Chronic Obstructive Pulmonary Disease (COPD) is a prevalent condition characterized by persistent respiratory symptoms and airflow limitation. Patients with COPD frequently experience acute exacerbations marked by worsening dyspnea, increased cough, and sputum production. However, in the acute care setting, it’s crucial to recognize that not all respiratory distress in COPD patients stems directly from COPD exacerbations. A range of cardiopulmonary conditions can mimic or aggravate COPD symptoms, leading to diagnostic complexities. This article provides a comprehensive overview of common cardiopulmonary diagnoses that should be considered in the differential diagnosis of acute respiratory distress in COPD patients, emphasizing a systematic approach to enhance diagnostic accuracy and improve patient outcomes.
Navigating the Complexities of Respiratory Distress in COPD Patients
The clinical course of COPD is often punctuated by acute exacerbations of COPD (ECOPDs), characterized by a sudden worsening of respiratory symptoms. These episodes not only diminish the patient’s quality of life but also contribute to a faster decline in lung function and elevate the risk of hospitalization and mortality. Alarmingly, a significant proportion of patients, nearly 20%, are readmitted to the hospital within 30 days of discharge following an ECOPD, imposing a substantial burden on both individuals and healthcare systems. The prognosis for patients hospitalized for ECOPD is grim, with approximately 25% and 65% mortality rates within 1 and 5 years, respectively.
COPD patients are typically older and often present with multiple comorbidities, including heart failure (HF), coronary artery disease (CAD), arrhythmias, interstitial lung diseases (ILDs), and bronchiectasis. These co-existing conditions not only increase the likelihood of ECOPDs but can also closely resemble or worsen their presentation. Notably, a substantial proportion of readmissions after ECOPD hospitalization, around 70%, are attributed to the decompensation of these underlying comorbidities. This highlights the critical need for clinicians to broaden their diagnostic perspective when evaluating COPD patients presenting with worsening dyspnea, especially when the typical features of ECOPD are not prominent. A meticulous investigation into these potential comorbidities can facilitate accurate diagnosis and enable timely and appropriate treatment.
For the majority of these mimicking or aggravating conditions, a comprehensive clinical assessment, supplemented by judicious use of clinical investigations, can guide healthcare providers towards a precise diagnosis. This article consolidates currently fragmented information and offers a practical, systematic approach for clinicians managing COPD patients who present with worsening respiratory symptoms, particularly dyspnea, in the acute care setting. Adopting such a systematic strategy is crucial for improving patient outcomes and mitigating the personal and societal costs associated with ECOPDs.
Methodological Approach to Differential Diagnosis
This perspective is based on the collective expertise of a panel of international experts in respiratory medicine, building upon previous work defining and classifying ECOPD severity. To ensure a robust and clinically relevant approach, a modified Delphi process was employed to identify and refine the most pertinent differential diagnoses. This involved a rigorous review of existing literature, with a focus on clinical guidelines and expert consensus documents. From a wide array of potential conditions that can manifest with respiratory symptoms similar to ECOPD, twelve key morbidities were identified as being most critical for consideration in clinical practice.
Working groups of experts conducted focused literature reviews for each of these identified morbidities. The culmination of this process was the development of a comprehensive diagnostic algorithm, along with detailed sub-algorithms for the most frequently encountered and clinically challenging conditions. These algorithms are designed to serve as practical tools for healthcare providers in the acute care setting. The algorithms and the accompanying guidance were iteratively refined through discussions and consensus-building among all expert panel members. For the purpose of this document, patients are categorized as having “confirmed COPD” if they have spirometric evidence of airflow limitation, and “suspected COPD” if spirometry is lacking but they have a history of smoking or environmental pollutant exposure, chronic respiratory symptoms, and/or are receiving COPD-directed pharmacological treatment.
