Introduction
Louse-borne relapsing fever (LBRF), a disease with a history stretching back to antiquity, is caused by the spirochete Borrelia recurrentis. Historically associated with war, famine, and displacement, LBRF is experiencing a re-emergence, particularly in Europe, due to increased refugee migration from endemic regions in Africa. This resurgence highlights the critical need for accurate and timely Borrelia recurrentis diagnosis to ensure effective patient management and public health control. This article provides a detailed overview of LBRF, with a specific focus on the diagnostic approaches for Borrelia recurrentis infection.
Aetiology
Borrelia recurrentis is the causative agent of LBRF. This bacterium is a large, motile spirochete closely related genetically to Borrelia duttonii and Borrelia crocidurae, the agents of tick-borne relapsing fever in East and West Africa, respectively. In fact, due to their genomic similarity, these three Borrelia species are sometimes considered ecotypes of a single genomospecies. Borrelia recurrentis has a relatively simple genome and can be cultured in specialized media, aiding in research and potentially advanced diagnostic development.
Transmission
The primary vector for Borrelia recurrentis is the human body louse (Pediculus humanus corporis). While Borrelia recurrentis has been found in head lice, transmission via this vector is not confirmed. Transmission occurs when infected lice are crushed, and their coelomic fluid or feces are introduced into the bloodstream through scratching or via mucous membranes. Nosocomial infections are also possible through contaminated blood products or needlestick injuries. Unlike ticks, lice do not transmit Borrelia recurrentis to their offspring, meaning humans are the primary reservoir for the bacteria between epidemics.
Epidemiology and Historical Background
LBRF is classically linked to conditions of poverty, poor hygiene, overcrowding, and displacement, which favor body louse infestation. Historically, LBRF has been responsible for significant epidemics across Europe, North Africa, and the Middle East. Descriptions of relapsing fevers, possibly LBRF, can be found as far back as Hippocrates. Major pandemics occurred in the 20th century, causing millions of cases. Currently, LBRF is endemic in the Horn of Africa, with ongoing outbreaks. The recent influx of refugees from these regions into Europe has led to a re-emergence of LBRF in countries where it was once eradicated, posing diagnostic challenges for clinicians unfamiliar with the disease.
Pathophysiology and Pathology
The Relapse Phenomenon
The hallmark of LBRF is its relapsing nature. This is driven by antigenic variation of the Borrelia recurrentis variable major outer membrane lipoprotein (vmp). The initial fever episode resolves as the immune system produces antibodies against the dominant vmp. However, Borrelia recurrentis can evade this response by switching to express a different vmp, leading to a relapse of spirochetemia and fever. This cycle can repeat multiple times in untreated individuals. The release of TNF-α during the immune response and the Jarisch–Herxheimer reaction (J-HR) following antibiotic treatment is also linked to vmp. Borrelia recurrentis also employs mechanisms to evade the complement system, further contributing to its persistence in the host.
Pathophysiology
The symptoms of LBRF, and particularly the J-HR, are associated with a systemic inflammatory response. Borrelia recurrentis vmps stimulate the release of pro-inflammatory cytokines such as TNF-α, IL-6, IL-8, and IL-1β. This cytokine storm is triggered by the phagocytosis of spirochetes, particularly during antibiotic treatment. The clinical manifestations, including fever, disseminated intravascular coagulation, and organ dysfunction, are consequences of this inflammatory cascade.
Pathology
Pathological findings in LBRF include the presence of spirochetes in blood vessels and tissues, especially in the spleen, liver, and central nervous system. Myocarditis, characterized by perivascular inflammation, is a significant finding and can contribute to sudden death. Other severe pathological features include splenic rupture, cerebral hemorrhage, and hepatic failure. Petechial hemorrhages are common throughout the body.
