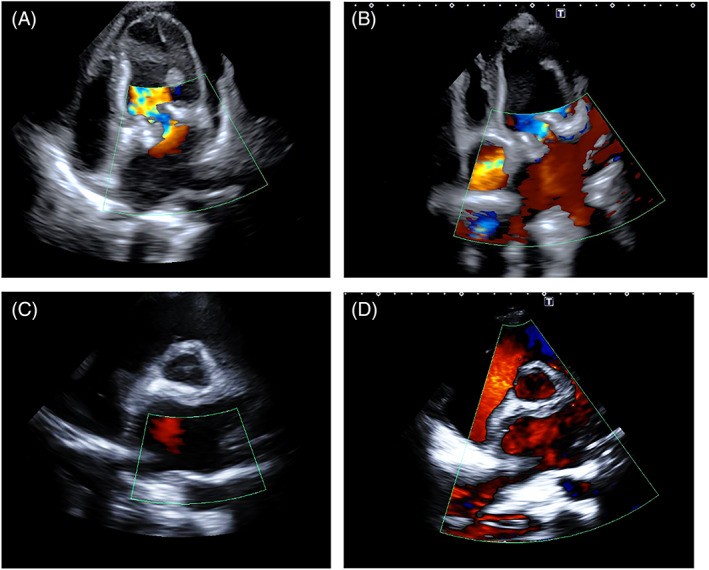
FIGURE 13. Variable Echogenicity in Different Tumor Types
Figure 13. Echocardiographic spectrum of tumor echogenicity. (A) Neuroendocrine tumor in septum: granular echogenicity. (B) Lymphoma in right chambers: mixed hypo- and hyperechogenic areas. (C) Angiosarcoma in right chambers: irregular echogenicity. (D) Myxosarcoma with hyperechogenic chondroid areas. (E) Extraskeletal osteosarcoma with calcifications (hyperechogenic). (F) Pericardial sarcoma with necrotic areas (hypoechogenic “holes” in 3D reconstruction).
Echogenicity can change post-therapy. Chemotherapy may induce necrosis (anechoic areas), while radiotherapy may cause fibrosis, increasing echogenicity (Figures 14, 15). Color Doppler assesses vascularity, especially in angiomas and angiosarcomas (Figure 16).
FIGURE 14. Echogenicity Changes Post-Chemotherapy
Figure 14. Echocardiographic changes in leiomyosarcoma after chemotherapy. Pre-chemotherapy (left) and post-chemotherapy (right) views showing development of anechoic areas indicative of necrosis.
FIGURE 15. Echogenicity Changes Post-Radiotherapy
Figure 15. Echocardiographic evolution of leiomyosarcoma during and after radiotherapy. Showing changes from homogenous low echogenicity (pre-RT), to mixed echoic/anechoic (during RT), to uniform high echogenicity (post-RT).
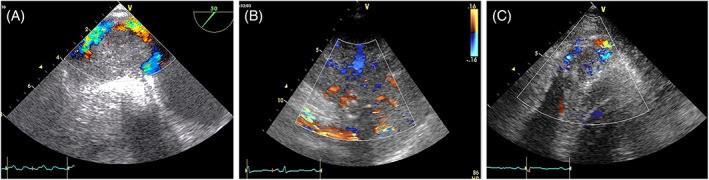
FIGURE 16. Hypervascularity in Angiosarcoma on Color Doppler
Figure 16. Color Doppler imaging of angiosarcoma vascularity. Demonstrating hypervascularization in three angiosarcoma cases: (A) Right atrium and IVC origin. (B) Right atrium to ventricle extension. (C) Right ventricular apex infiltration.
Ultrasound-enhancing agents (UEAs) improve tumor detection and characterization. UEA perfusion imaging differentiates vascular tumors from avascular thrombi. Malignant tumors typically show hyperenhancement due to neovascularization (Figure 17). Benign vascular tumors like hemangiomas also enhance. Myxomas show partial enhancement, while thrombi and fibroelastomas show no enhancement.
FIGURE 17. UEA Enhancement in Left Atrial Sarcoma
Figure 17. Contrast-enhanced echocardiography of left atrial sarcoma. (A) Standard TTE showing poorly defined, low echogenicity mass. (B, C) Enhanced echogenicity of the mass 5 minutes post-SonoVue injection. (D) Myocardial lymphoma infiltration showing granular enhancement post-SonoVue.
9. Hemodynamic Impact of Atrial Masses
Atrial tumors can cause obstruction if the mitral or tricuspid valve area becomes less than 2 cm², often due to prolapsing masses (Figure 8). Sarcomas and lymphomas can also cause obstruction by infiltrating the valve annulus. Pulmonary artery sarcomas frequently induce stenosis (Figure 18). Vena cavae or pulmonary veins can be obstructed by atrial sarcomas (Figures 5, 16). Valvular regurgitation is a rare complication of fibroelastomas, caused by cusp traction. Extracardiac tumors can compress vessels or chambers.