Initial Patient Assessment in Acute Respiratory Distress
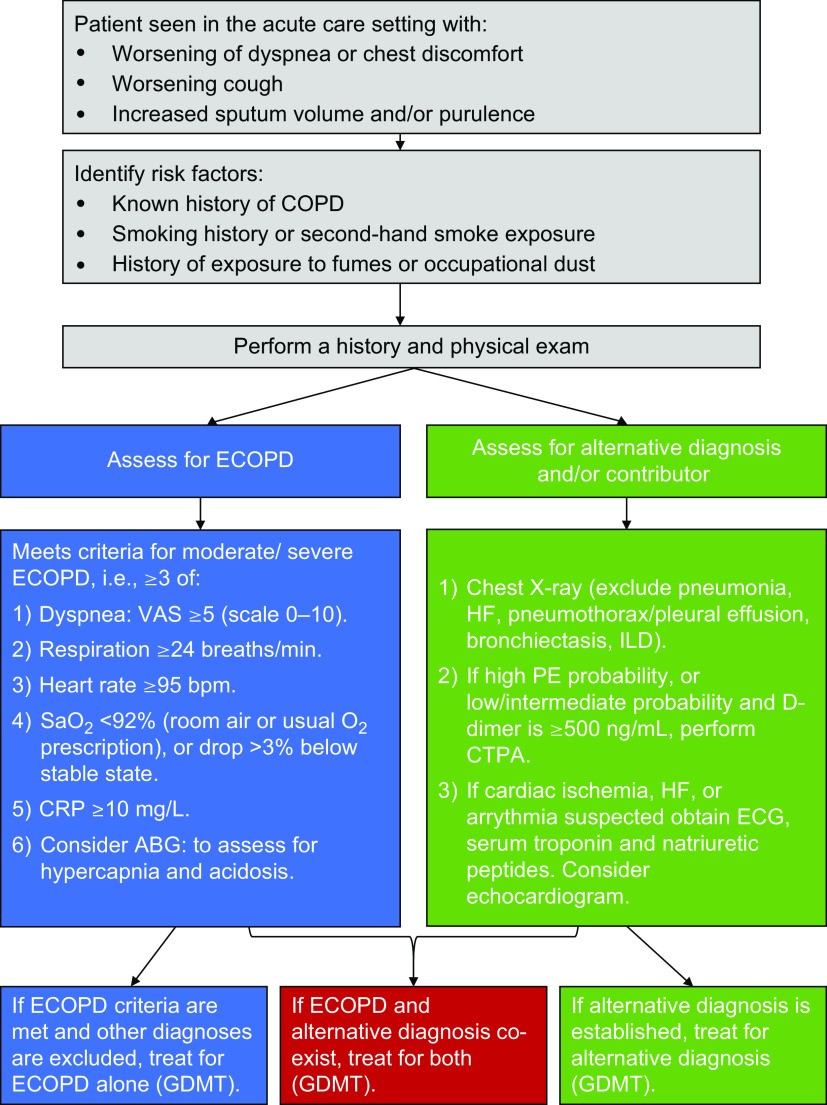
An initial assessment of a COPD patient presenting with worsening respiratory symptoms in the acute care setting should encompass a detailed medical history and thorough physical examination. This clinical evaluation, combined with the judicious use of objective measurements, can often establish ECOPD as the most probable diagnosis, particularly in milder cases seen in outpatient settings. However, it’s essential to recognize that the signs and symptoms of ECOPD, especially in more severe presentations, are not always specific. Other diseases can mimic or exacerbate ECOPD, complicating the clinical picture. This is especially pertinent in the acute care environment where patients often present with more severe and complex conditions. To aid in navigating this diagnostic complexity, a practical initial algorithm (Figure 2 from the original article) has been developed to guide clinicians toward a more precise diagnosis.
Table 1: The CASE Approach to Evaluating Worsening Respiratory Symptoms in COPD
| Category | Description |
|---|---|
| Complete | Conduct a thorough clinical assessment to identify underlying COPD and potential respiratory and non-respiratory comorbidities. Consider alternative causes for symptoms. |
| Assess | Symptoms: Evaluate dyspnea severity using a visual analog scale; document cough and sputum characteristics, presence and nature of pain. |
| Signs: Assess for tachypnea, tachycardia, arrhythmia, fever, sputum volume and color, respiratory distress signs (accessory muscle use), and thoracic abnormalities. | |
| Severity | Combine symptom and sign assessment with investigations like pulse oximetry. Consider CRP, chest X-ray, ECG, and arterial blood gases for severe cases. |
| Establish | Determine the likely cause of the event: viral, bacterial, environmental, medication non-adherence, alternative diagnosis, or other contributing factors. |
Definition of abbreviations: COPD = chronic obstructive pulmonary disease; CRP = C-reactive protein.
This CASE approach (Complete, Assess, Severity, Establish) provides a structured framework for the initial evaluation, ensuring a comprehensive and systematic assessment of patients presenting with acute respiratory symptoms.
Figure 2. Diagnostic algorithm for evaluating COPD patients with symptoms consistent with an exacerbated COPD in acute care settings. This algorithm emphasizes a step-wise approach incorporating clinical assessment, objective measurements, and targeted investigations to differentiate ECOPD from mimicking conditions.
