1. Introduction
1.1. Alzheimer’s Disease Pathophysiology and Clinical Presentation
Alzheimer’s disease (AD), the most prevalent cause of dementia, is a progressive neurodegenerative disorder characterized by the accumulation of amyloid-β (Aβ) plaques and neurofibrillary tangles (NFTs) composed of hyperphosphorylated tau protein within the brain. This accumulation disrupts neuronal function, leading to synaptic dysfunction, neuronal loss, and ultimately, cognitive decline. By the time AD is clinically diagnosed, significant neuronal damage has already occurred, making early detection crucial for effective intervention. The pathological hallmarks of AD, including Aβ plaques, NFTs, neuroinflammation, and oxidative stress, begin to develop years before clinical symptoms manifest. The prodromal stage, often referred to as mild cognitive impairment (MCI) due to AD, represents a critical window for early intervention, as symptoms are subtle and not yet overtly indicative of dementia.
The progression of Aβ deposition in the brain follows a hierarchical pattern, starting in the isocortex and spreading to other brain regions, eventually involving the cerebellum and brainstem nuclei. Similarly, tau pathology progresses from the transentorhinal and entorhinal cortex to the isocortex through the hippocampus, accompanied by heterogeneous neuronal loss across different brain areas. Neurofibrillary tangles (NFTs) form within neuronal cell bodies, while neuropil threads (NT) appear in dendrites, and axons contribute to senile plaque formation. The Braak staging system classifies AD neuropathology based on phospho-tau accumulation: stages I–II in the entorhinal cortex, III–IV in the hippocampus/limbic system, and V–VI in the frontal and parietal lobes, reflecting the advancing spread of the disease.
1.2. Current Diagnostic Approaches and the Need for Early Biomarkers
Advancements in neuroimaging techniques, such as magnetic resonance imaging (MRI) and positron emission tomography (PET), have significantly improved AD diagnosis. These techniques allow for in vivo visualization of brain structures, metabolic changes, and amyloid deposition. They enable the detection of pre-symptomatic diagnostic biomarkers in cognitively normal elderly individuals and are also used to monitor disease progression. Volumetric MRI approaches, particularly high-resolution subfield imaging, are valuable for studying early structural changes in brain regions most vulnerable to AD pathology. These tools aid in identifying protein accumulation and structural alterations, forming the basis of current diagnostic practices.
The rise of omics technologies (genomics, transcriptomics, proteomics, metabolomics, secretomics) has further enhanced our ability to analyze AD hallmarks in both sporadic and familial cases. These technologies facilitate the analysis of biofluids like blood, tears, urine, and saliva, offering non-invasive or minimally invasive diagnostic possibilities. Identifying reliable and sensitive biomarkers for early AD diagnosis is paramount. Early diagnosis provides patients with timely access to treatments that may slow disease progression, preserve cognitive function for longer, and allow for better future planning. This review aims to summarize and discuss potential biomarkers for Alzheimer’s disease early diagnosis across various accessible sources, including brain imaging, cerebrospinal fluid (CSF), blood, eyes, saliva, and urine, focusing on their correlation with early disease detection and prognosis.
2. Invasive Biomarkers
2.1. Brain Region-Specific Changes as Early Indicators
Specific brain regions exhibit early pathological changes in AD, making them potential targets for early diagnostic biomarkers.
Locus Coeruleus (LC): The locus coeruleus, a brainstem structure rich in neuromelanin and the primary source of noradrenaline in the brain, is implicated in attention and memory modulation. NFTs are observed in the LC in the asymptomatic stages of AD, preceding their appearance in other brain regions like the entorhinal cortex and neocortex. Tau accumulation in the LC occurs before significant neuronal loss during AD progression. Stereological studies have shown an average 8.4% decrease in LC volume with each Braak stage increment and neuronal loss progressing from 30% in the prodromal stage to 55% in diagnosed dementia. This neuronal loss correlates with cognitive dysfunction and reduced noradrenaline levels in the hippocampus and cortex. In vivo imaging of structural modifications like reduced LC volume or metabolic changes could aid early diagnosis and potentially allow for timely interventions to slow disease progression.
