Immunohistochemistry (IHC) stands as a cornerstone technique in modern medical diagnostics, particularly within the realm of lymphoma diagnosis. Utilizing the specific interactions between antigens and antibodies, IHC empowers pathologists to pinpoint precise proteins within cells. This capability unlocks a wealth of critical information, making IHC indispensable for characterizing lymphomas. For professionals dedicated to automotive diagnostics at xentrydiagnosis.store, understanding complex diagnostic processes, even in different fields like medicine, sharpens analytical skills applicable across disciplines.
This article delves into the fundamental applications of immunohistochemistry in lymphoma diagnosis, mirroring the depth and expertise you expect from xentrydiagnosis.store in automotive repair. We will explore how IHC aids in:
- Cell Lineage Identification: Determining whether lymphoma cells originate from B-cells, T-cells, or NK-cells.
- Maturation Phase Assessment: Identifying the stage of development of the lymphoma cells.
- Detection of Genetic Alterations: Revealing specific genetic changes through protein expression patterns.
- Cell Proliferation Visualization: Assessing the rate of lymphoma cell division using markers like Ki-67.
- Therapeutic Target Identification: Pinpointing markers like PD-L1 or CD30 that are crucial for targeted therapies.
Key markers such as CD3, a pan T-cell marker, and CD20, a pan B-cell marker, are foundational in differentiating lymphoma subtypes. Alternatives like CD79a can compensate when CD20 expression is lost. Neuroendocrine marker CD56 serves as an NK cell marker. Furthermore, IHC can detect overexpression of proteins like Bcl-2 and cyclin D-1, indicative of translocations in follicular and mantle cell lymphomas, respectively. The proliferation marker Ki-67 provides insights into tumor aggressiveness. In the era of immunotherapy, IHC plays a vital role in identifying targets like PD-L1, CD19, and CD30. Crucially, proper tissue fixation is paramount for accurate IHC results. Therefore, meticulous procedures from tissue collection to final diagnosis are essential for reliable lymphoma diagnosis.
Keywords: Immunohistochemistry, Lymphoma, Diagnosis, IHC Markers, Lymphoma Subtypes, Diagnostic Techniques
INTRODUCTION to Immunohistochemistry in Lymphoma Diagnostics
Immunohistochemistry (IHC) is a vital diagnostic technique that hinges on the precise interaction between antigens and antibodies to detect specific proteins within cells. Compared to other advanced diagnostic methods, IHC offers remarkable advantages, including cost-effectiveness, rapid turnaround times, and the crucial ability to be performed on formalin-fixed paraffin-embedded tissues, which are standard in pathology labs.
In lymphoma diagnosis, IHC is not merely helpful; it is indispensable. Under standard hematoxylin and eosin (H&E) staining, distinguishing between B and T lymphocytes, the two major types of lymphocytes involved in lymphomas, is often impossible. IHC overcomes this limitation and is routinely used for lymphoma diagnosis for several key reasons:
- Identifying Cell Lineage and Maturation Phase: IHC markers can definitively classify lymphoma cells as B-cell, T-cell, or NK-cell origin and determine their stage of maturity. This is fundamental to classifying the lymphoma accurately.
- Detecting Specific Genetic Alterations: IHC can indirectly reveal the presence of certain genetic abnormalities by detecting the overexpression of proteins that are products of these altered genes.
- Visualizing the Degree of Cell Proliferation: Markers like Ki-67, detectable by IHC, provide a clear picture of how rapidly lymphoma cells are dividing, a critical factor in prognosis and treatment planning.
- Identifying Therapeutic Targets: With the rise of targeted therapies, IHC is essential for identifying markers on lymphoma cells that can be targeted by specific drugs, such as PD-L1 for immunotherapy or CD30 for antibody-drug conjugates.
The selection and interpretation of IHC markers are highly context-dependent, varying with the clinical scenario and the suspected lymphoma type. To provide a practical understanding, this review will guide readers through recommended IHC panels for diagnosing various lymphoma subtypes, enhancing their ability to interpret pathology reports effectively.