Cardiovascular Conditions Mimicking COPD Exacerbations
Cardiovascular diseases are frequently observed in patients with COPD, and their presence significantly influences the clinical presentation of acute respiratory symptoms. Therefore, obtaining a detailed patient history focused on cardiovascular risk factors and prior cardiac diagnoses is paramount in assessing patients presenting with suspected ECOPD. Symptoms such as chest pain or discomfort, fluid retention (peripheral edema), and irregular pulse should raise suspicion for a cardiovascular etiology, either as an alternative diagnosis or a contributing factor to respiratory distress.
Acute Heart Failure (HF)
Acute heart failure is characterized by the rapid onset or worsening of dyspnea, particularly at rest or when lying down (orthopnea), accompanied by signs of fluid overload. These signs can include pulmonary congestion (rales or crackles on auscultation), elevated jugular venous pressure, and peripheral edema. Objective evidence of structural or functional cardiac abnormalities is essential for confirming the diagnosis. The symptomatic overlap between acute HF and ECOPD can make differentiation challenging. Compounding this difficulty is the frequent co-occurrence of COPD and HF, driven by shared risk factors like smoking and aging, as well as common underlying pathophysiological mechanisms such as systemic inflammation. Furthermore, diagnosing HF with preserved ejection fraction (HFpEF), which is common in COPD patients, can be particularly complex.
The interplay between COPD and HF is bidirectional. Hypoxemia and tachycardia during an ECOPD can decompensate pre-existing HF, while pulmonary edema in HF can exacerbate airflow obstruction in COPD. Wheezing, often associated with airway narrowing in COPD, is also present in approximately 30% of acute HF cases, further blurring the clinical distinction. Conversely, COPD can mask the classic basilar crackles of HF. However, the presence of basilar crackles, particularly when coupled with peripheral edema, elevated jugular venous pressure, and a third heart sound (S3 gallop), significantly increases the likelihood of HF contributing to the acute event.
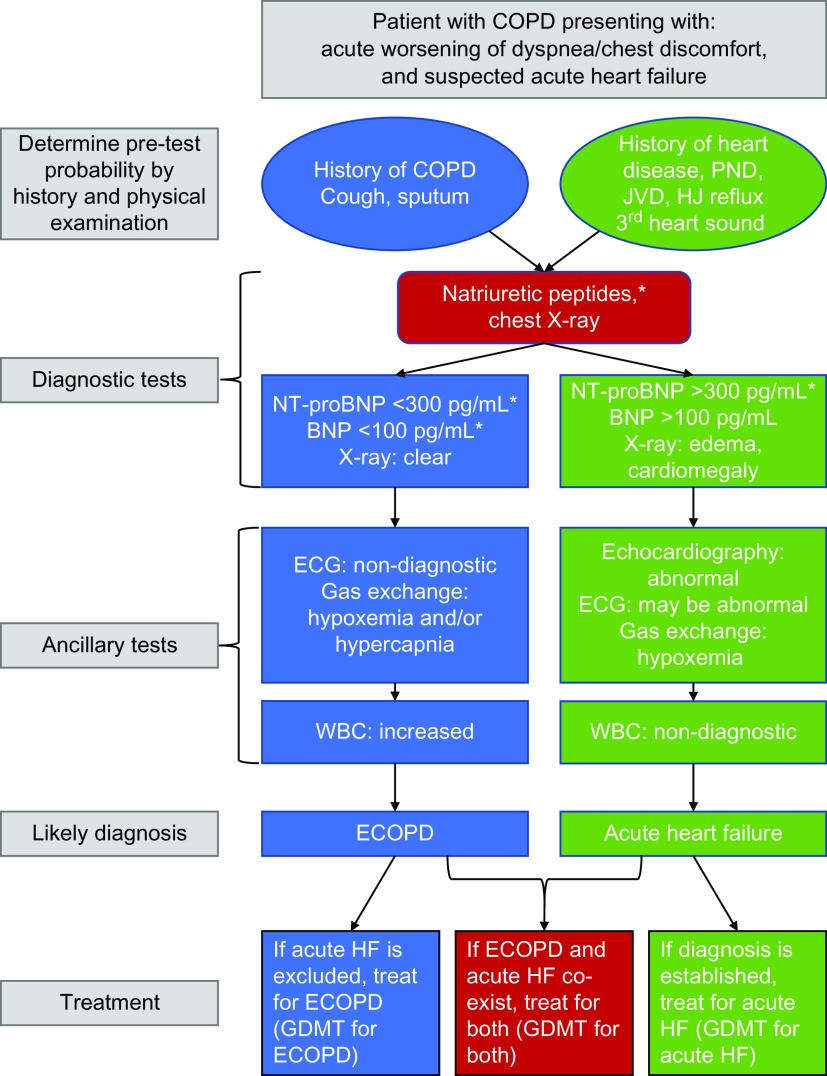
Chest radiography is a valuable tool in differentiating ECOPD from acute HF. Cardiomegaly supports underlying structural heart disease, and pulmonary edema on chest X-ray strongly suggests HF. Elevated levels of natriuretic peptides, such as brain natriuretic peptide (BNP) and N-terminal pro-brain natriuretic peptide (NT-proBNP), are robust biomarkers for HF. However, it’s crucial to interpret natriuretic peptide levels cautiously in COPD patients, as they can be elevated in stable COPD, pneumonia, and ECOPD even in the absence of acute HF. Therefore, higher diagnostic thresholds are often required to accurately diagnose concomitant acute HF and ECOPD. Importantly, low natriuretic peptide levels have a high negative predictive value for acute HF, even in the context of ECOPD. Elevated natriuretic peptides during ECOPD are associated with a poorer prognosis, including increased mortality risk. Point-of-care ultrasonography is emerging as a promising tool to differentiate between ECOPD and acute HF, but further research is needed to fully establish its impact on clinical outcomes. A diagnostic algorithm for evaluating suspected acute HF in COPD patients is provided in Figure 3 of the original article, offering a structured approach to diagnosis.
