Transthyretin amyloid cardiomyopathy (ATTR-CM) is a serious, often underdiagnosed, and potentially fatal form of heart failure that, fortunately, is now becoming increasingly treatable. The diagnostic challenges associated with ATTR-CM frequently lead to delayed or incorrect diagnoses. With the advent of therapies like tafamidis significantly improving patient outcomes, enhancing disease awareness and streamlining diagnostic pathways are paramount. Modern, cost-effective imaging techniques, including longitudinal strain imaging, cardiac magnetic resonance (CMR) T1 mapping, and cardiac scintigraphy, now enable the non-invasive diagnosis of ATTR-CM, removing the necessity for invasive tissue biopsies in many cases. This article provides an updated review of the essential diagnostic tools available to clinicians for the effective diagnosis of ATTR cardiac amyloidosis.
Keywords: Attr Cardiac Amyloidosis Diagnosis, Cardiac Amyloidosis, Cardiac Scintigraphy, Cardiomyopathy, Diastolic Dysfunction, Transthyretin Amyloid
Key Insights into ATTR Cardiac Amyloidosis Diagnosis
| Key Diagnostic Challenges & Advancements for ATTR-CM |
|---|
| ATTR-CM frequently presents as an elusive and life-threatening condition, often mimicking other cardiac diseases. |
| Recognizing a range of clinical indicators, both cardiac and extracardiac, is crucial for effective screening and early suspicion of ATTR-CM. |
| Definitive diagnosis of ATTR-CM is now achievable through non-invasive imaging, significantly reducing the need for biopsies. |
| Cardiac scintigraphy stands out as a vital imaging modality, playing a pivotal role in the accurate diagnosis of ATTR-CM. |
| While cardiac scintigraphy is highly indicative of ATTR-CM, it does not distinguish it from AL amyloid cardiomyopathy, necessitating a comprehensive, multidisciplinary diagnostic approach to rule out AL amyloidosis and confirm ATTR-CM. |
Introduction to ATTR Cardiac Amyloidosis Diagnosis
Despite significant progress in both diagnostic tools and treatment options, transthyretin (TTR) amyloid cardiomyopathy (ATTR-CM) remains a diagnostically challenging condition. The consequences of untreated amyloidosis are severe, often progressing to end-stage heart failure with a grim prognosis. ATTR-CM arises from the deposition of misfolded transthyretin protein within the heart muscle. This aberrant protein accumulates as amyloid fibrils, characterized by a cross-beta-sheet structure, which are toxic to various tissues. This mechanism contrasts with immunoglobulin light chain (AL) amyloidosis, where misfolded light chains secreted by plasma cells are the culprit [1].
The diagnosis of ATTR cardiac amyloidosis is often complicated because its clinical presentation can overlap significantly with numerous other cardiac conditions. Furthermore, many clinicians may lack familiarity with the specific diagnostic steps required for amyloid cardiomyopathies, potentially leading to confusion between ATTR-CM and AL amyloidosis. Consequently, a significant number of patients with ATTR-CM are initially misdiagnosed and subsequently treated for the incorrect condition [2]. Recognizing key clinical indicators is therefore crucial for prompting clinicians to consider ATTR-CM in their differential diagnosis. Timely and appropriate utilization of diagnostic tools is indispensable for accurate diagnosis. This review aims to comprehensively discuss the primary diagnostic tools that are essential for the diagnosis of ATTR cardiac amyloidosis. This article does not report on studies involving human or animal subjects conducted by the authors.
Prevalence of ATTR Cardiac Amyloidosis
The precise prevalence of ATTR-CM remains uncertain, largely due to underdiagnosis. Wild-type ATTR-CM (ATTR-CMwt) is the more prevalent form, with autopsy studies indicating an increasing incidence with advancing age [3]. Among older patients hospitalized for heart failure with preserved ejection fraction (HFpEF), bone scintigraphy revealed ATTR-CM in 13% of cases. All of these patients received a diagnosis of ATTR-CMwt by the age of 86 [4]. Hereditary ATTR-CM (ATTR-CMh) shares a similar clinical presentation to ATTR-CMwt, typically manifesting as late-onset restrictive cardiomyopathy. The average age at presentation for ATTR-CMh is around 69 years [5].