RECOMMENDED IHC PANELS for Diffuse Large B-Cell Lymphoma (DLBCL) and Variants
| Essential IHC Markers | CD3, CD20, Ki-67, CD10, Bcl-6, MUM-1, Bcl-2, c-myc, EBV ISH |
|---|---|
| Optional IHC Markers | CD79a, CD30, CD23, ALK, CD138 |
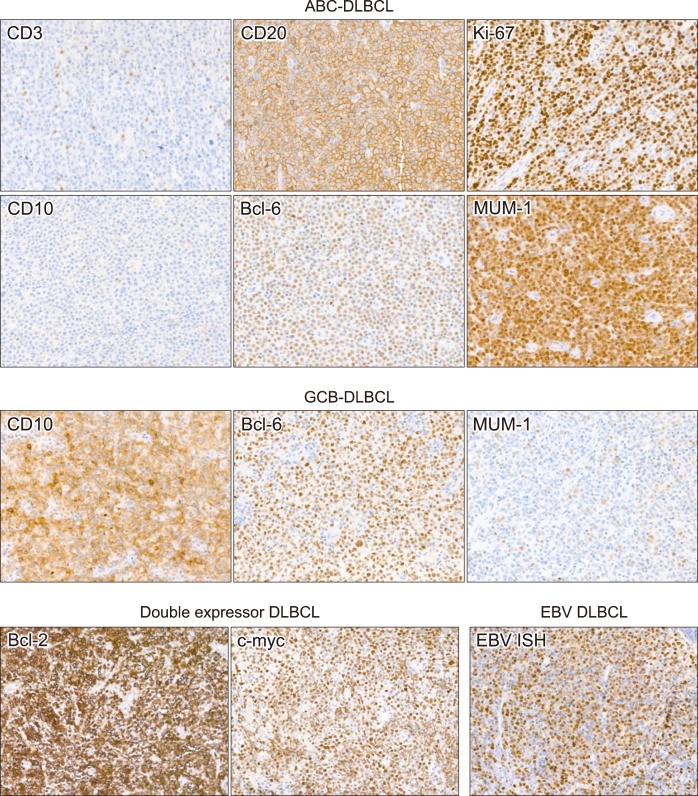
In the current World Health Organization (WHO) classification of tumors of hematopoietic and lymphoid tissues, Diffuse Large B-cell Lymphoma (DLBCL) encompasses DLBCL, Not Otherwise Specified (NOS), and several specific variants. The typical IHC profile of DLBCL NOS is characterized by large, atypical lymphocytes that are CD3-negative and CD20-positive, with a Ki-67 proliferation index exceeding 80%.
Further subclassification, crucial for prognosis and treatment, involves determining the Cell of Origin (COO) using the Hans algorithm. This algorithm relies on IHC staining for CD10 (cutoff: 30%), Bcl-6 (cutoff: 30%), and MUM-1 (Multiple Myeloma 1) (cutoff: 30%). Additionally, the double-expressor phenotype, associated with poorer prognosis, is assessed by staining for Bcl-2 (cutoff: 50%) and c-myc (cutoff: 40%). Epstein-Barr virus in situ hybridization (EBV ISH) is also a vital component, aiding in the detection of EBV-positive DLBCL (EBV DLBCL) (Fig. 1).
Fig. 1.
Alt Text: Immunohistochemistry images showing diffuse large B-cell lymphoma subtypes: ABC, GCB, and EBV DLBCL, illustrating marker expression differences.
Figure 1 illustrates representative IHC staining patterns for different types of diffuse large B-cell lymphomas. Abbreviations used are: ABC (activated B-cell type), DLBCL (diffuse large B-cell lymphoma), GCB (germinal center B-cell type), and EBV DLBCL (EBV-positive diffuse large B-cell lymphoma).
CD10 is normally expressed in centroblasts and centrocytes within reactive germinal centers. According to the Hans algorithm, a DLBCL is classified as germinal center B-cell (GCB) type if more than 30% of tumor cells are CD10-positive, irrespective of Bcl-6 and MUM-1 results. MUM-1 is typically found in plasma cells and post-GCBs; its expression in DLBCL suggests a non-GCB activated B-cell (ABC) type. Bcl-6, like CD10, is a marker of germinal center cells and is also expressed in many ABC-DLBCLs. Therefore, Bcl-6 positivity (≥30%) leads to GCB classification only if MUM-1 is negative.