Figure 3. Diagnostic algorithm for evaluating COPD patients presenting with increased respiratory symptoms and suspected acute heart failure in acute care settings. This algorithm integrates clinical features, natriuretic peptide levels, and ECG findings to guide the diagnostic process.
Ischemic Heart Disease (IHD)
Underlying ischemic heart disease (IHD) is associated with more prolonged and complicated ECOPDs. Myocardial injury is a frequent and clinically significant complication during ECOPDs, particularly in patients with pre-existing IHD. ECOPD significantly elevates the risk of acute myocardial infarction and stroke, not only in the immediate 30 days following the exacerbation but for up to a year afterward. Furthermore, COPD and IHD frequently coexist, with approximately one-third of IHD patients also having COPD. This co-occurrence is linked to more frequent emergency room visits and increased morbidity and mortality, with COPD independently contributing to IHD risk.
In outpatient settings, a clinical history of established IHD, current symptoms such as chest pain or discomfort and palpitations, and signs of systemic atherosclerosis (carotid bruit, reduced peripheral pulses) or acute myocardial ischemia (diaphoresis, S3 gallop) should raise suspicion for concomitant IHD in patients presenting with suspected ECOPD. Symptoms combined with an abnormal resting electrocardiogram (ECG) further strengthen the suspicion of acute myocardial ischemia. The diagnosis of myocardial injury or infarction, whether independent of or contributing to an ECOPD, should adhere to the fourth universal definition criteria, requiring elevated blood troponin levels at admission and/or during follow-up, along with at least one other criterion (ischemic symptoms, new ECG changes). It’s important to note that COPD itself can be associated with chronic, stable elevations in troponin levels, which may further increase during an ECOPD. When clinical suspicion for IHD is high, guideline-directed evaluation is warranted. Given the high prevalence of IHD in COPD patients experiencing exacerbations, assessment for atherosclerosis and related cardiovascular conditions should always be considered.
Arrhythmias
Cardiac arrhythmias, particularly atrial fibrillation (AF), are more prevalent in COPD patients. The risk of AF and other arrhythmias doubles during an ECOPD, and importantly, AF occurring during an ECOPD is associated with increased morbidity and mortality. Symptoms suggestive of AF include atypical chest discomfort, palpitations, lightheadedness, and dyspnea, along with an irregular pulse on palpation. Unlike some other conditions, the diagnostic differentiation between AF and ECOPD is often less challenging. Standard ECG and cardiac monitoring can effectively detect chronic, acute, or intermittent arrhythmias. The role of new-onset AF as a direct cause of acute dyspnea in COPD patients is still under investigation, and it remains a clinical question whether detected AF is pre-existing or a new event triggered by the acute respiratory decompensation.
