Introduction
In the intricate world of automotive repair, understanding complex systems is paramount. Just as a vehicle’s engine relies on a delicate balance of fluids and pressures, the human body depends on a sophisticated hemostatic system to prevent excessive bleeding. Hereditary bleeding disorders, stemming from deficiencies in specific clotting proteins, can significantly impact an individual’s health. As content creators for xentrydiagnosis.store and experts in automotive diagnostics, we recognize the importance of understanding complex systems, whether in vehicles or the human body. This article provides a comprehensive overview of hereditary bleeding disorders, focusing specifically on Bleeding Disorders Diagnosis, to enhance your understanding and awareness of these conditions.
The body’s natural ability to control bleeding after injury is crucial. This process, known as hemostasis, involves a complex interplay of blood vessels, platelets, and coagulation factors. When this system malfunctions due to inherited conditions, it leads to bleeding disorders. The three most prevalent hereditary bleeding disorders are hemophilia A (factor VIII deficiency), hemophilia B (factor IX deficiency), and von Willebrand disease. Early and accurate bleeding disorders diagnosis is essential for effective management and improving the quality of life for affected individuals. This article will delve into the evaluation process for these disorders, treatment options, and the collaborative care required for patients.
Understanding Hereditary Bleeding Disorders: Etiology and Epidemiology
Hereditary bleeding disorders, also known as congenital coagulopathies, arise from genetic mutations that disrupt the production or function of clotting factors. These disorders are distinct from acquired bleeding disorders, which develop later in life due to other medical conditions. Focusing on inherited conditions, hemophilia A and B are X-linked recessive disorders. This inheritance pattern means that males are predominantly affected, as they have only one X chromosome. Approximately one-third of hemophilia cases occur due to spontaneous genetic mutations, highlighting the unpredictable nature of these conditions.
Von Willebrand disease (VWD), in contrast, follows an autosomal dominant inheritance pattern. This means that both males and females can be affected, and a single copy of the mutated gene is sufficient to cause the disorder. VWD exhibits variable penetrance, meaning the severity of symptoms can differ significantly even within the same family. Homozygotes, who inherit two copies of the mutated gene, typically experience the most severe form of VWD. Neither hemophilia nor VWD shows a predisposition for any specific racial group, affecting populations globally.
Epidemiologically, hereditary bleeding disorders, while individually rare, collectively impact a significant number of individuals. Hemophilia A, the most common congenital coagulopathy, affects approximately 1 in 5,000 males. Hemophilia B is less frequent, occurring in about 1 in 30,000 male births. The higher prevalence of hemophilia in populations with increased consanguinity underscores the genetic component of these diseases. Females can be carriers of hemophilia genes, often without symptoms, or may experience mild bleeding tendencies due to partial factor deficiencies.
Von Willebrand disease is considerably more common, affecting approximately 1% of the general population, according to the Centers for Disease Control and Prevention (CDC). While VWD affects both sexes equally, women are more likely to experience noticeable symptoms due to increased bleeding during menstruation. The wide range in prevalence and symptom severity emphasizes the importance of accurate bleeding disorders diagnosis to tailor appropriate management strategies.
Pathophysiology and Clinical Presentation
The clinical presentation of bleeding disorders is closely linked to their severity. Mild cases might remain undiagnosed until a significant event like surgery or trauma occurs. However, severe forms often manifest early in life. In severe hemophilia, characterized by less than 1% of normal clotting factor levels, individuals may experience frequent spontaneous bleeding episodes, such as epistaxis (nosebleeds) occurring 20 to 30 times annually. Excessive bleeding after minor injuries, and hemarthrosis (bleeding into joints), are also common hallmarks. Diagnosis of severe hemophilia is often made within the first two years of life, sometimes becoming apparent immediately after circumcision or at birth with intracranial hemorrhages or umbilical cord bleeding.
Moderate hemophilia, with factor levels between 6% to 30% of normal, may present with less frequent spontaneous bleeding. However, these individuals are still at risk of excessive bleeding following surgery or major trauma. The variability in presentation highlights the need for comprehensive bleeding disorders diagnosis even in cases with milder symptoms.
Von Willebrand disease pathophysiology centers around a deficiency or dysfunction of von Willebrand factor (vWF), a crucial glycoprotein that facilitates platelet adhesion and stabilizes factor VIII. VWD symptoms can mimic platelet disorders, ranging from mild mucocutaneous bleeding like easy bruising, gingival bleeding, or menorrhagia (heavy menstrual bleeding), to more severe hemarthrosis in rarer instances. VWD is classified into three main types. Type 1 VWD, the most common (60-80% of cases), involves a quantitative deficiency of vWF, typically with 20% to 40% of normal levels, and may also include reduced factor VIII levels. Patients with Type 1 VWD often present with mild symptoms like easy bruising or mucosal bleeding. Certain medications, such as NSAIDs and aspirin, which inhibit prostaglandin and thromboxane synthesis, can exacerbate bleeding symptoms and may aid in initial bleeding disorders diagnosis.