For diagnosing double-hit lymphoma (high-grade B-cell lymphoma with MYC and BCL2 and/or BCL6 rearrangements), FISH (fluorescence in situ hybridization) testing for MYC rearrangements is ideally performed in all DLBCL cases. Patients with MYC rearrangements should then undergo FISH for BCL2 and BCL6 rearrangements. While c-myc IHC has been suggested as a screening tool for MYC FISH, its results do not reliably correlate with MYC translocation, making it a less recommended screening method.
EBV ISH, while methodologically similar to IHC in slide interpretation, is distinct. In this context, we discuss it alongside IHC due to its diagnostic complementarity. EBV DLBCL NOS is diagnosed if EBV ISH is positive in over 80% of tumor cells in a non-immunodeficient patient. It’s important to note that occasional EBV-positive cells can be bystander B cells in EBV-negative lymphomas.
In instances where DLBCL is suspected based on H&E but tumor cells are CD3 and CD20 negative, differential diagnoses should include poorly differentiated carcinoma or small round cell tumors. However, CD20-negative DLBCL and anaplastic large-cell lymphoma (ALCL) also remain in the lymphoma differential diagnosis. ALCL will be discussed further in the T-cell lymphoma section.
When a suspected DLBCL is CD20-negative, it’s essential to check for a history of rituximab administration, as rituximab can cause CD20 loss. In such cases, CD79a should be assessed to confirm B-cell lineage. For CD20-negative DLBCLs without rituximab history, plasmablastic lymphoma (PBL) or ALK-positive large B-cell lymphoma should be considered. PBL diagnosis is supported by CD138 staining, confirming plasma cell differentiation, and EBV expression. ALK-positive large B-cell lymphoma typically shows negative or weak pan-B-cell markers (CD20, CD79a, PAX5) but strong ALK positivity and plasma cell marker positivity, confirming its B-cell origin despite pan-B marker negativity.
While most DLBCL variants share IHC patterns with DLBCL NOS, primary mediastinal (thymic) large B-cell lymphoma (PMLBL) often expresses CD30 and CD23, aiding in diagnosis. However, sensitivities for CD30 and CD23 are around 80% and 70% respectively, meaning not all PMLBLs express these markers. When both are negative, differentiation from DLBCL NOS relies on tumor location, patient age, and PMLBL-specific histological features, though the distinction can sometimes be challenging.
RECOMMENDED IHC PANELS for Small/Low Grade B-Cell Lymphomas
| Essential IHC Markers | CD3, CD20, Ki-67, Bcl-2, CD10 |
|---|---|
| Optional IHC Markers | CD5, Cyclin D-1, SOX11, CD23, CD21, CD138, Kappa, Lambda, CD79a, IgD, IgG, IgM |
The category of “small/low-grade B-cell lymphomas” is used here for practical purposes to encompass mature B-cell lymphomas not classified as DLBCL or its variants. This avoids the inaccurate assumption that all small cell lymphomas are low-grade and potential confusion with “small lymphocytic lymphoma”. The primary differential diagnoses within this group are follicular lymphoma, chronic lymphocytic leukemia/small lymphocytic lymphoma (CLL/SLL), mantle cell lymphoma (MCL), and marginal zone lymphoma (MZL).