Hippocampus: The medial temporal lobe, particularly the hippocampus, is another brain structure early affected by NFTs and neurite loss. Hippocampal atrophy has been detected in AD patients up to 10 years before dementia diagnosis, even prior to MCI. The extent of atrophy in the hippocampus and its subfields is indicative of progression to MCI or AD. Studies using radial atrophy measurements have shown that CA1 and subicular atrophy in cognitively healthy individuals are associated with an increased risk of MCI, while the progression to CA2–3 subfield atrophy in amnestic MCI suggests future AD diagnosis. Apolipoprotein E ε4 allele (APOEε4), a significant genetic risk factor for AD, exacerbates hippocampal volume reduction and Aβ accumulation in cognitively normal elderly individuals. MRI imaging reveals significant hippocampal volume reduction in APOEε4 carriers with amnestic MCI, especially those progressing to AD. Even cognitively normal APOEε4 carriers exhibit hippocampal volume and cortical thickness reduction, alongside memory decline and accelerated brain atrophy before cognitive impairment onset.
Synaptic Density: Synaptic loss is an early event in AD pathology. A novel PET tracer targeting synaptic vesicle glycoprotein 2A (SV2A) allows for in vivo quantification of synaptic density, which can predict the stage of AD. Markers of neuronal death related to synaptic loss hold promise for early AD diagnosis.
In summary, in vivo morphological studies of brain regions like the LC and hippocampus, combined with genetic studies identifying risk alleles like APOEε4, offer valuable biomarkers for early AD detection. However, despite their high prognostic value, these techniques can be limited for routine clinical use due to cost and accessibility. Consequently, CSF biomarkers are extensively investigated as potential early diagnostic tools for AD.
2.2. Cerebrospinal Fluid (CSF) Biomarkers
Cerebrospinal fluid (CSF) is in direct contact with the brain and reflects biochemical changes occurring in the CNS, making it a rich source of potential AD biomarkers. While CSF collection is invasive, it provides valuable diagnostic information, particularly in the early stages of AD.
Classical CSF Biomarkers: Decreased levels of Aβ-42 and elevated levels of total tau and phospho-tau in CSF are well-established as core AD biomarkers. These biomarkers can predict cognitive decline and are used to support AD diagnosis.
Advantages and Disadvantages of CSF Biomarkers:
Table 1 summarizes the advantages and disadvantages of CSF, blood, and other biofluids as sources of AD biomarkers. CSF offers high diagnostic accuracy and the ability to measure a wide range of pathophysiological biomarkers at high concentrations due to its proximity to the brain. However, its invasive collection limits routine screening and widespread use.
Novel CSF Biomarkers: Advancements in omics technologies are enabling the evaluation of novel molecular markers in CSF. Mass spectrometry-based analyses have revealed that while total ApoE levels in CSF correlate with total tau and phospho-tau, ApoE isoforms do not differ significantly between AD and non-AD individuals in CSF.
APP-Related Proteins: Proteins involved in amyloid precursor protein (APP) processing are also potential early AD biomarkers. Presenilin 1 (PSEN1) and β-secretase 1 (BACE1), enzymes involved in APP cleavage, are found to be increased in CSF of MCI patients. Elevated BACE1 activity is associated with the APOEε4 genotype and is particularly increased in MCI patients progressing to AD. BACE1 shows promise as an early biomarker for amyloidogenic process alterations in APOEε4 carriers but may be less reliable in non-carriers.
Neuroinflammation and Synaptic Dysfunction Markers: Markers of neuroinflammation and synaptic dysfunction in CSF could correlate more directly with cognitive decline. Proteins involved in vesicular transport (secretogranin II (SCG2), chromogranin A (CHGA)), synapse formation (neurexins (NRXNs), neuronal pentraxin 1 (NPTX1), neurocan core protein (NCANP)), and immune response (lysozyme C (LysC) and β2-microglobulin (β2M)) are elevated in CSF of MCI patients, especially those progressing to AD. Higher CHGA levels in CSF of healthy elderly individuals are predictive of future Aβ-42 decreases. Inflammatory proteins YKL-40 and visinin-like protein-1 (VILIP-1) are increased in CSF of MCI and AD patients. YKL-40 levels increase from the prodromal to severe AD stages, while VILIP-1 is elevated primarily in the prodromal stage. Increased YKL-40 is associated with progression from normal cognition to MCI. Interferon-γ-induced protein 10 (IP-10) and monocyte chemoattractant protein 1 (MCP-1), other inflammatory markers, are also elevated in CSF of asymptomatic elderly adults with elevated tau levels and in MCI/AD patients, respectively. Triggering receptor expressed on myeloid cells 2 (TREM2), involved in immune response regulation in the brain, shows increased soluble CSF TREM2 levels years before symptom onset in autosomal dominant AD mutation carriers.