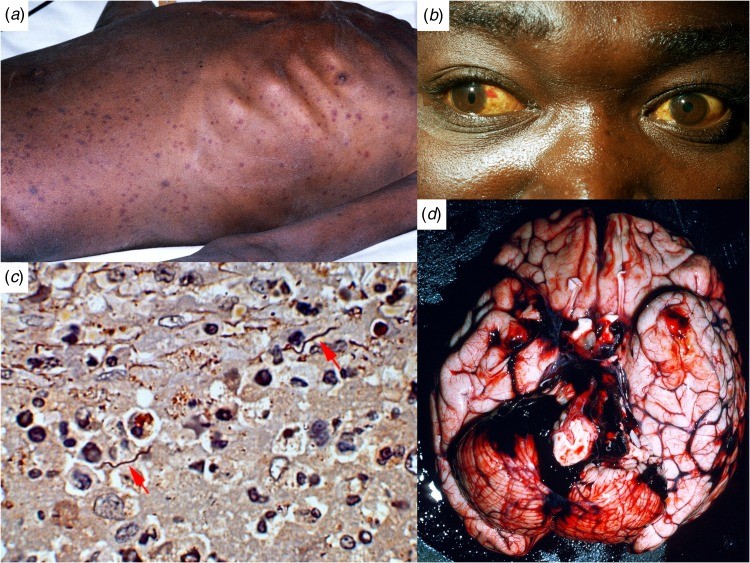
Fig. 1. LBRF Manifestations in Ethiopian Patients
Figure 1. Clinical presentations of LBRF in Ethiopian patients. (A) Petechial rash on the trunk of a patient co-infected with Salmonella enterica serovar Typhi. (B) Subconjunctival hemorrhages and jaundice indicating liver damage and coagulopathy. (C) Borrelia recurrentis spirochetes in splenic pulp (silver stain). (D) Cerebral hemorrhage, a severe complication of LBRF.
Symptoms and Signs
The incubation period for LBRF ranges from 4 to 18 days. The onset of illness is abrupt, with high fever and rigors. Common symptoms include headache, myalgia, arthralgia, anorexia, nausea, vomiting, and abdominal pain. Epistaxis and cough may develop. Hepatic tenderness and hepatomegaly are frequent signs, while splenomegaly is less common. Jaundice and petechial rash are variable but important clinical features. Subconjunctival hemorrhages and epistaxis are also common. Neurological symptoms are less frequent compared to tick-borne relapsing fevers. Untreated attacks typically resolve after several days, followed by afebrile periods and subsequent relapses. Pregnant women are at higher risk of severe disease and adverse pregnancy outcomes.
Clinical Presentation in Refugees Arriving in Europe
Studies comparing LBRF in refugees arriving in Europe to patients in endemic regions show some differences. While fever, headache, and myalgia are common in both groups, jaundice appears to be more frequently reported in refugees. Bleeding and meningism, and fatalities were less frequent in refugees in one study, potentially reflecting earlier diagnosis and treatment in European settings. Elevated C-reactive protein and procalcitonin levels are observed in refugees with LBRF.
Prognosis
Historically, untreated LBRF had a high case fatality rate. However, with antibiotic treatment, the prognosis is significantly improved, with mortality reduced to below 5% in most treated cases. Factors associated with poor prognosis include severe manifestations like coma, shock, hyperpyrexia, myocarditis, hepatic failure, and complicating co-infections.
Severe Louse-Borne Relapsing Fever
Severe LBRF is characterized by complications such as hyperpyrexia, shock, myocarditis leading to pulmonary edema, acute respiratory distress syndrome, hepatic failure, splenic rupture, and hemorrhage. Co-infections can worsen the prognosis.
Spontaneous Crisis and J-HR
Both the spontaneous crisis at the end of a fever paroxysm and the J-HR following antibiotic treatment involve a dramatic physiological response. The crisis or J-HR typically begins with rigors, followed by a rapid rise in temperature, heart rate, and blood pressure, potentially accompanied by delirium. This is followed by a “flush phase” with profuse sweating, hypotension, and temperature decline. Hypotension during this phase can be dangerous, particularly in patients with myocarditis. Tetracycline is associated with a higher incidence of J-HR compared to penicillin.
Laboratory Investigations
Laboratory findings in LBRF often include spirochetemia, anemia, and neutrophilic leukocytosis, followed by leukopenia during crisis or J-HR. Thrombocytopenia and mild coagulopathy are common. Elevated liver enzymes and bilirubin indicate hepatocellular damage. Mild renal impairment can occur. ECG changes may reflect myocarditis.
Diagnosis
Accurate and rapid Borrelia recurrentis diagnosis is crucial for effective management of LBRF. Several diagnostic methods are available:
Microscopy
Microscopy of blood films remains the most rapid and readily available method for Borrelia recurrentis diagnosis, particularly in endemic settings. Both thick and thin blood smears, stained with Giemsa or similar stains, can be used to visualize the spirochetes. Thick smears are more sensitive due to the larger volume of blood examined. The sensitivity of microscopy depends on the spirochete density in the blood, with thin smears detecting around 105 spirochetes/mL and thick smears around 104 spirochetes/mL. Concentration methods, like centrifugation, can improve sensitivity. Quantitative buffy coat technique is another microscopic method. The high spirochetemia in LBRF generally makes microscopy more reliable compared to other Borrelia infections. A potential diagnostic pitfall is misidentification of Plasmodium vivax microgametes as spirochetes.