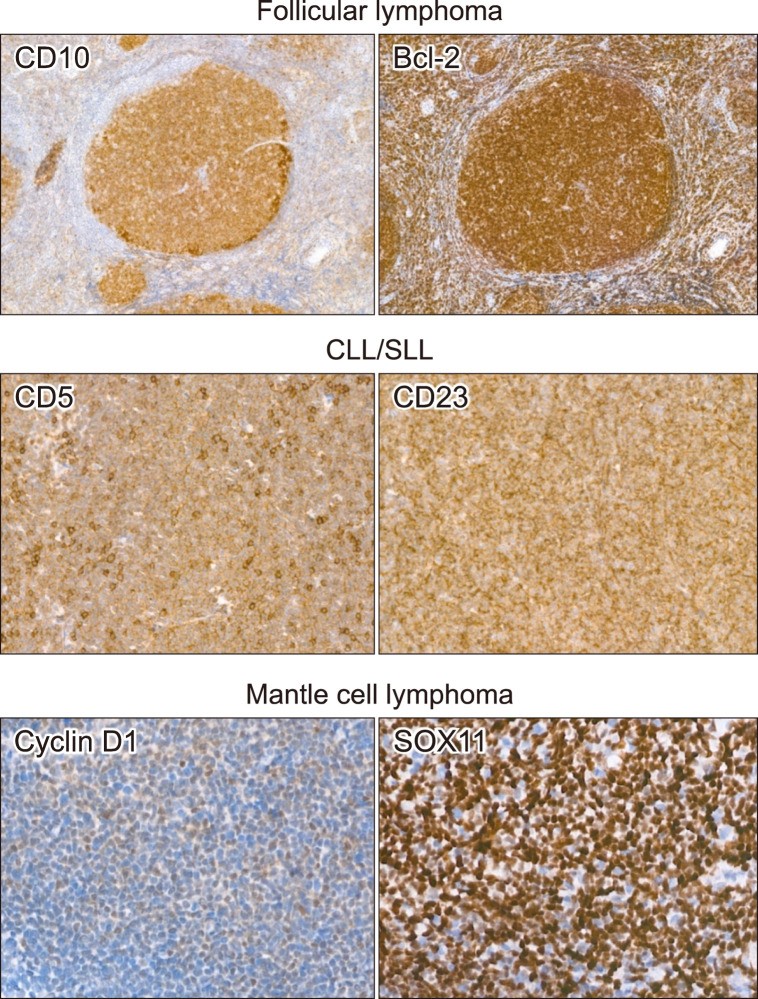
Follicular lymphoma is characterized by the co-expression of CD10 and Bcl-2 (Fig. 2), a highly suggestive IHC finding. CD10 expression indicates origin from centrocytes or centroblasts, similar to GCB-DLBCL. Bcl-2 is typically negative in normal germinal center cells, but BCL2 translocation leads to its aberrant overexpression in follicular lymphoma. While some other small/low-grade B-cell lymphomas may also express both CD10 and Bcl-2, this combination strongly points towards follicular lymphoma. A diagnostic challenge arises from follicular lymphomas lacking CD10 expression, especially in higher grades (3A and 3B), where CD10 negativity is more frequent. Although alternative germinal center markers like LMO2, GCET1, and HGAL (GCET2) exist, they are not routinely recommended. The Ki-67 labeling index in follicular lymphoma is generally around 30% in high-grade forms (grades 3A and 3B), but Ki-67 alone is not used for grading.
Fig. 2.
Alt Text: IHC images showing small B-cell lymphoma subtypes: follicular lymphoma (CD10+, Bcl-2+), CLL/SLL (CD5+, CD23+), and mantle cell lymphoma (Cyclin D-1+, SOX11+).
Figure 2 provides representative IHC staining images of small/low-grade B-cell lymphomas. It highlights follicular lymphoma with CD10 and Bcl-2 positivity, chronic lymphocytic leukemia/small lymphocytic lymphoma (CLL/SLL) with CD5 and CD23 positivity, and mantle cell lymphoma with cyclin D-1 and SOX11 positivity. Abbreviation: CLL/SLL, chronic lymphocytic leukemia/small lymphocytic lymphoma.
For CD10-negative, Bcl-2-positive small/low-grade B-cell lymphomas, the differential diagnosis should include:
CLL/SLL, while less common in Asia compared to the West, remains a crucial differential diagnosis. Characteristic IHC findings include CD3-negative, CD20-positive tumor cells co-expressing CD5 and CD23 (Fig. 2). CD5, primarily a T-cell marker, when diffusely expressed in a B-cell lymphoma, is a strong indicator of CLL/SLL. Reactive T-cells are commonly present in B-cell lymphoma tumor areas, so diffuse CD5 staining pattern in the tumor cells is key. While CD23 is highly sensitive for CLL/SLL, it can be expressed in other small/low-grade B-cell lymphomas, so CD23 expression alone is insufficient for diagnosis.