Subtypes and Pathogenesis of ATTR Cardiac Amyloidosis
ATTR-CM is genetically classified based on the TTR gene into two main subtypes: wild-type (ATTR-CMwt) and hereditary (ATTR-CMh). The TTR gene, located on chromosome 18, encodes a 127-amino acid protein. ATTR-CMwt occurs without any identifiable genetic mutation, while ATTR-CMh is characterized by a specific single amino acid mutation. Recent data suggest a higher prevalence of ATTR-CMh in certain populations, with approximately 3.5% of African Americans carrying the gene variant. This allele is found in about 10% of African Americans over 65 years of age with congestive heart failure [6]. Table 1 summarizes the key distinctions between ATTR-CMwt and ATTR-CMh.
Table 1. Differentiating Wild-Type and Hereditary ATTR Cardiac Amyloidosis
| Feature | ATTR-CMwt | ATTR-CMh |
|---|---|---|
| Age of Onset | Typically > 60 years | Variable, depending on mutation (30–80 years) |
| Genetic Basis | Normal TTR gene sequence | Nucleotide mutations in the TTR gene |
| Survival | Approximately 3.5 years post-diagnosis | Variable, dependent on specific genetic mutation |
| Patient Demographics | Predominantly Male; Increased prevalence with age; Higher incidence in African Americans | Mutations are geographically clustered (e.g., Ireland, Japan, Sub-Saharan Africa) |
TTR protein is synthesized in the liver, choroid plexus, and retinal pigment epithelium. In its native state, TTR is a tetramer composed of four beta-sheet-rich monomers circulating in the bloodstream. It functions as a transport protein for thyroxine and retinol-binding protein [7]. Genetic studies have identified that single amino acid mutations within the 127-amino acid TTR sequence can lead to protein misfolding and aggregation [8]. This misfolded TTR accumulates in tissues, resulting in clinical ATTR amyloidosis [9]. In ATTR-CM, the accumulation of aggregated and misfolded TTR creates a rigid, space-occupying infiltrate within the myocardium, causing restrictive physiology and cardiac dysfunction [10].
Clinical Manifestations of ATTR Cardiac Amyloidosis
Cardiac Involvement and Differential Diagnoses in ATTR-CM
A typical clinical presentation of ATTR-CM is an elderly patient diagnosed with heart failure with preserved ejection fraction (HFpEF). These individuals often present with a history of worsening heart failure that proves resistant to standard therapies. Echocardiographic findings commonly reveal increased left ventricular wall thickness. Despite treatment, patients experience progressive decline, and their hemodynamic profiles often exhibit a restrictive pattern. Notably, these patients may not tolerate beta-blockers well and can experience spontaneous improvement in hypertension, even without medication or lifestyle modifications [10]. The average age at diagnosis for ATTR-CMwt is around 74 years, with a higher prevalence in males [11], leading to the term “senile amyloidosis” for this population.
ATTR-CM can be diagnostically challenging as its presentation can mimic other cardiac conditions, most notably aortic stenosis, hypertrophic cardiomyopathy, and AL amyloidosis. Other conditions in the differential diagnosis include dilated cardiomyopathies, toxic cardiomyopathies, peripartum cardiomyopathy, cardiac sarcoidosis, and stress-induced cardiomyopathy. Consequently, ATTR-CM is often underdiagnosed in patients initially presenting with these conditions.
Retrospective studies have indicated a relatively high prevalence of ATTR-CM in patients with severe aortic stenosis undergoing surgical valve replacement [12]. In patients with low-flow, low-gradient severe aortic stenosis, the presence of coexisting ATTR-CM is suspected as a cause for restrictive physiology and limited improvement post-valve replacement. These patients have demonstrated poorer outcomes despite valve replacement and hemodynamic improvements [13]. As long-term data from transcatheter aortic valve replacement (TAVR) patients accumulates, further insights into the relationship between ATTR-CM and aortic stenosis will emerge.
Studies have also shown that asymmetric septal hypertrophy, a hallmark of hypertrophic cardiomyopathy, can be observed in up to 25% of ATTR-CMwt patients [2]. These patients are frequently misdiagnosed with hypertrophic cardiomyopathy. Histological examination has revealed incidental ATTR-CM in patients undergoing septal myectomy for left ventricular outflow tract obstruction (LVOTO) [14]. Clinicians managing hypertrophic cardiomyopathy patients should therefore consider ATTR-CM in the differential diagnosis. Furthermore, histopathological analysis should be considered in septal myectomy specimens to identify potential coexisting ATTR-CM.