Polymerase Chain Reaction (PCR)
Polymerase Chain Reaction (PCR) assays have significantly advanced Borrelia recurrentis diagnosis. Multiplex real-time PCR (MR-TPCR) assays can differentiate between African Borrelia species, including B. recurrentis, B. duttonii, B. crocidurae, and B. hispanica. These assays typically target genes like 16S rRNA, glpQ, recN, and recC. PCR offers higher sensitivity than microscopy, detecting as few as 100 copies of bacterial DNA, and can identify Borrelia recurrentis even in microscopy-negative cases. While highly sensitive for B. duttonii/B. recurrentis, current PCR assays may not reliably distinguish between these two closely related species. PCR is particularly valuable for confirming Borrelia recurrentis diagnosis in returning travelers and refugees and is being implemented in point-of-care settings in endemic regions.
Serology
Serological tests for LBRF have historically been unreliable due to low specificity and sensitivity. While some assays using recombinant antigens like glpQ or monoclonal antibodies have shown promise in research settings, serology is not routinely used for Borrelia recurrentis diagnosis. Serological tests may produce false-positive results for syphilis and are often not sensitive enough to detect acute infections. Currently, serology plays a limited role in the diagnostic algorithm for LBRF.
Differential Diagnosis
In febrile patients, particularly those from or with travel history to endemic areas, Borrelia recurrentis diagnosis requires careful consideration of differential diagnoses. Severe falciparum malaria is a critical differential, especially given overlapping geographical distribution and similar symptoms like fever, jaundice, and thrombocytopenia. Other considerations include:
- Yellow fever and other viral hemorrhagic fevers: Important in endemic areas, presenting with jaundice and bleeding.
- Viral hepatitis: Can cause jaundice and fever.
- Rickettsial infections, especially louse-borne typhus: Louse-borne typhus can occur in mixed epidemics with LBRF and share symptoms like fever and rash.
- Leptospirosis: Considered if acute kidney injury is present, can co-occur with LBRF.
- Trench fever (Bartonella quintana): Louse-borne, causes relapsing fever but typically lacks jaundice and severe bleeding.
- Typhoid fever: Common co-infection in endemic areas, presenting with fever and abdominal symptoms.
- Sepsis and meningitis: Important to rule out in febrile patients, especially in refugee populations.
Treatment
Antibiotic treatment is highly effective for LBRF. Single-dose oral tetracycline or erythromycin can achieve complete cure and prevent relapses. However, due to frequent vomiting, parenteral administration, such as single-dose intravenous tetracycline or erythromycin (for pregnant women and children), is often preferred. Doxycycline is effective in areas with potential co-endemic louse-borne typhus. Penicillin is also effective but may be less reliable in preventing relapses, particularly with long-acting formulations. Lower doses of penicillin may be used initially in severe cases or pregnant women to mitigate the J-HR. Chloramphenicol is another effective option but may have limited availability in some regions.
Preventing the J-HR
The Jarisch–Herxheimer reaction is a common complication of antibiotic treatment for LBRF. While various strategies have been investigated to prevent or mitigate the J-HR, none have proven consistently effective in clinical practice. Pre-treatment with corticosteroids, hydrocortisone, paracetamol, and pentoxifylline have not shown reliable benefit. Meptazinol and anti-TNF-α antibodies have shown some promise in research settings but are not widely available. Supportive care remains crucial during the J-HR, including managing hyperpyrexia, hypotension, and dehydration.
Supportive Treatment
Supportive care is essential, particularly during the spontaneous crisis or J-HR. This includes aggressive management of hyperpyrexia with antipyretics and cooling measures, fluid resuscitation to address dehydration and hypovolemia, and monitoring for and managing hypotension. In cases of myocarditis, digoxin may be considered. Oxygen therapy is indicated in severe cases. Complicating infections should be promptly identified and treated.
Prevention and Control of Epidemics
Prevention of LBRF epidemics relies on breaking the cycle of louse transmission. This involves:
- Delousing clothing: Heat treatment (>60 °C) or washing at 52 °C for 30 minutes effectively kills lice.
- Personal hygiene: Regular bathing with soap.
- Pediculicides: Topical pediculicides can be used, but louse resistance is a growing concern.
- Separation of infested clothing: Removing clothing from the wearer for 10 days will starve lice to death.
Vaccine development for LBRF has not been prioritized.
Conclusion
Borrelia recurrentis diagnosis is critical for managing louse-borne relapsing fever, a re-emerging infectious disease of global health concern. Microscopy remains a rapid and accessible diagnostic tool, while PCR offers improved sensitivity and species-specific identification. Clinicians in both endemic and non-endemic regions, particularly in areas receiving refugee populations, should be aware of LBRF and its diagnostic approaches to ensure prompt and effective patient care and contribute to public health efforts to control this ancient yet persistent disease.
References
[References] (as in original article)