Pulmonary Embolism (PE)
The prevalence of pulmonary embolism (PE) in patients presenting with ECOPD varies widely, ranging from 3.3% to as high as 29.4% in some studies. PE is more likely in patients with atypical or unexplained ECOPD and necessitates prompt diagnosis and specific management to improve outcomes. PE should be considered in COPD patients presenting with worsening dyspnea, especially in the presence of risk factors such as a history of venous thromboembolic disease, recent hospitalization for heart failure, or malignancy. While both PE and ECOPD share the primary symptom of worsened dyspnea, patients with PE more frequently report pleuritic chest pain, hemoptysis (coughing up blood), and signs of right heart failure. They are also less likely to present with a cough producing increased or purulent sputum, which is more typical of ECOPD.
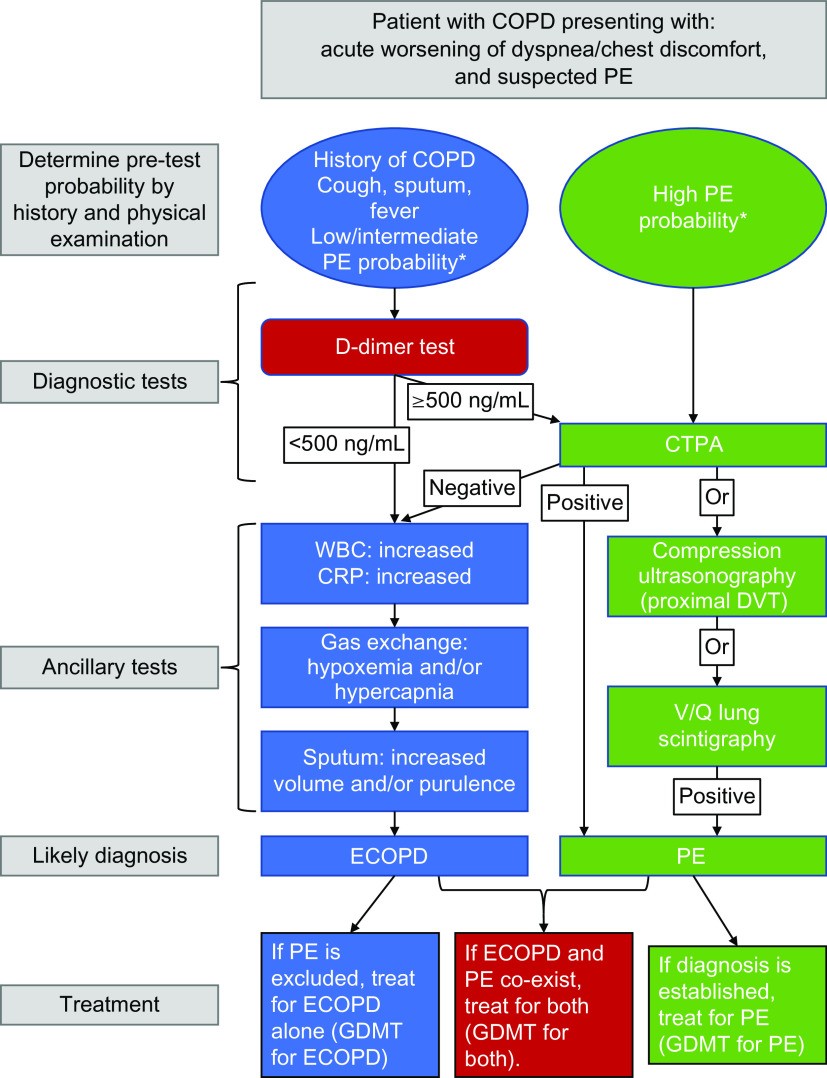
Practical diagnostic approaches for evaluating PE in hospitalized ECOPD patients have been investigated. Risk stratification scores, such as the revised Geneva score, can be used to assess the pretest clinical probability of PE. Patients with a high pretest probability should proceed directly to computed tomography pulmonary angiography (CTPA). In those with a low pretest probability, D-dimer testing can be used as an initial screening tool. A negative D-dimer result (below 500 ng/mL) can effectively rule out PE, obviating the need for further imaging. Conversely, a positive D-dimer result warrants further investigation, typically with CTPA. Studies have demonstrated that using such risk-stratified approaches can effectively identify PE while minimizing unnecessary imaging. Given the high mortality associated with PE, CTPA imaging is strongly recommended in patients with a high clinical suspicion of PE based on pretest probability scores and in those with atypical ECOPD presentations. In patients with a low pretest probability, a negative D-dimer can help exclude PE and avoid unnecessary CTPA. Lower-limb compression ultrasonography (CUS) can be a valuable adjunct, particularly to diagnose deep vein thrombosis (DVT) in patients with suspected PE, especially when CTPA is contraindicated or negative. In the context of suspected PE, a focused four-point CUS examination of the groin and popliteal fossa can be sufficient. The probability of detecting proximal DVT with CUS is higher in patients with leg symptoms suggesting DVT. A diagnostic algorithm for evaluating suspected PE in COPD patients is presented in Figure 4 from the original article, providing a step-by-step guide to diagnosis.