Extracardiac Involvement in ATTR Cardiac Amyloidosis Diagnosis
Extracardiac manifestations of ATTR can serve as valuable screening tools, enabling clinicians to suspect ATTR-CM even before significant cardiac symptoms develop. These extracardiac signs include involvement of the central nervous system, nephrotic syndrome, ocular manifestations, gastrointestinal issues, autonomic neuropathy, and peripheral neuropathy. However, the most commonly observed and consistent extracardiac findings in ATTR are bilateral carpal tunnel syndrome, spinal stenosis, and spontaneous biceps tendon rupture [15, 16]. While ATTR-CMwt and ATTR-CMh generally share similar extracardiac manifestations, genetic studies indicate that the specific extracardiac features of ATTR-CMh can vary slightly depending on the underlying genotype. For instance, the VAL122lle (pV1421) mutation typically does not cause polyneuropathy [17], while the Thr60AIa (pT80A) mutation, prevalent in Ireland, is associated with a high incidence of carpal tunnel syndrome [18].
ATTR amyloid fibril deposition in soft tissues can lead to nerve entrapment, with median nerve entrapment, or carpal tunnel syndrome, being the most common. Interestingly, research has shown that carpal tunnel syndrome symptoms can precede clinical ATTR-CM by up to 10 years [19]. Approximately 50% of patients with ATTR-CMwt experience carpal tunnel syndrome [20]. These findings underscore the importance of considering carpal tunnel syndrome as a significant clinical indicator for screening patients with heart failure symptoms and a history of median nerve entrapment. This approach can facilitate earlier and more efficient diagnosis. Furthermore, if patients have undergone carpal tunnel release surgery, pathological examination of tenosynovial tissue can be informative, as amyloid deposits, if present, can aid in diagnosis [21].
Similar to carpal tunnel syndrome, ATTR deposition in musculoskeletal tissues can cause spinal canal narrowing and compression via ligamentum flavum infiltration. Studies have shown that over 45% of older patients undergoing surgery for spinal stenosis had ATTR deposition [22]. Additionally, ATTR deposits were found in 33% of patients with spontaneous distal biceps tendon rupture [15]. Orthopedic studies have also identified amyloid deposits in tissue samples from patients undergoing rotator cuff repairs [23] and total knee and hip arthroplasties [24]. These musculoskeletal manifestations of ATTR serve as crucial clues in screening patients at risk for ATTR-CM.
Diagnostic Tools for ATTR Cardiac Amyloidosis Diagnosis
Serology in ATTR-CM Diagnosis
Unlike AL amyloidosis, where circulating biomarkers like light chains are present, ATTR-CM lacks specific serological biomarkers for routine testing. However, emerging serologic tests for endogenous TTR ligand retinol-binding protein 4 show promise as potential biomarkers in the future [25]. Classic cardiac biomarkers, such as troponin and B-type natriuretic peptide (BNP), are consistently elevated in ATTR-CM and are incorporated into staging and prognostic assessments [26]. Serum troponin levels can be persistently elevated even in the absence of overt cardiomyopathy. Similarly, pro-B-type natriuretic peptide (proBNP) levels may be disproportionately elevated relative to the clinical severity of heart failure [27].
Electrocardiogram (ECG) in ATTR-CM Diagnosis
An electrocardiogram (ECG) can provide initial clues suggestive of ATTR-CM. Patients with increased left ventricular wall thickness who exhibit a low-voltage ECG pattern should be further evaluated for ATTR-CM. Typically, hypertrophic cardiomyopathy and hypertensive heart disease, which also cause left ventricular wall thickening, are associated with ECG criteria for left ventricular hypertrophy, not low voltage. However, it is important to note that low voltage is only present in up to 40% of ATTR-CM patients [28].
Atrioventricular (AV) block is another significant ECG finding in ATTR-CM. Amyloid infiltration of the sinus and AV nodes can lead to conduction system abnormalities, often requiring pacemaker implantation. AV block is observed in up to 22% of patients with cardiac amyloidosis [29].
Imaging Modalities for ATTR Cardiac Amyloidosis Diagnosis
Imaging is central to the non-invasive diagnosis of ATTR-CM. Transthoracic echocardiography (TTE), cardiovascular magnetic resonance (CMR), and cardiac scintigraphy are the three primary imaging modalities proven to be valuable and, in some cases, diagnostic for ATTR-CM.