Neuronal Damage Markers: Neurogranin, a protein important for memory enhancement and post-synaptic signaling, shows promise as a CSF biomarker, with levels differentiating early symptomatic AD patients from controls. Neurofilament light chain (NF-L) and neuron-specific enolase (NSE), markers of neuronal damage, are elevated in CSF of MCI patients compared to healthy elderly and advanced AD patients. High CSF NF-L levels in advanced AD correlate with cognitive decline and neuronal loss.
Lipid Biomarkers: Lipid alterations in CSF are also relevant. Sulfatide levels are reduced, while phosphocholine and sphingomyelin levels are increased in CSF of prodromal AD patients, correlating with amyloid and tau pathology. Fatty acid-binding protein (FABP3) is elevated in CSF of MCI and AD patients and is linked to early brain structural changes.
MicroRNAs (miRNAs): MicroRNAs (miRNAs) are small non-coding RNAs that regulate gene expression. CSF miRNAs are being explored as novel AD biomarkers. While CSF collection is invasive, miRNAs offer stability, detectability at low concentrations, and potential to target multiple AD-related genes. Specific miRNAs, such as miRNA-125b and miRNA-146a, are upregulated in CSF of AD patients. miRNA-125b targets synaptic proteins and cell cycle regulators, while miRNA-146a targets immune system regulators and Aβ accumulation. miRNA-29a, which targets BACE1, is increased in CSF of AD patients, despite being decreased in the AD cortex. MiRNA-9, involved in neurogenesis and also targeting BACE1, is upregulated in CSF, neocortex, and hippocampus of AD patients. These miRNAs show potential as both diagnostic and prognostic biomarkers and even as therapeutic targets. Table 2 provides a summary of AD-related miRNAs in CSF and blood.
In conclusion, CSF biomarkers, including classical markers and novel proteins and miRNAs, offer valuable tools for early AD diagnosis, reflecting various aspects of AD pathology from protein misfolding to neuroinflammation and neuronal damage.
3. Noninvasive Biomarkers
3.1. Blood Biomarkers
Blood biomarkers are highly desirable due to their non-invasive nature, making them suitable for routine screening and longitudinal monitoring.
Blood-Based Proteins and Peptides: While blood is a complex matrix, it offers accessibility for AD biomarker research. Blood pressure has been linked to AD pathology, with hypertension associated with increased AD risk. Brain-derived substances can cross the blood-brain barrier and be detected in the bloodstream. Plasma levels of Aβ-42, Aβ-40, and the Aβ-42/Aβ-40 ratio are decreased in preclinical AD patients. Conversely, some studies report elevated Aβ-0 levels in AD patients. Plasma tau levels are also reported to be elevated in AD patients, but low concentrations require highly sensitive detection techniques. Serum neurofilament protein levels correlate with AD, but are not specific to AD. Plasma brain-derived neurotrophic factor (BDNF) and clusterin levels are altered in AD, but their diagnostic utility requires further validation.
Extracellular RNA (exRNA) in Blood: Brain-derived exRNAs can reach the bloodstream via lymphatic drainage or blood-brain barrier leakage, offering a window into CNS gene expression. Phosphoglycerate dehydrogenase (PHGDH) exRNA is upregulated in plasma of presymptomatic AD individuals.
MicroRNAs (miRNAs) in Blood: Blood-based miRNAs are promising non-invasive AD biomarkers. Several miRNAs that downregulate AD-related proteins like BACE-1 and APP are being investigated. Some miRNAs, including miR-31, miR-93, miR-143, and miR-146a, are decreased in serum of AD patients and can differentiate AD from other dementias. miR-206, involved in cognitive decline, is increased in AD plasma. Circulating exRNAs, often protected within exosomes, are stable and detectable in blood.
Brain-Derived Extracellular Vesicles (BEVs) in Blood: Extracellular vesicles (EVs), including exosomes, are membrane-bound vesicles involved in cell-to-cell communication and can cross the blood-brain barrier. Brain-secreted EVs (BEVs) isolated from blood offer a minimally invasive way to sample brain tissue components. BEVs from neurons, neural precursor cells, and astrocytes are under investigation as AD biomarkers.