MCL diagnosis heavily relies on CD5 and cyclin D-1 markers. Cyclin D-1 positivity, resulting from CCND1 translocation, is highly specific for MCL, unlike CD5 which is also positive in CLL/SLL. Cyclin D-1-negative MCLs exist; in these cases, SOX11 staining is diagnostically helpful. Monoclonal SOX11 expression has comparable sensitivity and specificity to cyclin D-1 in MCL, often making it a primary marker for differentiating MCL (Fig. 2). The Ki-67 labeling index in MCL is variable. Unlike other B-cell lymphomas that transform into DLBCL upon large cell transformation, MCL maintains its diagnosis even after high-grade transformation, including blastoid and pleomorphic variants, which can exhibit Ki-67 indices as high as DLBCL.
Nodal and extranodal MZLs lack specific IHC markers or genetic alterations, posing diagnostic challenges. MZL is often diagnosed by exclusion, when a small B-cell lymphoma is negative for CD10, CD5, CD23, cyclin D-1, and SOX11. While some pathologists caution against MZL as a “waste basket diagnosis,” the current WHO classification lacks a more defined category for these cases. MZL originates from post-GCB cells, often showing plasma cell differentiation. In such cases, confirming plasma cell clonality through kappa and lambda light-chain staining can aid in MZL diagnosis. Monoclonality is indicated by a kappa to lambda ratio exceeding 4:1 or lambda to kappa ratio exceeding 2:1, outside the normal range of 2.8–0.7:1. However, plasmacytic differentiation in MZL can blur the distinction from lymphoplasmacytic lymphoma (LPL). While immunoglobulin (IgG, IgA, IgM) staining can help differentiate MZL and LPL, IHC staining alone may not always yield a definitive diagnosis.
RECOMMENDED IHC PANELS for T/NK-Cell Lymphomas
| Essential IHC Markers | CD3, CD20, Ki-67, EBV ISH |
|---|---|
| Optional IHC Markers | CD4, CD8, CD56, CD30, ALK, TIA-1, granzyme B, PD-1, CXCL13, CD10, Bcl-6, ICOS, CD2, CD5, CD7, CD21, β-F1, TCR-delta |
The WHO classification lists numerous T/NK-cell lymphoma types, but initial differential diagnosis typically focuses on peripheral T-cell lymphoma, not otherwise specified (PTCL NOS), angioimmunoblastic T-cell lymphoma (AITL), anaplastic large cell lymphoma (ALCL), and extranodal NK/T-cell lymphoma, nasal type (ENKTL).
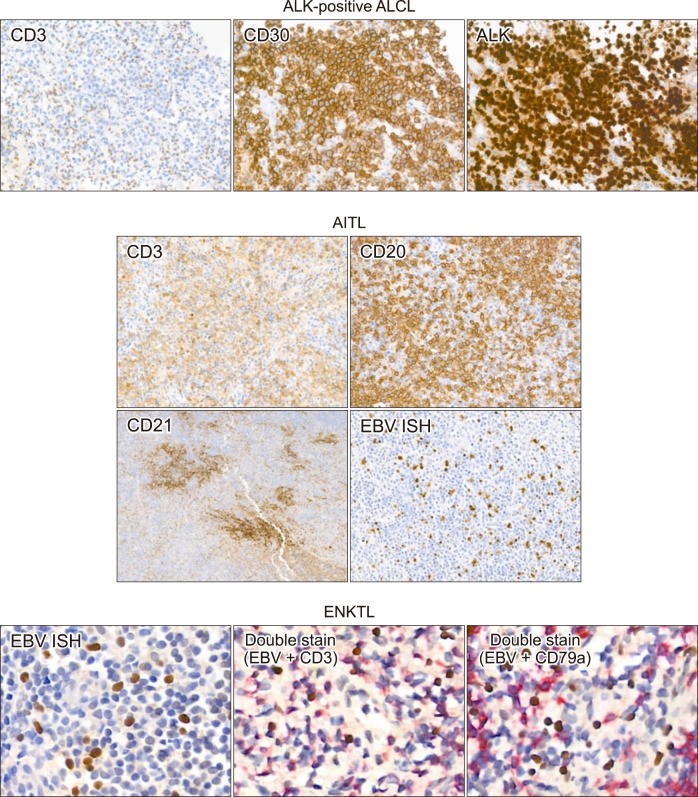
ALCL, although a T-cell lymphoma, often lacks CD3, the pan-T-cell marker. Conversely, CD30 shows strong, diffuse positivity. ALK staining further classifies ALCL into ALK-positive and ALK-negative subtypes (Fig. 3). Due to CD3 negativity in ALK-negative ALCL, additional T-cell markers (CD2, CD5, CD7) and cytotoxic granule markers (TIA-1, granzyme B) are crucial to confirm T-cell origin. Expression of any of these markers supports T-cell lineage. CD30 expression varies in intensity and proportion across different lymphocytes. Differentiating CD30-positive PTCL and ALK-negative ALCL can be challenging, but diffuse and strong CD30 expression is characteristic of ALCL.