Transthoracic Echocardiography (TTE) in ATTR-CM Diagnosis
While TTE has limitations, several echocardiographic findings should raise suspicion for cardiac amyloidosis. Symmetrical thickening of the left ventricular (LV) walls is a notable feature, often misidentified as LV hypertrophy. In ATTR-CM, this thickening is due to extracellular amyloid protein deposition, not myocyte hypertrophy as seen in true LV hypertrophy. The myocardium may exhibit a characteristic “sparkling” or “speckled” appearance due to the dense amyloid infiltration. Pleural and pericardial effusions, although often small, may also be present. Diastolic function parameters typically show a restrictive filling pattern and significant bi-atrial enlargement, although these findings often manifest in later stages of the disease [30]. However, these findings are not specific to ATTR-CM. For example, increased LV wall thickness and a speckled pattern can also be seen in end-stage renal disease and glycogen storage diseases.
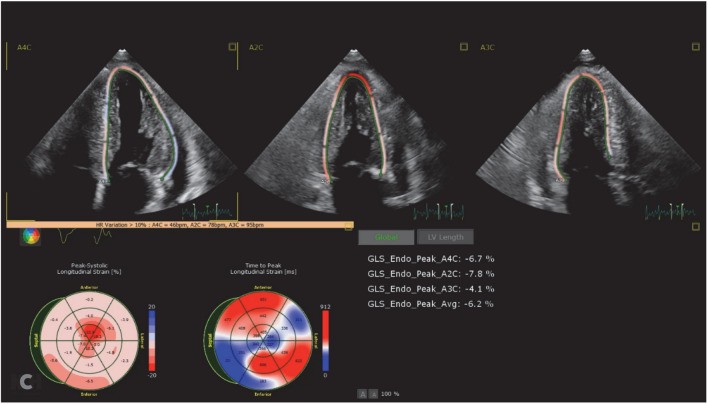
Advanced echocardiographic techniques, particularly longitudinal systolic strain imaging, have improved diagnostic accuracy. Longitudinal systolic strain is reduced in ATTR-CM, with a characteristic pattern of apical sparing—preserved strain in the apical region of the LV compared to reduced strain in the mid and basal segments. This creates a distinctive “cherry on top” pattern on longitudinal strain imaging maps (Fig. 1). An abnormal apical to mid/basal strain ratio has demonstrated high diagnostic accuracy for cardiac amyloidosis and aids in differentiating it from other conditions [31]. This is particularly useful when differentiating ATTR-CM from hypertensive heart disease or aortic stenosis, which can present with similar basic echocardiographic features. However, longitudinal strain imaging alone cannot differentiate between ATTR-CM and AL amyloidosis, as AL amyloidosis can produce a similar strain pattern.
Fig. 1.
Transthoracic echocardiography demonstrating a classic cardiac amyloid longitudinal strain pattern of the “cherry on top,” highlighting apical sparing in ATTR cardiac amyloidosis diagnosis.
Cardiovascular Magnetic Resonance (CMR) Imaging in ATTR-CM Diagnosis
CMR is increasingly utilized for evaluating a wide range of cardiac pathologies. Gadolinium enhancement in CMR is particularly helpful in detecting infiltrative cardiomyopathies. Inability to suppress the myocardial signal (failure of gadolinium to wash out) or diffuse subendocardial or transmural late gadolinium enhancement (LGE) is highly suggestive of amyloidosis, demonstrating impressive sensitivity and specificity [32]. This pattern of LGE is not typically observed in other cardiac conditions like hypertensive heart disease, where gadolinium washes out from the myocardium (Fig. 2).
Fig. 2.
a Four-chamber phase-sensitive inversion recovery CMR sequences demonstrating late gadolinium enhancement across all chambers, indicating transmural involvement and inability to null the myocardium in ATTR cardiac amyloidosis. b Short-axis phase-sensitive inversion recovery CMR sequence showing diffuse late gadolinium enhancement and transmural involvement, characteristic of amyloid deposition in ATTR cardiac amyloidosis. Figure images courtesy of Dr Mohammad Al-Ani, Division of Cardiovascular Medicine, University of Florida, Gainesville, Florida.