3.1.1. Neuron-Derived BEVs in Blood
Neuron-derived BEVs in blood show elevated Aβ-42 levels in AD dementia patients compared to cognitively normal individuals. Plasma Aβ-42 in neuron-derived BEVs has potential as an early AD biomarker and a marker of disease progression, comparable to CSF Aβ-42. Tau levels in neuron-derived BEVs are less consistent, with some studies showing elevated phospho-tau in AD, reaching a plateau early in the disease course, and others finding no significant differences. Synaptic proteins in neuron-derived BEVs, such as neurogranin, synaptotagmin, synaptopodin, and synaptophysin, are reduced in AD dementia, but also in MCI and Parkinson’s disease, limiting their specificity. Growth-associated protein 43 (GAP43), synaptosomal-associated protein-25 (SNAP-25), and synapsin 1 are also decreased in neuron-derived BEVs from AD patients. Insulin pathway proteins, specifically phospho-Ser312-insulin receptor substrate-1 (IRS-1) and phospho-panTyr-IRS-1, are deregulated in neuron-derived BEVs in AD. Lysosomal proteins in neuron-derived BEVs, including cathepsin D, lysosome-associated membrane protein, and ubiquitinated proteins, are increased in AD, while heat-shock protein-70 is decreased, suggesting early neuronal lysosomal dysfunction in AD. Repressor element 1-silencing transcription factor (REST) levels are lower in neuron-derived BEVs from AD and MCI patients.
3.1.2. Neural Precursor Cell-Derived BEVs in Blood
Neural precursor cell-derived BEVs, specifically from chondroitin sulfate proteoglycan (CSPG4) cells, show reduced levels of neurotrophic factors (hepatocyte growth factor, fibroblast growth factors 2 and 13, and type 1 insulin-like growth factor) in preclinical AD patients, suggesting their potential as early biomarkers.
3.1.3. Astrocyte-Derived BEVs in Blood
Astrocyte-derived BEVs carry tau, Aβ-42, and APP pathway proteins. Levels of BACE1, sAPPβ, and complement proteins are increased, while glial-derived neurotrophic factor (GDNF) levels are decreased in astrocyte-derived BEVs from AD patients.
3.1.4. MicroRNA Cargo of Blood-Isolated EVs
Exosomal miRNAs in blood are stable and show promise as AD biomarkers. Numerous differentially expressed plasma- and serum-isolated EV-associated miRNAs have been identified in AD and MCI. Exosomal miR-342-3p, miR-125a-5p, miR-125b-5p, and miR-451a are decreased in AD patients and correlate with cognitive impairment. Exosomal miR-223 and exosomal miR-29a also show altered levels in AD. Combinations of miRNAs, such as miR-135a, miR-193b, and miR-384, may offer improved diagnostic accuracy. Table 2 summarizes AD-related miRNAs found in blood and CSF.
Despite the promise of blood biomarkers, challenges remain, including the complexity of blood as a matrix, low concentrations of brain-derived biomarkers, methodological variability, and conflicting results across studies. Further validation is needed for blood biomarkers to be clinically adopted for AD diagnosis.
3.2. Ocular Biomarkers
Ocular biomarkers are attractive due to the eye’s accessibility and the shared pathophysiology between the eye and brain in AD. Retinal and lens changes, and tear fluid analysis are being explored.
Retinal Amyloid and Tau: Aβ depositions are found in the retina and lens of AD patients. Retinal amyloid plaques appear to precede brain plaques in AD models. Retinal Aβ can be detected non-invasively using hyperspectral imaging microscopy or modified endoscopes. Aβ isoforms Aβ1-40 and Aβ1-42 accumulate in the lens of AD patients, correlating with brain amyloid load.
Retinal Structural and Functional Changes: Retinal nerve cell layer thinning, optic nerve atrophy, and retinal ganglion cell loss are reported in AD, leading to visual impairment and circadian rhythm disturbances. Melanopsin retinal ganglion cells (mRGCs), responsible for circadian rhythms, are reduced in AD patients and show Aβ accumulation around them.
Corneal Biomarkers: The cornea, the outermost layer of the eye, is easily accessible. APP and BACE1 expression have been found in the cornea, suggesting its potential as a biomarker source.