Fig. 3.
Alt Text: IHC images of T/NK-cell lymphomas: ALK+ ALCL (CD3-, CD30+, ALK+), AITL (mixed T/B-cells, CD21+ FDC meshwork, EBV+ B-cells), ENKTL (EBV+, CD3+, CD79a- double stain).
Figure 3 displays representative IHC staining images of T/NK-cell lymphomas. These include ALK-positive anaplastic large cell lymphoma with CD3 negativity, CD30 and ALK positivity; angioimmunoblastic T-cell lymphoma showing a mixed T- and B-cell pattern on CD3 and CD20 stains, expanded follicular dendritic cell meshwork on CD21 stain, and scattered large EBV-positive B-cells; and extranodal NK/T-cell lymphoma with EBV-positive cells positive for CD3 and negative for CD79a in double stains. Abbreviations: AITL, angioimmuno-blastic T-cell lymphoma; ALCL, ana-plastic large cell lymphoma; ENKTL, extranodal NK/T-cell lymphoma.
AITL diagnosis based solely on marker immunostaining is often challenging. Tumor cells in AITL do not form diffuse sheets, making them difficult to recognize as T-cell lymphoma using only CD3 and CD20. AITL tumor cells are typically CD4-positive and CD8-negative, being T follicular helper (TFH) cell lymphomas. However, discerning tumor cell staining from background cells with CD4 and CD8 can be difficult. TFH markers like PD-1, CXCL13, CD10, Bcl-6, and ICOS can be more helpful, though each has limited sensitivity and specificity. CD21 is a particularly important IHC marker for AITL. As a follicular dendritic cell (FDC) marker, CD21 highlights the characteristic expansion of the FDC meshwork, which is highly specific for AITL (Fig. 3). AITL frequently induces monoclonal B-cell proliferation, which can lead to misdiagnosis of EBV-positive (or negative) large B-cell proliferation as (EBV-positive) DLBCL (Fig. 3). Therefore, AITL diagnosis requires comprehensive evaluation of IHC results alongside clinical and pathological factors.
Reliable ENKTL diagnosis is significantly enhanced by including EBV ISH in all T/NK-cell lymphoma panels. ENKTL tumor cells are almost always EBV ISH-positive. EBV primarily infects B cells and epithelial cells, not typically T/NK cells. Thus, EBV ISH positivity in T or NK cells strongly suggests a T/NK-cell lymphoproliferative disorder. Most ENKTL tumor cells are CD3-positive (sometimes weakly). CD56 expression is variable depending on the cell lineage. When tumor cell density is low, distinguishing between EBV-positive bystander B cells and EBV-positive tumor T cells can be difficult. In such cases, double staining with EBV ISH–CD3 and EBV ISH–CD20 (or CD79a) is crucial to identify the lineage of EBV-positive cells (Fig. 3).
PTCL NOS encompasses remaining PTCLs not classified into specific types. Tumor cells are CD3-positive, and CD3 staining often accentuates lymphocyte atypia better than H&E staining. Careful assessment for cytologic atypia on CD3-stained slides is essential, requiring robust quality control for immunostaining techniques. Tumor cells in PTCL NOS may express CD4 or CD8, or be negative for both. While individual cells are unlikely to be both CD4- and CD8-positive, both cell types may appear diffusely in low-power views, necessitating careful interpretation. CD56 expression varies within PTCL NOS subtypes. β-F1 and T-cell receptor delta (TCR delta) staining can help determine αβ T-cell or γδ T-cell origin, aiding in differential diagnosis. With the advent of CD30-targeted therapies, CD30 staining is increasingly used in T-cell lymphomas.