Advanced CMR techniques, such as T1 mapping, further enhance sensitivity and provide quantitative measures of amyloid infiltration in the myocardium. Quantitative metrics like extracellular volume fraction (ECV) are significantly elevated in cardiac amyloidosis compared to other myocardial diseases [33].
While CMR is highly effective in diagnosing infiltrative processes like cardiac amyloidosis, it cannot, on its own, differentiate between ATTR-CM and AL amyloid cardiomyopathy [34]. Therefore, additional diagnostic modalities are necessary to determine the specific type of amyloidosis.
Cardiac Scintigraphy with 99m Technetium Pyrophosphate (Tc-99m PYP) in ATTR-CM Diagnosis
Currently, cardiac scintigraphy using bone-avid radiotracers is the only imaging modality capable of accurately identifying the specific type of cardiac amyloidosis, particularly ATTR-CM.
Three technetium-labeled radiotracers are commonly used to detect ATTR-CM: technetium-99m pyrophosphate (Tc-99m PYP), technetium-99m 3,3-diphosphono-1,2-propanodicarboxylic acid (Tc-99m DPD), and technetium-99m hydroxymethylene diphosphonate (Tc-99m HMDP) [10]. Myocardial uptake of these radiotracers is visually compared to rib uptake, and cardiac uptake greater than rib uptake is indicative of ATTR-CM. Although the precise mechanism of radiotracer uptake in the myocardium is not fully understood, several hypotheses have been proposed [35]. Studies have demonstrated that these radiotracers provide 100% specificity for ATTR-CM diagnosis when grade 2 or 3 myocardial uptake is observed in heart failure patients without monoclonal protein (ruling out AL amyloidosis) and with supportive echocardiographic or CMR findings of amyloidosis [36]. Thus, cardiac scintigraphy offers a non-invasive pathway to diagnose ATTR-CM.
Despite the significant advantage of cardiac scintigraphy, the diagnostic approach to ATTR-CM heavily relies on effectively excluding AL amyloidosis. A multidisciplinary approach is essential, requiring thorough hematologic and nephrologic evaluations to confidently rule out AL amyloidosis. AL amyloid cardiomyopathy itself can also exhibit increased radiotracer uptake on cardiac scintigraphy. Common serological tests for AL amyloidosis, such as serum and urine protein electrophoresis, can be unreliable and challenging to interpret for non-specialists. Furthermore, monoclonal gammopathy can coexist with ATTR-CM in some patients [37]. Therefore, a multidisciplinary evaluation, along with genetic testing and tissue biopsy, should be strongly considered in complex diagnostic cases. Table 2 summarizes non-invasive diagnostic indicators that raise suspicion for ATTR-CM.
Table 2. Non-Invasive Diagnostic Indicators and Tests for Suspecting ATTR Cardiac Amyloidosis
| Diagnostic Indicator/Test | Key Features Suggestive of ATTR-CM |
|---|---|
| Electrocardiogram (ECG) | Discrepancy between left ventricular thickness and QRS voltage; Presence of atrioventricular block |
| Echocardiogram (TTE) | Increased left ventricular wall thickness; “Speckled” myocardial texture; Reduced longitudinal strain with apical sparing on strain imaging; Restrictive diastolic filling pattern |
| Cardiovascular Magnetic Resonance (CMR) | Marked extracellular volume expansion; Abnormal myocardial nulling on T1 mapping; Delayed gadolinium enhancement (LGE) |
| Cardiac Scintigraphy (Tc-99m PYP) | Increased radiotracer uptake in the myocardium (Grade 2 or 3) |
| Serology | Mild, chronic elevation of cardiac biomarkers (troponin, BNP); Absence of monoclonal protein indicative of AL amyloidosis |
| Clinical Features | Heart failure with preserved ejection fraction (HFpEF); Intolerance to beta-blockers or ACE inhibitors; Resolution of hypertension without medication; Peripheral neuropathy; Carpal tunnel syndrome (bilateral); Biceps tendon rupture; Lumbar spinal stenosis |
Genetic Testing in ATTR Cardiac Amyloidosis Diagnosis
Genetic testing is recommended for all patients diagnosed with ATTR-CM, regardless of age, primarily due to the potential implications for family members. Currently, the optimal surveillance strategies for individuals with a variant genotype are still under investigation [10]. Genetic counseling is a crucial component of managing ATTR-CM, particularly for hereditary forms.