Tear Fluid Biomarkers: Tear fluid is a readily accessible biofluid containing proteins and miRNAs. Total protein concentration and composition in tears, as well as tear flow rate and function, are altered in AD patients. Lipocalin-1, dermcidin, lysozyme C, lacritin, and elongation initiation factor 4E (eIF4E) in tears show potential as AD biomarkers. miR-200b-5p is elevated in AD tear fluid. Tear fluid analysis could be a valuable first-line screening tool for AD.
3.3. Salivary Biomarkers
Saliva is a non-invasive and easily collected biofluid. Salivary glands express APP and Aβ, and salivary changes may reflect CSF alterations.
Salivary Aβ and Tau: Studies on salivary Aβ levels in AD show conflicting results. Some studies report no changes in Aβ-40 and variable Aβ-42 levels, while others show increased salivary Aβ-40 and Aβ-42 levels with AD severity. Salivary Aβ-42 may be more associated with familial AD. Salivary total tau levels do not show significant changes in AD, but the phospho-tau/total-tau ratio appears promising.
Lactoferrin and Acetylcholinesterase (AChE) in Saliva: Low salivary lactoferrin levels are associated with increased risk of amnestic MCI and AD dementia. Salivary AChE activity does not appear to be significantly altered in AD.
Further longitudinal studies with larger sample sizes are needed to validate salivary biomarkers for AD.
3.4. Urine Biomarkers
Urine offers a completely non-invasive source of biomarkers, reflecting systemic metabolic changes, including oxidative stress.
Oxidative Stress Markers in Urine: Oxidative stress is prominent in AD. Urinary isoprostane 8,12-iso-iPF(2alpha)-VI levels are elevated in advanced AD stages and may predict MCI-to-AD progression. Oxidized nucleosides in urine, reflecting oxidative DNA damage, are also elevated in AD patients.
Protein Biomarkers in Urine: Alzheimer’s disease-associated neuronal thread protein (AD7c-NTP) in urine shows promise for predicting Aβ plaques in MCI patients. Meta-analyses suggest urinary AD7c-NTP for early AD diagnosis. Albumin, osteopontin, gelsolin, and insulin-like growth factor-binding protein 7 levels in urine are also altered in AD and may serve as novel urinary biomarkers.
Further research is needed to validate urine biomarkers for AD diagnosis.
4. Concluding Remarks and Future Perspectives
Figure 1 and Figure 2 provide schematic overviews of invasive and non-invasive biomarkers discussed. In addition to established CSF biomarkers, accessible fluid biomarkers from blood, tears, saliva, and urine offer promising avenues for AD diagnosis and prognosis.
Blood, particularly neuron-derived BEVs, shows promise, with Aβ-42 levels in BEVs being a consistently reported biomarker. Synaptic proteins and neurotrophic factors in BEVs also warrant further investigation. BEVs from other brain cell types, such as cerebrovascular cells, could enhance blood-based biomarker sensitivity.
Tear fluid, saliva, and urine biomarkers, including proteins, lipids, metabolites, and miRNAs, are emerging as potential non-invasive AD diagnostic tools. Specifically, tear lipocalin-1, salivary lactoferrin and Aβ, and urinary AD7c-NTP and oxidative stress markers are promising candidates.
Inflammatory markers, such as YKL-40, TREM2, and MCP-1, are consistently altered in AD, but may lack specificity for AD versus other neurodegenerative conditions. More specific inflammatory biomarkers are needed for accurate AD diagnosis.
While fluid biomarkers show strong potential for early AD diagnosis, further research is crucial for clinical validation, regulatory qualification, and clinical application. Establishing normative ranges for biomarker levels is essential for clinical utility. Early diagnosis and intervention, guided by these biomarkers, hold the promise to improve the quality of life for AD patients and potentially mitigate the irreversible consequences of this devastating disease.
Table 1. Advantages and disadvantages for each category of biological fluids used to isolate Alzheimer’s disease biomarkers.