Figure 4. Diagnostic algorithm for evaluating COPD patients presenting with increased respiratory symptoms and suspected pulmonary embolism (PE) in acute care settings. This algorithm incorporates clinical probability assessment, D-dimer testing, CTPA, and lower limb ultrasound to guide the diagnostic pathway for PE.
Infectious Diseases in the Differential Diagnosis
Pneumonia
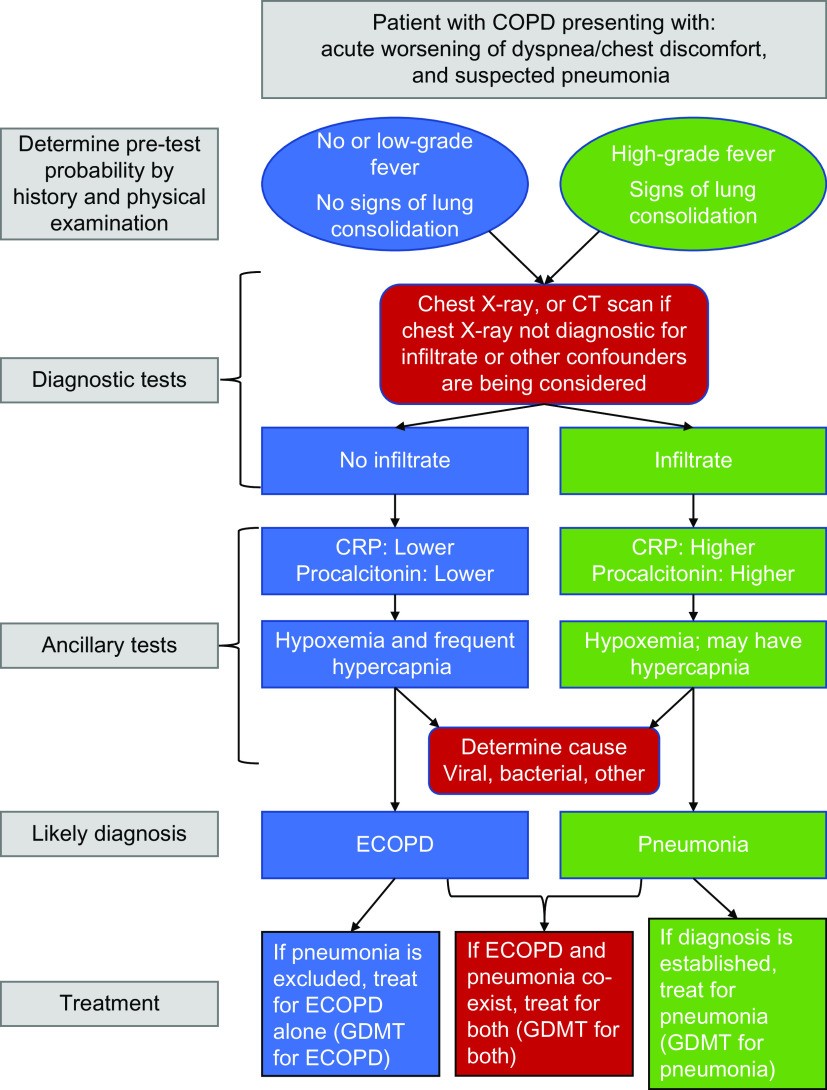
Distinguishing between pneumonia and ECOPD can be challenging, as both conditions share overlapping symptoms such as worsened cough, increased sputum production, dyspnea, and fever. However, pneumonia should always be considered in the differential diagnosis of COPD patients presenting with symptoms suggestive of ECOPD, as COPD is a significant risk factor for pneumonia. Physical examination can provide clues for differentiation. Signs of lung consolidation, such as localized crackles, crepitus, and dullness to percussion, are more frequently observed in pneumonia.