Endomyocardial Biopsy in ATTR Cardiac Amyloidosis Diagnosis
Tissue biopsy with histopathology and immunohistochemistry has been the traditional gold standard for definitively diagnosing amyloidosis. Extracardiac tissue biopsies, such as abdominal fat pad aspirates, have variable and often unreliable sensitivity [38]. Tissue samples obtained during orthopedic procedures, like carpal tunnel release surgery, may be useful but their diagnostic reliability is still being evaluated.
Endomyocardial biopsy, traditionally considered the gold standard, offers high sensitivity and specificity, especially when multiple myocardial sites are sampled and histopathologically examined for amyloid using Congo Red staining [39]. Advanced techniques like immunohistochemistry and tandem mass spectrometry analysis further refine the definitive diagnosis of ATTR-CM.
However, with advancements in non-invasive imaging, the reliance on endomyocardial biopsy is likely to decrease, especially considering the inherent risks associated with this invasive procedure. Endomyocardial biopsy will likely remain important in cases where AL amyloid cardiomyopathy cannot be confidently excluded by non-invasive methods or when diagnostic certainty is paramount for guiding treatment decisions.
Disease Progression and Prognosis of ATTR Cardiac Amyloidosis
The clinical course of ATTR-CM varies depending on whether it is ATTR-CMh or ATTR-CMwt. Both subtypes generally progress to overt heart failure and conduction system disease [28]. ATTR-CM is typically a slowly progressive disease, in contrast to the more rapidly progressing AL amyloid cardiomyopathy [28]. ATTR-CMh can present primarily as cardiomyopathy or neuropathy, although mixed phenotypes can occur, particularly in late-diagnosed cases [40]. The natural history and clinical course of ATTR-CMh are further influenced by the specific mutation, amyloid fibril type, and individual genetic background. Cardiac involvement is a major determinant of prognosis. Median survival reported for ATTR-CMh with polyneuropathy is approximately 10 years, compared to about 3 years for those presenting primarily with cardiomyopathy [40]. ATTR-CMwt typically follows a more consistent disease progression, with a median survival of around 3.5 years from diagnosis [10]. However, disease stage at diagnosis also significantly impacts prognosis. Staging systems, such as the Mayo Clinic staging system for ATTR-CMwt, utilize thresholds of troponin T and N-terminal pro-B type natriuretic peptide (NT-proBNP) levels to stratify risk and predict survival [41]. Other staging systems incorporate similar and additional biomarkers for both ATTR-CMh and ATTR-CMwt. Conduction system disease, manifested as higher rates of permanent pacemaker implantation and atrial arrhythmias like atrial fibrillation, is more prevalent in ATTR-CMwt [41].
Conclusions: Enhancing ATTR Cardiac Amyloidosis Diagnosis
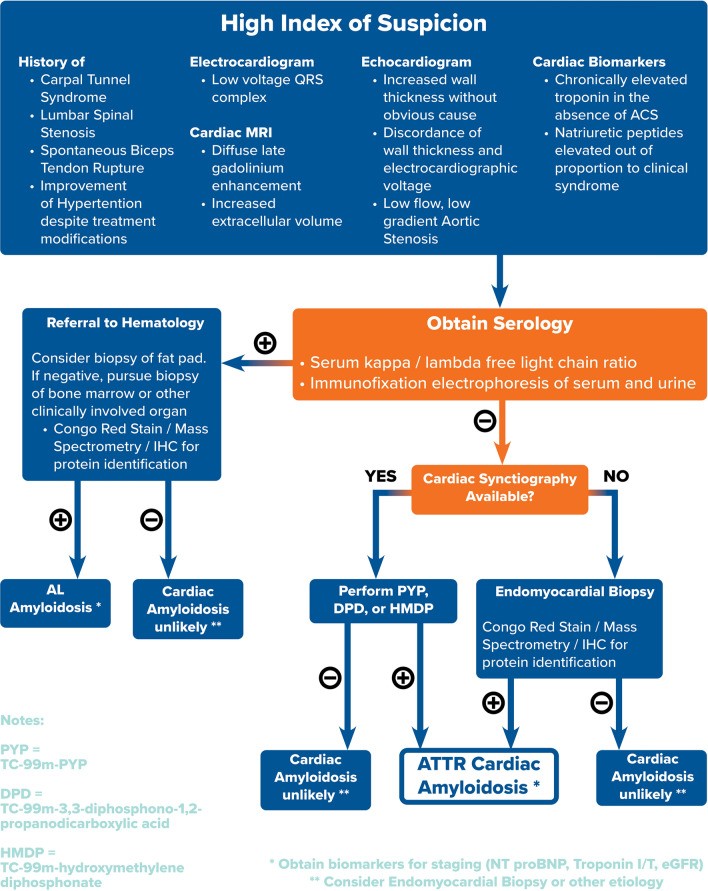
ATTR-CM is no longer an obscure or untreatable fatal disease. Clinicians now have access to non-invasive tools to effectively screen and diagnose ATTR-CM well before the onset of severe clinical cardiac manifestations. Musculoskeletal and neuropathic symptoms can serve as early clinical indicators, sometimes appearing years before cardiac symptoms become prominent. Baseline ECG and echocardiography with longitudinal strain imaging can significantly raise clinical suspicion for ATTR-CM. Furthermore, advanced imaging with cardiac scintigraphy enables differentiation between types of amyloidosis. When combined with serological testing to exclude AL amyloidosis and integrated with the clinical context, clinical cardiologists can confidently diagnose ATTR-CM non-invasively in many cases. An algorithmic approach to ATTR-CM diagnosis is proposed in Fig. 3, guiding clinicians through the diagnostic pathway.