Type 2 VWD (approximately 17% of cases) is characterized by a qualitative defect in vWF function. Subtypes within Type 2 include 2A (abnormal vWF multimers), 2B (hyperactive vWF leading to thrombocytopenia), 2M (decreased platelet binding), and 2N (decreased factor VIII binding). Type 3 VWD, the rarest and most severe form (about 3% of cases), involves a near-complete absence of vWF and significantly reduced factor VIII levels. Understanding these different types is crucial for accurate bleeding disorders diagnosis and targeted treatment strategies.
Figure: Visual representation of various bleeding disorders impacting different stages of the coagulation cascade.
History, Physical Examination, and Evaluation
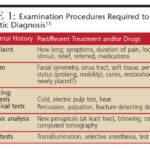
A thorough patient history and physical examination are crucial first steps in bleeding disorders diagnosis. In hemophilia, while mild cases may only surface after trauma or surgery, moderate to severe forms often present with characteristic signs. These include mucosal and gingival bleeding, easy bruising, and hematoma formation. Hemarthrosis, bleeding into joints like ankles, knees, and elbows, is a significant concern. Recurrent hemarthrosis can lead to chronic joint damage, synovitis, and hemophilic arthropathy, characterized by joint narrowing, bone cysts, and limited range of motion. Muscle bleeds, even from minor injuries, can cause anemia and compartment syndrome due to compression of nerves and vital structures. Intracranial hemorrhage is a life-threatening emergency in hemophilia patients. Infants with hemophilia may present with cephalohematoma after vaginal delivery or significant bleeding post-circumcision.
Von Willebrand disease often manifests in childhood with easy bruising and bleeding history. While very mild VWD may be asymptomatic, those with qualitative or quantitative vWF deficiencies typically exhibit mucocutaneous bleeding and recurrent epistaxis. Menorrhagia is a common presenting symptom in women with VWD, often prompting investigation and bleeding disorders diagnosis. Similar to hemophilia, VWD may remain undiagnosed until major surgery or trauma.
Laboratory evaluation is indispensable for confirming bleeding disorders diagnosis. Initial screening tests usually include platelet count, prothrombin time (PT), partial thromboplastin time (aPTT), international normalized ratio (INR), and bleeding time. A normal platelet count ranges from 150,000 to 500,000/ml, and platelet function is assessed by bleeding time. Prolonged bleeding times can indicate vWF deficiency or platelet function-inhibiting medications like NSAIDs and aspirin.
PT measures the function of coagulation factors II, V, VII, and X, vitamin K-dependent factors synthesized in the liver. PT and INR are prolonged by warfarin, which interferes with vitamin K-dependent factor synthesis. INR, typically 0.8 to 1.2, estimates the percentage of functional clotting factors; an INR of 2 to 3 suggests approximately 10% factor activity. At least 30% factor activity is needed for normal coagulation. aPTT assesses the intrinsic pathway, involving factors VIII, IX, XI, XII, and vWF.
In hemophilia, aPTT is characteristically prolonged, while PT/INR, bleeding time, and platelet count are usually normal. Elevated aPTT prompts further investigation of individual factor levels, specifically factor VIII and IX, to confirm bleeding disorders diagnosis and determine disease severity. Family history, particularly of maternal male relatives, is also crucial.
Bleeding disorders diagnosis for von Willebrand disease involves assessing bleeding time and vWF levels using the ristocetin cofactor activity assay. A history of bleeding and positive family history further support the diagnosis. Due to vWF level fluctuations with stress, pregnancy, exercise, and inflammation, repeated testing (at least three times) is recommended for accurate diagnosis. Subtype-specific diagnosis of VWD requires factor multimer assays. aPTT may be mildly elevated in about 50% of VWD patients due to reduced factor VIII levels.
Treatment and Management Strategies
The primary treatment for hemophilia is factor replacement therapy, administered either prophylactically to prevent bleeding or on-demand to treat acute bleeds. Recombinant factor VIII and IX concentrates are preferred due to their safety profile, eliminating the risk of viral transmission associated with plasma-derived products. Recombinant factor VIII raises plasma concentration by 2% per IU/kg, and factor IX by 0.8% per IU/kg. Treatment goals aim for 50% plasma factor levels for managing bleeding risks like surgery, and 100% for major trauma. Factor levels should be achieved pre-operatively and maintained post-surgery, with dosing frequency based on factor half-life (typically 8-12 hours).