In cases where the suspected ECOPD is deemed mild to moderate and there are no clinical signs of lung consolidation, further diagnostic testing to differentiate pneumonia from ECOPD may not be immediately necessary. However, if the episode is severe or prolonged, or if abnormal chest examination findings are present, a thorough evaluation for pneumonia is warranted. Mortality rates following hospitalization for pneumonia are higher than those for ECOPD, emphasizing the importance of accurate diagnosis. While chest X-ray is often the initial imaging modality, it may miss subtle opacities. In such cases, more sensitive CT scans can be considered, especially in severe or diagnostically uncertain cases.
Serum biomarkers are increasingly valuable in differentiating pneumonia from ECOPD, particularly in the acute care setting. Elevated C-reactive protein (CRP) levels are more suggestive of pneumonia, especially bacterial pneumonia, compared to ECOPD. Procalcitonin can also be helpful; a low procalcitonin level in a patient with low pretest probability for pneumonia may favor a non-bacterial etiology. Other biomarkers like interleukin-6 (IL-6), white blood cell count, and platelet count may also have diagnostic utility, although validated cut-off values are still under investigation.
In the context of ECOPD, clinicians should consider viral respiratory infections, such as respiratory syncytial virus (RSV), severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) (COVID-19), and influenza, as causative agents, particularly during periods of community outbreaks. Specific antiviral treatments are available for these infections, and COPD patients are often prioritized for such interventions due to their increased risk of severe outcomes. Sputum purulence is a clinical marker that supports bacterial infection and can predict the benefit of antibiotic treatment. In severe ECOPD or suspected pneumonia, sputum Gram stain and culture, along with molecular point-of-care testing, can guide antibiotic therapy, especially as difficult-to-treat bacterial pathogens may be present. Urine antigen tests for Streptococcus pneumoniae and Legionella pneumophila should also be considered in suspected pneumonia cases. A diagnostic algorithm for evaluating suspected pneumonia in COPD patients is provided in Figure 5 from the original article, guiding clinicians through the diagnostic process.
Figure 5. Diagnostic algorithm for evaluating COPD patients presenting with increased respiratory symptoms and suspected pneumonia in acute care settings. This algorithm integrates clinical assessment, chest imaging, and biomarker testing to guide the diagnosis of pneumonia in the context of COPD.
Bronchiectasis
Bronchiectasis, characterized by permanent dilatation of the bronchi, is a common comorbidity in COPD patients, with prevalence rates ranging widely depending on the population studied and diagnostic criteria used. Patients with both COPD and bronchiectasis tend to experience more frequent exacerbations, increased respiratory symptoms, a higher risk of pneumonia, and increased mortality compared to those with either condition alone. This increased exacerbation risk may be related to more frequent colonization with pathogenic bacteria, including Pseudomonas aeruginosa.
COPD diagnosis is primarily based on physiological criteria (spirometry), whereas bronchiectasis diagnosis is radiological (CT scan). However, patients with severe bronchiectasis can also exhibit poorly reversible airflow obstruction, meeting spirometric criteria for COPD, leading to diagnostic overlap. The ROSE criteria (Radiology, Airflow Obstruction, Symptoms, Exposure) can be helpful in characterizing COPD/bronchiectasis overlap. In patients with overlapping COPD and bronchiectasis, differentiating between an exacerbation of bronchiectasis and an ECOPD can be clinically challenging. Bronchiectasis guidelines recommend investigating for underlying treatable causes, such as antibody deficiency. CT scans are logically indicated to exclude bronchiectasis in COPD patients with frequent or severe exacerbations.
Inflammatory Conditions to Consider
Asthma Exacerbation
Comorbid bronchial asthma is prevalent in COPD patients, with reported rates varying widely based on diagnostic definitions. Factors suggesting asthma comorbidity in a COPD patient include a family history of asthma, younger age, and a history of atopy or childhood asthma. Patients with both COPD and asthma tend to be more symptomatic, have poorer lung function, and are at a higher risk of exacerbations and hospitalizations compared to patients with either condition alone.
Currently, there is limited research directly comparing exacerbations in patients with comorbid COPD and asthma to exacerbations of asthma alone or ECOPD. Clinical guidelines often define asthma exacerbations and ECOPDs using similar criteria. Viral infections and environmental pollutants are common triggers for both conditions. While sputum purulence is typically associated with ECOPD, it can also be observed in asthma exacerbations, further complicating differentiation. In practice, the acute management of concomitant COPD and asthma often aligns closely with asthma exacerbation management guidelines, focusing on bronchodilation and anti-inflammatory therapies.