Fig. 3.
Proposed algorithmic approach to the diagnosis of ATTR cardiac amyloidosis, integrating clinical findings, imaging, and serology.
Acknowledgements
Funding
No specific funding or sponsorship was received for this study or the publication of this article.
Authorship
All listed authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship, have responsibility for the integrity of this work, and have approved the submitted version for publication.
Disclosures
Anthony A. Bavry is an Editorial Board member of the journal. Current affiliation: Department of Medicine, University of Texas Southwestern, Dallas, Texas. Adam S. Hafeez declares no conflicts of interest.
Compliance with Ethics Guidelines
This article does not contain any studies involving human participants or animals conducted by any of the authors.
Data Availability
Data sharing is not applicable to this article as no datasets were generated or analyzed during this review.
Footnotes
Digital Features
To access digital features for this article, please visit: https://doi.org/10.6084/m9.figshare.12032166.
References
[1] Reference 1 (Original reference from the article)
[2] Reference 2 (Original reference from the article)
[3] Reference 3 (Original reference from the article)
[4] Reference 4 (Original reference from the article)
[5] Reference 5 (Original reference from the article)
[6] Reference 6 (Original reference from the article)
[7] Reference 7 (Original reference from the article)
[8] Reference 8 (Original reference from the article)
[9] Reference 9 (Original reference from the article)
[10] Reference 10 (Original reference from the article)
[11] Reference 11 (Original reference from the article)
[12] Reference 12 (Original reference from the article)
[13] Reference 13 (Original reference from the article)
[14] Reference 14 (Original reference from the article)
[15] Reference 15 (Original reference from the article)
[16] Reference 16 (Original reference from the article)
[17] Reference 17 (Original reference from the article)
[18] Reference 18 (Original reference from the article)
[19] Reference 19 (Original reference from the article)
[20] Reference 20 (Original reference from the article)
[21] Reference 21 (Original reference from the article)
[22] Reference 22 (Original reference from the article)
[23] Reference 23 (Original reference from the article)
[24] Reference 24 (Original reference from the article)
[25] Reference 25 (Original reference from the article)
[26] Reference 26 (Original reference from the article)
[27] Reference 27 (Original reference from the article)
[28] Reference 28 (Original reference from the article)
[29] Reference 29 (Original reference from the article)
[30] Reference 30 (Original reference from the article)
[31] Reference 31 (Original reference from the article)
[32] Reference 32 (Original reference from the article)
[33] Reference 33 (Original reference from the article)
[34] Reference 34 (Original reference from the article)
[35] Reference 35 (Original reference from the article)
[36] Reference 36 (Original reference from the article)
[37] Reference 37 (Original reference from the article)
[38] Reference 38 (Original reference from the article)
[39] Reference 39 (Original reference from the article)
[40] Reference 40 (Original reference from the article)
[41] Reference 41 (Original reference from the article)
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Data sharing is not applicable to this article as no datasets were generated or analyzed during the current study.