RECOMMENDED IHC PANELS for Precursor Lymphomas
| Essential IHC Markers | CD3, CD20, CD79a, Ki-67, TdT, CD99 |
|---|---|
| Optional IHC Markers | CD10, CD19, PAX5, CD5 |
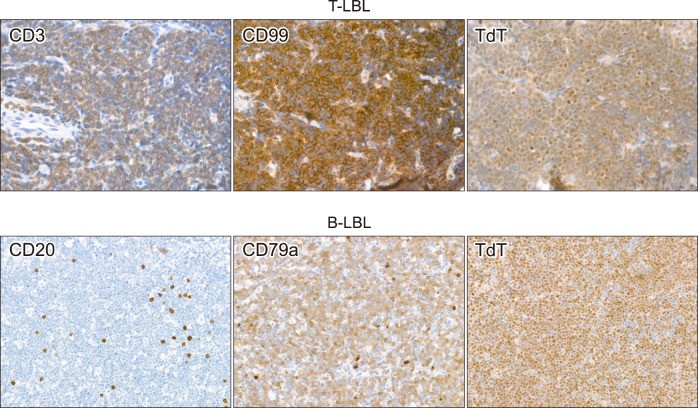
T-lymphoblastic leukemia/lymphoma (T-LBL) and B-lymphoblastic leukemia/lymphoma (B-LBL) are morphologically indistinguishable on H&E-stained slides. Confirmation of a precursor lymphoid neoplasm using terminal deoxynucleotidyl transferase (TdT) and CD99 staining is followed by lineage determination (T-cell or B-cell). T-LBL typically expresses CD3, while B-LBL often lacks CD20. In cases negative for both CD3 and CD20, B-cell lineage confirmation requires additional markers like CD79a, PAX5, or CD19 (Fig. 4). Routine practice often starts with CD3, CD20, and Ki-67 staining, followed by TdT and CD99 based on clinical suspicion of LBL and blastoid morphology.
Fig. 4.
Alt Text: IHC images of precursor lymphomas: T-LBL (CD3+, CD99+, TdT+) and B-LBL (CD20-, CD79a+, TdT+).
Figure 4 presents representative IHC staining images of precursor lymphomas. T-lymphoblastic leukemia/lymphoma shows tumor cells positive for CD3, CD99, and TdT. B-lymphoblastic leukemia/lymphoma shows tumor cells negative for CD20, positive for CD79a, and TdT.
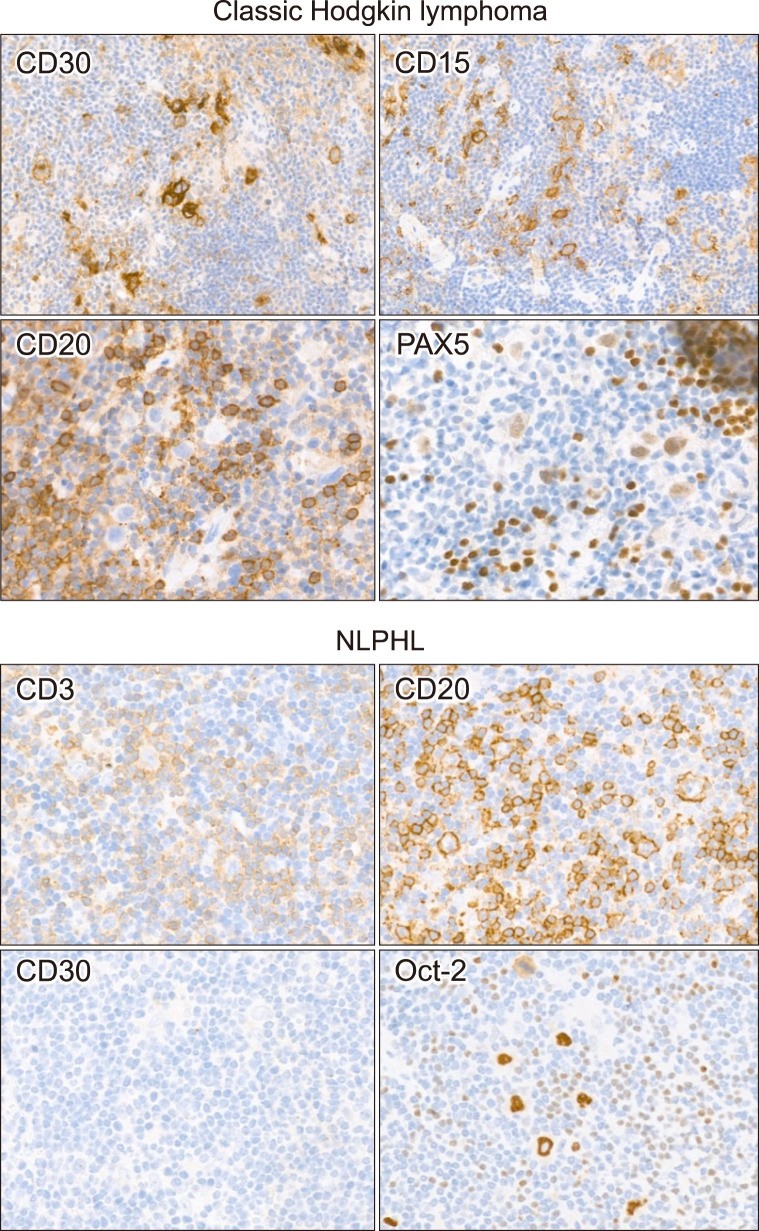
RECOMMENDED IHC PANELS for Hodgkin Lymphomas
| Essential IHC Markers | CD3, CD20, Ki-67, CD30, CD15, PAX5, EBV ISH |
|---|---|
| Optional IHC Markers | Oct-2, BOB1, EMA |
Hodgkin lymphoma broadly divides into classic Hodgkin lymphoma (cHL) and nodular lymphocyte-predominant Hodgkin lymphoma (NLPHL). The diagnostic approach starts with an IHC panel for cHL, with additional staining for NLPHL if cHL is excluded.
