Diamond Blackfan anemia (DBA) is a rare, inherited bone marrow failure syndrome characterized by a failure of the bone marrow to produce red blood cells. This condition often presents in infancy and is associated with congenital anomalies and an increased predisposition to cancer. Accurate and timely Blackfan Diamond Anemia Diagnosis is crucial for effective management and improved patient outcomes. Recent advancements in understanding the genetic and molecular basis of DBA have refined diagnostic approaches, making it possible to identify both classical and non-classical presentations of this complex disorder.
Understanding Diamond Blackfan Anemia (DBA)
Diamond Blackfan anemia (DBA; MIM #205900) belongs to a group of genetic disorders known as inherited bone marrow failure syndromes (IBMFS). These syndromes share common features, including pro-apoptotic hematopoiesis, bone marrow failure, birth defects, and, in many cases, a predisposition to cancer. DBA specifically is recognized as a “ribosomopathy,” stemming from defects in ribosome biogenesis. Ribosomes are essential cellular components responsible for protein synthesis, and their malfunction in DBA leads to a cascade of cellular stresses, particularly affecting erythroid progenitor cells.
Key Characteristics of DBA
The hallmark of DBA is erythroid failure, meaning the bone marrow fails to produce sufficient red blood cells. This leads to severe anemia, often presenting in the first year of life. Beyond anemia, DBA is characterized by:
- Congenital Anomalies: A significant proportion of individuals with DBA have birth defects, affecting various organ systems.
- Cancer Predisposition: Patients with DBA have an increased risk of developing certain cancers, including leukemia and solid tumors.
Diagnostic Criteria for Diamond Blackfan Anemia
The classical diagnostic criteria for DBA, established in 1976, include:
- Anemia presenting before the first birthday.
- Near normal or slightly decreased neutrophil counts.
- Variable platelet counts.
- Reticulocytopenia (low count of immature red blood cells).
- Macrocytosis (abnormally large red blood cells).
- Normal bone marrow cellularity with a marked reduction in red cell precursors.
These criteria have been the cornerstone of Blackfan Diamond anemia diagnosis for decades. However, with increased understanding of the genetic heterogeneity of DBA, the diagnostic landscape has broadened to include non-classical presentations.
Supporting Features in DBA Diagnosis
In addition to the core criteria, certain supporting features enhance the likelihood of a Blackfan Diamond anemia diagnosis:
- Elevated Fetal Hemoglobin (HbF): Increased levels of HbF are commonly observed in DBA, reflecting stress erythropoiesis.
- Elevated Erythrocyte Adenosine Deaminase (eADA) Activity: An unexplained but significant finding in about 85% of DBA patients is elevated eADA activity in red blood cells.
- Macrocytosis: While part of the classical criteria, the degree of macrocytosis can be a strong indicator, especially when other causes are excluded.
It’s important to note that macrocytosis and elevated HbF are not exclusive to DBA and can be seen in other bone marrow failure conditions or during recovery from anemia. However, in the context of other suggestive findings, they strengthen the suspicion for DBA.
Non-Classical DBA Diagnosis
The discovery of genes mutated in DBA has revealed that the clinical presentation can be highly variable. This has led to the recognition of “non-classical” DBA, where individuals may not fully meet the traditional diagnostic criteria. Non-classical DBA presentations may include:
- Minimal or No Anemia: Some individuals, particularly gene carriers within families, may have very mild or no anemia.
- Macrocytosis Alone: Isolated macrocytosis without significant anemia can be an indicator in some cases.
- Adult Onset Presentation: While rare, DBA can present in adulthood.
- Phenotypically Normal Parents of Affected Offspring: Due to variable penetrance, parents carrying a DBA gene mutation may show no hematologic abnormalities.
- Congenital Anomalies with Minimal Erythroid Abnormalities: In some instances, congenital anomalies may be the most prominent feature, with subtle or absent hematologic signs.
This expanded understanding of DBA necessitates a comprehensive diagnostic approach, considering both classical and non-classical features, especially in familial cases or individuals with suggestive congenital anomalies.
Differential Diagnosis of Diamond Blackfan Anemia
When considering a Blackfan Diamond anemia diagnosis, it’s essential to differentiate it from other conditions presenting with red cell aplasia or anemia in infancy and childhood.