| Advantages | Disadvantages |
|---|---|
| CSF | Close relationship with the brain; High accuracy in the diagnostic process; Ability to test a large number of candidate pathophysiological biomarkers; High concentration of the biomarkers |
| Blood | Noninvasive, fast and convenient; Inexpensive and reproducible; Simple to measure (well-established as part of clinical routines globally); No prior training of the clinicians is required; Can be performed in a large variety of settings (primary care, hospitals, patient’s home…); Easy to implement in large populations; Ability to test a large number of candidate pathophysiological biomarkers; First-step of the multi-stage diagnostic process (identification of patients at the earliest stages of the disease) |
| Other matrices (tears, saliva, and urine) | Extremely noninvasive; Repeatable collections; Easy, no risk of infection, can be self-collected by the patient; Cheap; Stress-free |
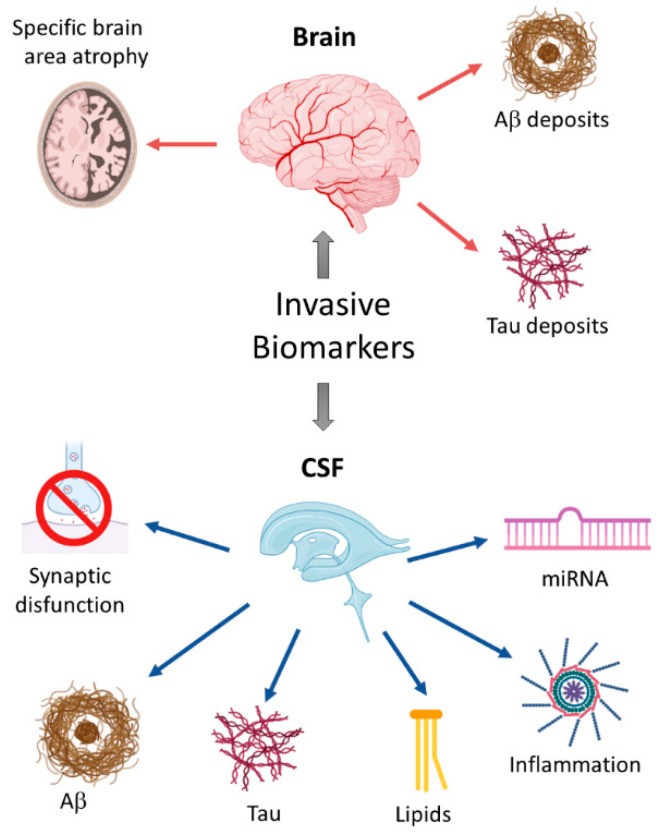
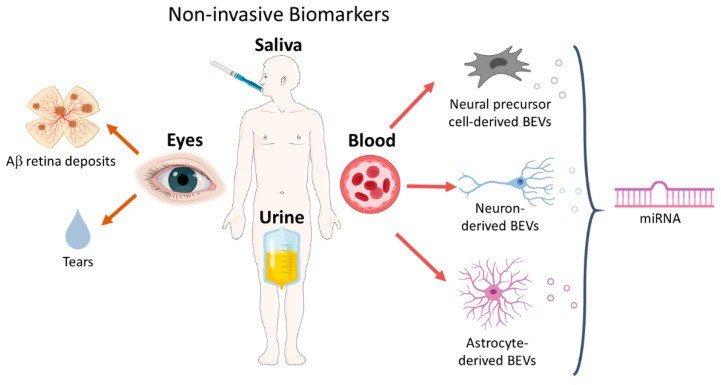
Figure 1. Schematic overview of invasive biomarkers. Different biomarkers have been used to detect early anatomical changes in the brains of people with mild cognitive impairment, including the atrophy of specific brain areas like the locus coeruleus or the hippocampus, and the presence of typical protein aggregates such as extracellular amyloid plaques or intracellular Tau-containing neurofibrillary tangles (upper panel). Additionally, biomarkers of Alzheimer’s disease (AD)-related degenerative processes like synaptic dysfunction, neuroinflammation, oxidative stress, or neuronal loss can be measured in the cerebrospinal fluid of AD patients. The detection of miRNAs represents a novel and promising tool for the early AD diagnosis (lower panel).
Figure 2. Schematic overview of noninvasive biomarkers: eyes, saliva, urine and blood. Besides fluid biomarkers (tears) that can be collected from the eyes, the promising advances in in vivo retinal imaging could provide an AD diagnosis tool in the near future. Blood-isolated brain secreted extracellular vesicles (BEVs) derive from three possible brain cell types: neural precursors, neurons and astrocytes. The content of this blood-isolated BEVs, mainly miRNAs, have been investigated as potential AD biomarker.