cHL tumor cells (Hodgkin-Reed-Sternberg cells or HRS cells) have distinctive morphology. IHC reveals strong CD30 expression (membranous and Golgi patterns). CD20 is negative, and PAX5 is weakly positive in these cells (Fig. 5). CD30 has extremely high sensitivity, being positive in almost all cHL tumor cells. However, CD30 intensity can vary, requiring careful interpretation. While less sensitive than CD30, CD15, when co-expressed with CD30 in the same tumor sample, strongly supports cHL diagnosis. CD15 interpretation requires caution due to frequent positivity in small granulocytes within tumors. EBV ISH positivity varies with cHL variants and does not alter the diagnosis.
Fig. 5.
Alt Text: IHC images of Hodgkin lymphomas: cHL (CD30+, CD15+, CD20-, weak PAX5+ in HRS cells) and NLPHL (CD20+, Oct-2+, CD30- in tumor cells, CD3+ peritumoral rosettes).
Figure 5 shows representative IHC staining images of Hodgkin lymphomas. In classic Hodgkin lymphoma, tumor cells are positive for CD30 and CD15, CD20 is negative, and PAX5 is weakly positive in large tumor cells. In nodular lymphocyte predominant Hodgkin lymphoma, tumor cells are positive for CD20 and Oct-2, and CD30 is negative in tumor cells. CD3 stain shows peritumoral rosette-like T-cells. Abbreviation: NLPHL, nodular lymphocyte predominant Hodgkin lymphoma.
In NLPHL, tumor cells resemble cHL cells morphologically but are CD20-positive and CD30-negative. Differentiation from T-cell/histiocyte-rich large B-cell lymphoma (THRLBL) can be challenging due to scattered CD20-positive, large, atypical cells in both. Nodular patterns of background B-cells on CD20 staining favor NLPHL, but variable low-power patterns complicate differentiation. NLPHL tumor cells are strongly Oct-2-positive, moderately BOB1-positive, and sometimes EMA-positive, but this pattern is similar in THRLBL and does not aid in distinguishing between them (Fig. 5).
CONCLUSION: The Indispensable Role of Basic Immunohistochemistry in Lymphoma Diagnosis
Authors’ Disclosures of Potential Conflicts of Interest
No potential conflicts of interest relevant to this article were reported.