FIGURE 18. Pulmonary Artery Sarcoma with Stenosis
Figure 18. Echocardiographic assessment of pulmonary artery sarcoma. (A) Poorly echogenic mass causing pulmonary artery stenosis. (B) Stenosis severity. (C) Improved mass definition via TOE.
10. Integrating Echocardiography into Clinical Atrial Mass Differential Diagnosis
Echocardiography provides essential information for atrial mass diagnosis, including location, attachment, size, shape, tissue characteristics, mobility, and hemodynamic impact. Its temporal resolution is ideal for detecting mobile masses, especially valvular ones. TTE is the initial tool, but advanced techniques like TOE and UEAs are often needed for detailed atrial mass evaluation. Misdiagnosis of malignant atrial tumors as myxomas has been reported, highlighting the need for thorough assessment.
TABLE 3. Comparison of Echocardiographic Modalities for Cardiac Tumor Diagnosis
| Feature | TTE-2D | TTE-3D | TOE-2D | TOE-3D | UEAs |
|---|---|---|---|---|---|
| Detection | +++ | + | ++++ | ++ | − |
| Location | +++ | ++ | ++++ | ++++ | + |
| Attachment | ++ | ++ | +++ | ++++ | − |
| Size | ++ | +++ | +++ | ++++ | − |
| Shape/Surface | ++ | ++ | +++ | ++++ | − |
| Mobility | +++ | +++ | ++++ | ++++ | |
| Tissue Characterization | ++ | ++ | +++ | ++ | ++++ |
| Hemodynamic Impact | +++ | ++ | +++ | ++ | − |
Note: Utility: – no, + fair, ++ good, +++ very good, ++++ very very good.
TOE-2D is crucial for precise atrial mass assessment and relationships with pulmonary veins and vena cava. TOE-3D adds value by improving accuracy in location, attachment, size, shape, tissue characterization, and adjacent structure relationships. Table 3 summarizes echocardiographic modality utility.
Echocardiography limitations include origin assessment, extent, and tissue characterization. Morphologically similar benign and malignant tumors necessitate pathology for definitive diagnosis. PET/CT is superior in differentiating benign from malignant masses (myxoma vs. myxosarcoma, etc.) based on metabolic activity using 18FDG and heterogeneity factor (HF). Optimizing 18FDG sensitivity requires dietary and heparin protocols. Specific tracers are needed for neuroendocrine tumors and paragangliomas. CT or MRI with angiography are useful for ventricular tumors near coronary arteries.
TABLE 4. Preliminary Differential Diagnosis Based on Clinical and Echocardiographic Features
| Intracavitary |
|---|
| (a) Left atrium: Myxoma, sarcoma, lung metastasis |
| – Septal, thin stalk: Myxoma likely |
| – Other sites: Sarcoma considered |
| – Broad base: Sarcoma likely (vs. thrombus) |
| – Pulmonary vein continuity: Lung cancer, sarcoma |
| (b) Right atrium/ventricle: Angiosarcoma, liver/ovarian metastasis, lymphoma |
| – Atrial wall infiltration: Angiosarcoma, lymphoma |
| – IVC continuity: Liver/gynecological cancer (vs. thrombus, tumor thrombus) |
| – SVC continuity: Lymphoma |
| – Thin stalk, single: Myxoma |
| (c) Left ventricle: Leiomyosarcoma, myxoma |
| (d) Valve: Papillary fibroelastoma (vs. endocarditis) |
| (e) Pulmonary artery: Intimal sarcoma |
| Intramural |
| 1. Infants/children: Rhabdomyoma, fibroma, angioma |
| – Adults with systemic malignancy: Metastasis |
| – Adults without other diseases: Sarcoma, fibroma, angioma |
| Intrapericardial |
| 1. Adults with systemic malignancy: Metastasis |
| – Right chamber infiltration: Angiosarcoma |
| – Diffuse encasement: Angiosarcoma, lung cancer, lymphoma |
Clinical context and echocardiographic features guide preliminary differential diagnosis (Tables 2, 4), informing further diagnostic steps.
11. Conclusion
Echocardiography is pivotal in cardiac tumor evaluation, guiding diagnosis and management. Integrated with clinical context, it helps determine need for surgery, medical treatment, or observation. However, multimodality imaging, including CT, MRI, and PET/CT, is often necessary for definitive diagnosis and therapy planning. A multidisciplinary team approach, including cardiologists, oncologists, radiologists, and nuclear medicine specialists, is crucial for optimal management of patients with atrial masses and suspected cardiac tumors.
References
[List of references from the original article – to be included here in the final version]
Supporting Information
Video S1 Videoclips (Link to original supplementary material)
Data Availability Statement
Review article, no experimental data.