Von Willebrand disease treatment options include desmopressin (DDAVP) and cryoprecipitate. DDAVP enhances vWF release in Type 1 VWD. It is a synthetic vasopressin analog that stimulates vWF release from endothelial cells. Intravenous DDAVP (0.3mcg/kg) can increase vWF and factor VIII levels by 3 to 5 times baseline within an hour. DDAVP can also be given subcutaneously or intranasally for home prophylaxis. However, DDAVP can cause fluid retention and hyponatremia, requiring fluid restriction during treatment. DDAVP response varies; about 10% of Type 1 VWD patients may not respond. Patients with Type 2 and 3 VWD typically require factor VIII and vWF replacement products. Notably, DDAVP is contraindicated in Type 2B VWD due to thrombotic risks.
Replacement products for bleeding disorders include factor concentrates and plasma-derived products. Factor VIII replacement dosing using human plasma-derived concentrate is calculated as: weight (kg) x desired factor VIII level x 0.5. Alphanate, a virally inactivated product with factor VIII and vWF activity, is approved for hemophilia A and VWD. Allogenic replacement therapies like fresh frozen plasma (FFP) and cryoprecipitate are less preferred due to large volume requirements (FFP) and lack of factor IX (cryoprecipitate). Platelet transfusions may be necessary for persistent bleeding despite factor replacement.
Antifibrinolytic agents like aminocaproic acid and tranexamic acid are adjunct therapies. Aminocaproic acid inhibits fibrinolysis, promoting clot stability, and is available in IV and oral formulations. Tranexamic acid, a more potent antifibrinolytic, is also available in IV and oral forms, useful for minor bleeding and dental procedures. Hormonal treatments, such as oral contraceptives, can elevate vWF levels in women with VWD and menorrhagia.
Differential Diagnosis and Prognosis
When considering bleeding disorders diagnosis, it’s crucial to differentiate them from other conditions causing bleeding. The differential diagnosis includes acquired conditions such as malignancy, severe liver failure, thrombocytopenia, vitamin K deficiency, antithrombin III deficiency, disseminated intravascular coagulation (DIC), lupus anticoagulant, protein C and S deficiencies, and medication side effects. Idiopathic thrombocytopenic purpura (ITP) should also be considered.
The prognosis for patients with hereditary bleeding disorders is generally good with appropriate medical care, education, and management plans. Patients can live full, productive lives. Medical identification bracelets are recommended to ensure proper management in emergencies.
Enhancing Healthcare Team Outcomes
Effective management of bleeding disorders requires a collaborative interprofessional healthcare team including physicians, specialists, nurses, and pharmacists. The World Bleeding Disorder Registry (WBDR) is a global initiative to collect patient data, improving understanding and treatment of these conditions. Hematology professionals should be familiar with the WBDR to enhance patient care.
Conclusion
Accurate and timely bleeding disorders diagnosis is crucial for effective management of hereditary bleeding disorders. This article has provided a comprehensive overview of the etiology, pathophysiology, clinical presentation, evaluation, and treatment of these conditions, emphasizing the importance of a multidisciplinary approach to patient care. Understanding these complex disorders is essential for healthcare professionals to improve outcomes and quality of life for affected individuals.
References
1.Méndez Rojano R, Mendez S, Lucor D, Ranc A, Giansily-Blaizot M, Schved JF, Nicoud F. Kinetics of the coagulation cascade including the contact activation system: sensitivity analysis and model reduction. Biomech Model Mechanobiol. 2019 Aug;18(4):1139-1153. [PubMed: 30900051]
2.Sultan Y, Caen J, Bernard J. [Demonstration of recessive transmission in Willebrands diseases. Detection of heterozygotes]. C R Acad Hebd Seances Acad Sci D. 1974 Sep 23;279(13):1139-42. [PubMed: 4219324]
3.Edlund M. [Menorrhagia–a symptom not sufficiently surveyed. The path to diagnosis and treatment lined with ambiguity and misunderstandings]. Lakartidningen. 2001 Nov 28;98(48):5505-6, 5509-10. [PubMed: 11769366]