Table 2. AD-related main miRNA.
| miRNAs | Regulation and Localization | References |
|---|---|---|
| miR-let-7d-5p, miR-let-7g-5p, miR-26b-5p, miR-191-5p | ↓ Blood | [131] |
| miR-125a-5p | ↓ Blood | [128] |
| miR-126-3p, miR-23a-3p, miR-151a-3p | ↓ Blood | [129] |
| miR-135b | ↓ Blood | [132] |
| miR-181a | ↓ Blood | [133] |
| miR-194-5p | ↓ Blood | [134] |
| miR-19b-3p, miR-29c-3p, miR-125b-3p | ↓ Blood | [135] |
| miR-31, miR-93 | ↓ Blood | [99] |
| miR-3613-3p, miR-3916, miR-4772-3p, miR-185-5p, miR-20b-3p | ↓ Blood | [123] |
| miR-501-3p | ↓ Blood | [136] |
| miR-545-3p | ↓ Blood | [137] |
| miR-181c | ↓ Blood, ↓ Brain | [133,138] |
| miR-139-5p, miR-141-3p, miR-150-5p, miR-152-3p, miR-23b-3p, miR-24-3p, miR-338-3p, miR-342-3p, miR-125b-5p, miR-342-5p | ↓ Blood, ↓ CSF | [123] |
| miR-1306-5p | ↓ Blood, ↓ CSF | [122,139] |
| miR-143 | ↓ Blood, ↓ CSF | [99,133] |
| miR-15b | ↓ Blood, ↓ CSF | [131,133] |
| miR-15b-3p | ↓ Blood, ↓ CSF | [122,139] |
| miR-193b | ↓ Blood, ↓ CSF | [121,124] |
| miR-223 | ↓ Blood, ↓ CSF | [125,140] |
| miR-451a | ↓ Blood, ↓ CSF | [128,139] |
| miR-106, miR-107, miR-181 | ↓ Brain | [69] |
| miR-106b | ↓ Brain | [138] |
| miR-137, miR-139, miR-153, miR-183, miR-135, miR-124b | ↓ Brain | [66] |
| miR-15a, miR-19b, miR-26b, miR-330 | ↓ Brain | [138] |
| miR-425 | ↓ Brain | [133] |
| miR-146b | ↓ Brain, ↓ CSF | [133] |
| miR-210 | ↓ Brain, ↓ CSF | [133,141] |
| miR-10, miR-126, miR-127, miR-154, miR-194, miR-195, miR-199a, miR-214, miR-221, miR-338, miR-422b, miR-451, miR-455, miR-497, miR-99a, miR-27a-3p | ↓ CSF | [133] |
| miR-16-2, miR-16-5p, miR-605-5p, mir-9-5p, miR-598, miR-136-3p | ↓ CSF | [139] |
| miR-200b | ↓ CSF | [142] |
| miR-214-3p, miR-299-5p | ↓ CSF | [132,143] |
| miR-29b-3p | ↓ CSF | [123] |
| miR-29c | ↓ CSF | [134] |
| miR-29 | ↓ Blood, ↓ Brain, ↑Brain | [69,131,133] |
| miR-125b | ↓ Blood, ↑ Brain, ↑ CSF | [65,66,123] |
| miR-146a | ↓ Blood, ↑ Brain, ↑ CSF | [69,71,99] |
| miR-26a | ↓ Brain (frontal cortex), ↑ Brain (hippocampus) | [133] |
| miR-3065-5p | ↓ Blood, ↑ Brain | [122,123] |
| let-7i-5p | ↓ Blood, ↑ CSF | [129,134] |
| miR-106a-5p, miR-20-5p, miR-425-5p, miR-18b-5p, miR-582-5p | ↑ Blood | [122] |
| miR-106b-3p, miR-20b-5p, miR-146a-5p, miR-195-5p, niR-497-5p | ↑ Blood | [135] |
| miR-455-3p, miR-4668-5p | ↑ Blood | [144] |
| miR-5001-3p | ↑ Blood | [123] |
| miR-519 | ↑ Blood | [140] |
| miR-548at-5p | ↑ Blood | [123] |
| miR-590-5p | ↑ Blood | [134] |
| miR-101-3p, miR-106b-5p, miR-143-3p, miR-335-5p, miR-361-5p, | ↑ Blood, ↑ CSF | [122] |
| miR-138-5p | ↑ Blood, ↑ CSF | [123] |
| miR-155 | ↑ Blood, ↑ CSF | [71,131] |
| miR-15a-5p | ↑ Blood, ↑ CSF | [122,134] |
| miR-659-5p | ↑ Blood, ↑ CSF | [123] |
| miR-100, miR-145, miR-148a, miR-27, miR-34a, miR-381, miR-422a, miR-423, miR-92 | ↑ Brain | [133] |
| miR-128 | ↑ Brain | [66] |
| miR-34 | ↑ Brain | [69] |
| miR-98 | ↑ Brain | [138] |
| miR-let-7b, miR-let7e | ↑ CSF | [145] |