Distinguishing DBA from Transient Erythroblastopenia of Childhood (TEC)
Transient erythroblastopenia of childhood (TEC) is the most common differential diagnosis in children presenting with red cell failure. TEC is an acquired condition characterized by a temporary suppression of red blood cell production, typically resolving within a few months. Key distinctions between DBA and TEC are summarized below:
| Feature | Diamond Blackfan Anemia (DBA) | Transient Erythroblastopenia of Childhood (TEC) |
|---|---|---|
| Age of Onset | Younger than 1 year | Older than 1 year |
| Inheritance | Sporadic and dominant | Not inherited |
| Congenital Anomalies | Present | Absent |
| Mean Corpuscular Volume (MCV) | Elevated | Normal (elevated during recovery) |
| Fetal Hemoglobin (HbF) | Elevated | Normal (elevated during recovery) |
| Erythrocyte ADA (eADA) | Elevated (in ~85% cases) | Normal |
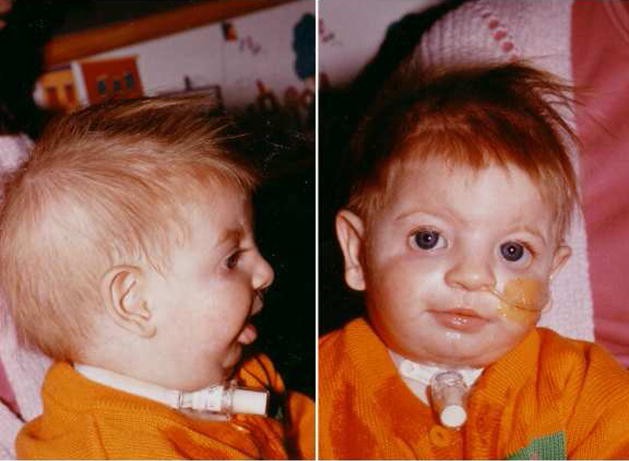
TEC is thought to be an acquired, likely post-viral, autoimmune condition. Unlike DBA, TEC is not associated with congenital anomalies or a family history of anemia. While macrocytosis and elevated HbF can occur in TEC during recovery (“stress erythropoiesis”), eADA activity remains normal, a crucial differentiating factor. In cases where differentiation is challenging, close monitoring and potentially limited red cell transfusions may be necessary until a definitive diagnosis can be established.
Other Conditions in Differential Diagnosis
Besides TEC, other conditions to consider in the differential diagnosis of DBA include:
- Pearson Syndrome: Another rare inherited bone marrow failure syndrome, often presenting with pancreatic insufficiency and sideroblastic anemia.
- Acquired Pure Red Cell Aplasia (PRCA): Various acquired conditions can lead to PRCA, including autoimmune disorders, infections (especially parvovirus B19), thymoma, and certain medications.
- Congenital Infections: Infections like cytomegalovirus or rubella can cause anemia in newborns.
- Nutritional Deficiencies: Severe folate or vitamin B12 deficiencies, though rare in developed countries, can cause macrocytic anemia.
- Other Inherited Bone Marrow Failure Syndromes: While distinct, syndromes like Fanconi anemia and Shwachman-Diamond syndrome share some overlapping features and should be considered, especially in atypical presentations.
A thorough clinical evaluation, appropriate laboratory investigations, and genetic testing are essential to accurately distinguish DBA from these other conditions.
The Diagnostic Process for DBA
The process of Blackfan Diamond anemia diagnosis involves a combination of clinical assessment, hematological investigations, and genetic testing.
Clinical Evaluation and History
A detailed clinical evaluation is the first step in the diagnostic process. This includes:
- Medical History: Assessing the age of onset of anemia, history of transfusions, and any other hematologic abnormalities.
- Family History: Inquiring about family history of anemia, congenital anomalies, or known cases of DBA or other inherited disorders.
- Physical Examination: Careful examination for congenital anomalies, particularly craniofacial features, thumb abnormalities, and short stature, which are commonly associated with DBA.
Hematological Tests
Specific hematological tests are crucial for confirming the diagnosis and excluding other causes of anemia:
- Complete Blood Count (CBC) with Red Cell Indices: To assess hemoglobin levels, red blood cell count, white blood cell count, platelet count, MCV (mean corpuscular volume), and other red cell indices. Macrocytosis is a key finding.
- Reticulocyte Count: Typically low in DBA, indicating bone marrow failure to produce new red blood cells.
- Bone Marrow Aspirate and Biopsy: Essential to evaluate bone marrow cellularity and the presence or absence of red cell precursors (erythroid precursors). In DBA, the marrow is usually cellular but shows a marked reduction in erythroid precursors.
- Fetal Hemoglobin (HbF) Level: Measurement of HbF levels, which are often elevated in DBA.
- Erythrocyte Adenosine Deaminase (eADA) Activity: eADA levels in red blood cells should be measured, as elevation is a strong supporting feature for DBA.
Genetic Testing for DBA
Genetic testing has become an increasingly important tool in Blackfan Diamond anemia diagnosis. Identifying a causative gene mutation can:
- Confirm the Diagnosis: Especially in cases with atypical presentations or when classical criteria are not fully met.
- Facilitate Family Screening: Allows for testing of family members at risk, including asymptomatic carriers.
- Inform Genetic Counseling: Provides valuable information for reproductive planning and assessing recurrence risk.
- Potentially Guide Prognosis and Treatment: While genotype-phenotype correlations are still being investigated, specific gene mutations might have implications for disease severity or cancer risk.