4.Kurth AA, Ludwig G, Scharrer I. [Prevalence, pathophysiology, diagnosis and treatment of von Willebrand syndrome in orthopedic trauma patients]. Orthopade. 1999 Apr;28(4):366-74. [PubMed: 10335531]
5.Bertamino M, Riccardi F, Banov L, Svahn J, Molinari AC. Hemophilia Care in the Pediatric Age. J Clin Med. 2017 May 19;6(5) [PMC free article: PMC5447945] [PubMed: 28534860]
6.Sholzberg M, Teitel J, Hicks LK. A 24-year-old woman with heavy menstrual bleeding. CMAJ. 2017 Jun 05;189(22):E779-E780. [PMC free article: PMC5461127] [PubMed: 28584042]
7.Moon LM, Perez-Milicua G, Dietrich JE. Evaluation and management of heavy menstrual bleeding in adolescents. Curr Opin Obstet Gynecol. 2017 Oct;29(5):328-336. [PubMed: 28858896]
8.Karaman K, Akbayram S, Garipardıç M, Öner AF. Diagnostic evaluation of our patients with hemophilia A: 17-year experience. Turk Pediatri Ars. 2015 Jun;50(2):96-101. [PMC free article: PMC4523992] [PubMed: 26265893]
9.Yoo MC, Jeong BO, Ahn J. Anterior osteophyte resection of the ankle joint to increase range of motion in haemophilic ankle arthropathy. Haemophilia. 2019 May;25(3):e159-e164. [PubMed: 30866148]
10.Demers C, Derzko C, David M, Douglas J. No. 163-Gynaecological and Obstetric Management of Women With Inherited Bleeding Disorders. J Obstet Gynaecol Can. 2018 Feb;40(2):e91-e103. [PubMed: 29447730]
11.Neutze D, Roque J. Clinical Evaluation of Bleeding and Bruising in Primary Care. Am Fam Physician. 2016 Feb 15;93(4):279-86. [PubMed: 26926815]
12.Jain S, Acharya SS. Management of rare coagulation disorders in 2018. Transfus Apher Sci. 2018 Dec;57(6):705-712. [PubMed: 30392819]
13.Saif MA, Thachil J, Brown R, Bigger BW, Wynn RF, Nash M, Hay CR. Is it congenital or acquired von Willebrands disease? Haemophilia. 2015 Jan;21(1):e113-5. [PubMed: 25381916]
14.Anzengruber J, Lubich C, Prenninger T, Gringeri A, Scheiflinger F, Reipert BM, Malisauskas M. Comparative analysis of marketed factor VIII products: recombinant products are not alike vis-a-vis soluble protein aggregates and subvisible particles. J Thromb Haemost. 2018 Jun;16(6):1176-1181. [PubMed: 29665242]
15.Guddati AK, Rosovsky RP, Van Cott EM, Kuter DJ. Quantitative analysis of desmopressin (DDAVP) response in adult patients with type 1 von Willebrand disease. Int J Lab Hematol. 2019 Jun;41(3):325-330. [PubMed: 30735311]
16.Barinsky GL, Buziashvili D, Svider PF, Carron MA, Folbe AJ, Hsueh WD, Eloy JA, Johnson AP. Perioperative Desmopressin for Patients Undergoing Otolaryngologic Procedures: A Systematic Review. Otolaryngol Head Neck Surg. 2019 Jul;161(1):36-45. [PubMed: 30857487]
17.Kruse-Jarres R, Johnsen JM. How I treat type 2B von Willebrand disease. Blood. 2018 Mar 22;131(12):1292-1300. [PubMed: 29378695]
18.Miesbach W, Krekeler S, Wolf Z, Seifried E. Clinical use of Haemate® P in von Willebrand disease: a 25-year retrospective observational study. Thromb Res. 2015 Mar;135(3):479-84. [PubMed: 25595881]
19.Ockelford PA, Lowe G, Johns AS, Berry EW. Comparison of methods of plasma volume determination for dose calculation of factor VIII in patients with classic haemophilia. N Z Med J. 1986 Feb 26;99(796):116-9. [PubMed: 3081842]
20.Bar-Natan M, Hymes KB. Management of Intraoperative Coagulopathy. Neurosurg Clin N Am. 2018 Oct;29(4):557-565. [PubMed: 30223968]
21.Engelen ET, Schutgens RE, Mauser-Bunschoten EP, van Es RJ, van Galen KP. Antifibrinolytic therapy for preventing oral bleeding in people on anticoagulants undergoing minor oral surgery or dental extractions. Cochrane Database Syst Rev. 2018 Jul 02;7(7):CD012293. [PMC free article: PMC6513563] [PubMed: 29963686]
22.Asensio JA, Cáceres ARR, Pelegrina LT, Sanhueza MLÁ, Scotti L, Parborell F, Laconi MR. Allopregnanolone alters follicular and luteal dynamics during the estrous cycle. Reprod Biol Endocrinol. 2018 Apr 10;16(1):35. [PMC free article: PMC5894215] [PubMed: 29636114]
23.Hamilton A, Ozelo M, Leggo J, Notley C, Brown H, Frontroth JP, Angelillo-Scherrer A, Baghaei F, Enayat SM, Favaloro E, Lillicrap D, Othman M. Frequency of platelet type versus type 2B von Willebrand disease. An international registry-based study. Thromb Haemost. 2011 Mar;105(3):501-8. [PubMed: 21301777]