| miR-let-7f, miR-105, miR-138, miR-141, miR-151, miR-186, miR-191, miR-197, miR-204, miR-205, miR-216, miR-302b, miR-30a-3p, miR-30a-5p, miR-30b, miR-30d, miR-32, miR-345, miR-362, miR-371, miR-374, miR-375, miR-380-3p, miR-429, miR-448, miR-449, miR-494, miR-501, miR-517, miR-518, miR-520, miR-526 | ↑ CSF | [133] |
| miR-20a-5p | ↑ CSF | [122] |
| miR-222 | ↑ CSF | [146] |
| miR-331-5p, miR-485-5p, miR-132-5p | ↑ CSF | [139] |
| miR-613 | ↑ CSF | [147] |
| miR-200b-5p | ↑ Eyes | [148] |
| miR-93-5p | ↑ ↓ Blood, ↑ CSF | [122,135] |
| miR-101 | ↑ Blood, ↓ Brain | [131,138] |
| miR-132, miR-212 | ↑ Blood, ↓ Brain | [126,133] |
| miR-200c | ↑ Blood, ↓ Brain (frontal cortex), ↑ Brain (hippocampus) | [133,149] |
| miR-9 | ↑ Blood, ↓ Brain (frontal cortex, cortex), ↑ Brain (hippocampus), ↑ CSF | [66,71,131,133,138] |
| miR-30e-5p | ↑ Blood, ↑ Brain, ↑ CSF, | [122,133] |
| miR-29a | ↑ Blood, ↓ Brain, ↑ CSF | [73,74,130] |
| miR-206 | ↑ Blood, ↑ CSF, ↑ Eyes | [100,150] |
| miR-142-5p | ↑ Blood, ↓ CSF | [133,134] |
| miR-384 | ↑ Blood, ↓ CSF | [124] |
| miR-135a | ↑ Blood, ↓ CSF, ↑ CSF | [124,133,142] |
| miR-125a | ↓ Brain, ↑ CSF | [66,133] |
| miR-29b | ↓ Blood, ↓ Brain, ↑ CSF | [73,74,131] |
| miR-30c | ↑ Brain (frontal cortex), ↓ Brain (hippocampus), ↑ CSF | [133] |
Acknowledgments
Figures created with BioRender.com.
Author Contributions
E.A. and G.E. carried out the literature review, conceptualized and prepared the initial draft. G.E. provided critical inputs. V.G.-V. edited and contributed in the final manuscript. All authors have read and agreed to the final version of the manuscript.
Funding
This research has not received any specific funding.
Conflicts of Interest
The authors declare no conflict of interest.
References
[1] Swerdlow, R.H. Alzheimer’s Disease, Mitochondria, and Noncanonical Glucose Metabolism. J. Pers. Med. 2020, 10, 114. https://doi.org/10.3390/jpm10030114
[2] Braak, H.; Alafuzoff, I.; Arzberger, T.; Kretzschmar, H.; Del Tredici, K. Staging of Alzheimer disease-associated neurofibrillary pathology using paraffin sections and immunocytochemistry. Acta Neuropathol. 2006, 112, 389–404.
[3] Braak, H.; Braak, E. Neuropathological stageing of Alzheimer-related changes. Acta Neuropathol. 1991, 82, 239–259.
[4] Thal, D.R.; Rüb, U.; Orantes, M.; Braak, H. Phases of A beta-deposition in the human brain and its relevance for the development of AD. Neurology 2002, 58, 1791–1800.
[5] German, D.C.; Manaye, K.F.; Sonsalla, P.K.; Brooks, B.A. Midbrain dopaminergic cell loss in Parkinson’s disease and incidental Lewy body cases. Ann. Neurol. 1989, 26, 507–514.
[6] Gibb, W.R.; Lees, A.J. The relevance of the Lewy body to the pathogenesis of idiopathic Parkinson’s disease. J. Neurol. Neurosurg. Psychiatry 1988, 51, 745–752.
[7] Baker-Nigh, A.T.; Lucin, K.M.; Veglia, E.;diameter,diameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameterdiameter