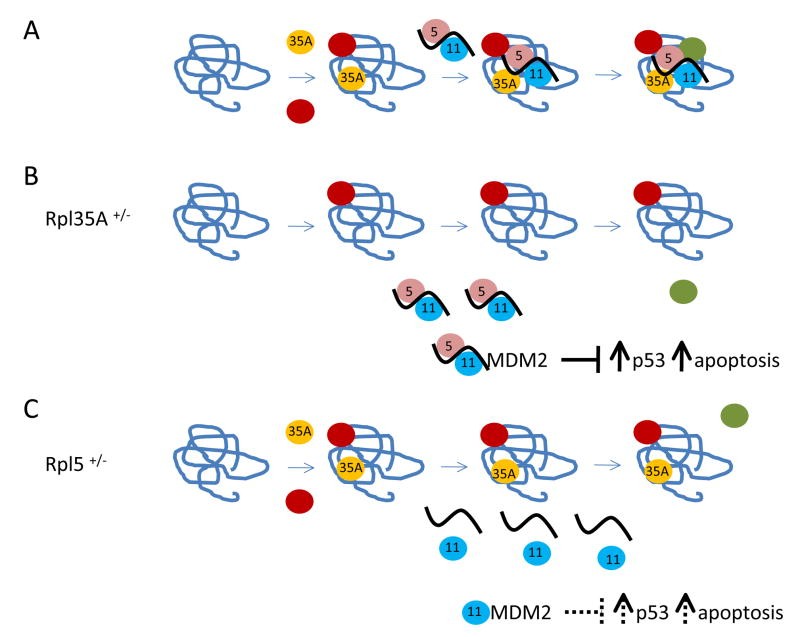
Currently known DBA genes primarily encode ribosomal proteins. Mutations in RPS19 were the first to be identified and remain the most common, accounting for approximately 25% of cases. Other identified genes include RPL5, RPL11, RPS24, RPS17, and RPL35A. Ongoing research continues to identify additional DBA-related genes.
Genetic testing typically involves sequencing these known genes. If a mutation is identified in a proband (the first affected family member), family members can be tested for the same mutation, even if they are asymptomatic, to identify carriers or individuals with non-classical DBA.
Importance of Early and Accurate DBA Diagnosis
Early and accurate Blackfan Diamond anemia diagnosis is paramount for several reasons:
Impact on Treatment Decisions
A definitive DBA diagnosis guides appropriate treatment strategies. The mainstays of treatment for DBA include:
- Corticosteroids: Effective in inducing remission in a significant proportion of patients. However, long-term use is associated with side effects.
- Red Blood Cell Transfusions: Used to manage anemia, especially in steroid-refractory patients. Chronic transfusions lead to iron overload, requiring chelation therapy.
- Hematopoietic Stem Cell Transplantation (HSCT): Potentially curative option, particularly for transfusion-dependent patients or those with severe steroid side effects.
Accurate diagnosis ensures that patients receive timely and appropriate treatment, minimizing complications and improving quality of life.
Genetic Counseling and Family Screening
A confirmed DBA diagnosis has significant implications for genetic counseling. Understanding the inheritance pattern (primarily autosomal dominant, but sporadic cases occur) and recurrence risk is crucial for families planning future pregnancies. Family screening can identify other affected individuals or carriers, allowing for proactive management and genetic counseling for extended family members.
Cancer Risk Surveillance
Given the increased cancer predisposition in DBA, early diagnosis allows for appropriate cancer surveillance strategies. While specific guidelines are still evolving, increased awareness and vigilance for signs and symptoms of cancer are essential in DBA patients.
Advancements in DBA Diagnosis
Significant advancements have been made in Blackfan Diamond anemia diagnosis, driven by research and collaborative efforts.
Genetic Discoveries and Expanded Diagnostic Capabilities
The identification of multiple DBA-related genes has revolutionized diagnosis. Genetic testing is now a crucial component, enabling the diagnosis of non-classical cases and facilitating family studies. Ongoing research to identify the remaining DBA genes promises to further refine diagnostic capabilities and potentially uncover genotype-phenotype correlations.
Role of DBA Registries in Improving Diagnosis and Understanding
International DBA registries play a vital role in advancing our understanding of DBA. These registries collect clinical, genetic, and outcome data from a large number of patients, providing invaluable insights into the natural history of the disease, genotype-phenotype relationships, and treatment outcomes. Registry data has contributed to refined diagnostic criteria, improved treatment guidelines, and a better understanding of the long-term complications of DBA.
Conclusion
Blackfan Diamond anemia diagnosis is a multifaceted process requiring careful clinical evaluation, hematological investigations, and increasingly, genetic testing. The recognition of both classical and non-classical presentations of DBA, coupled with advancements in genetic diagnostics, has improved our ability to accurately identify affected individuals. Early and accurate diagnosis is crucial for guiding treatment decisions, facilitating genetic counseling, and implementing appropriate cancer surveillance strategies. Ongoing research and collaborative efforts, particularly through patient registries, continue to refine our understanding of DBA and pave the way for further improvements in diagnosis and management.