Interstitial Lung Diseases (ILDs)
Interstitial lung diseases (ILDs) are found in a notable proportion of individuals with a history of tobacco smoke exposure. Despite differing underlying pathophysiologies and static respiratory mechanics, both ILDs and COPD share dyspnea and dry cough as prominent symptoms. Dyspnea in both conditions is often attributed to a mismatch between the effort required to breathe and the effective volume of air inhaled. Similar to COPD, ILDs, particularly idiopathic pulmonary fibrosis (IPF), can have a course punctuated by acute exacerbations, which are typically severe and associated with clinical decline. IPF exacerbations generally carry a worse prognosis compared to ECOPDs.
Some patients present with combined pulmonary fibrosis and emphysema (CPFE). In these patients, differentiating between an ECOPD and an IPF exacerbation during an acute respiratory worsening can be challenging. Imaging findings of new widespread alveolar abnormalities, such as ground-glass opacities, on chest imaging in a patient with symptoms suggestive of ECOPD should raise suspicion for an ILD exacerbation contributing to the acute event.
Miscellaneous Conditions in Differential Diagnosis
Anxiety and Depression
The prevalence of depression and anxiety in COPD patients experiencing ECOPDs is substantial and varies based on assessment methods. Patients with COPD and comorbid depression and/or anxiety have a higher risk of ECOPDs and hospitalizations compared to those without these mental health conditions. Furthermore, a significant proportion of current and former smokers have co-existing anxiety and depression, which independently predict exacerbation risk. This creates a negative feedback loop, where each exacerbation worsens symptoms, muscle weakness, and functional capacity, consequently exacerbating anxiety and depression.
The direct causal role of anxiety and depression in triggering ECOPDs is not fully understood. Panic attacks and anxiety can induce tachypnea, which in COPD patients can lead to hyperinflation and potentially acute hypercapnic respiratory failure. Conversely, hypercapnia itself can trigger panic attacks through activation of brainstem reflexes, leading to hyperventilation and further panic symptoms. In COPD patients experiencing a panic attack, the absence of increased cough and sputum, along with the lack of inflammatory markers or other clear causes for dyspnea and tachypnea, can help differentiate panic from ECOPD.
Pneumothorax
Pneumothorax, the presence of air in the pleural space, is a potentially life-threatening condition that can occur in COPD patients and mimic an ECOPD. Subpleural blebs and bullae, common in older males with emphysema and smoking history, predispose to pneumothorax. In COPD patients with pneumothorax, dyspnea is often accompanied by chest pain, hypoxemia, and in some cases, acute respiratory failure. Diagnosis is typically confirmed by chest X-ray, carefully differentiating pneumothorax from pre-existing bullae. Lung ultrasound can also be a rapid and readily available diagnostic tool, especially in the ICU or emergency department setting.
Pleural Effusion
Large pleural effusions can contribute to worsening dyspnea in COPD patients by altering lung and chest wall mechanics and causing a restrictive ventilatory defect. If a large effusion is associated with elevated natriuretic peptides, a cardiovascular etiology, particularly acute HF, is more likely. Conversely, if associated with elevated CRP, parapneumonic effusion or empyema due to infection is more probable. Lung cancer should also be considered in the differential diagnosis of pleural effusion. Chest X-ray, ultrasound, and chest CT are all valuable for assessing pleural effusion in COPD patients with acute respiratory symptoms, helping to determine the presence, size, and characteristics of the effusion.
Anemia
Hemoglobin assessment is important in COPD patients. Polycythemia may indicate chronic hypoxemia, while anemia, a common comorbidity in stable COPD, can contribute to breathlessness. Anemic COPD patients tend to be older, have more severe airflow limitation, and a higher burden of cardiac and metabolic comorbidities. While anemia may not directly increase ECOPD incidence, it can logically worsen the dyspnea associated with exacerbations, potentially impacting ECOPD severity and duration. COPD patients with anemia tend to have longer hospital stays and increased mortality risk, potentially linked to their higher comorbidity burden.
Conclusion: Towards Precise Diagnosis in Acute Respiratory Distress
In conclusion, while many COPD patients presenting with acute respiratory symptom worsening may indeed be experiencing an ECOPD, it is imperative for clinicians in acute care settings to maintain a broad differential diagnosis. Acute decompensation of co-existing morbidities, particularly cardiovascular conditions, or the development of acute events such as pneumonia or pulmonary embolism, can significantly contribute to or even be the primary cause of respiratory distress in COPD patients. Therefore, a systematic approach to evaluating COPD patients with worsening respiratory symptoms, especially dyspnea without clear infectious signs, is crucial. This approach should include a thorough investigation for common comorbidities that can mimic or aggravate ECOPDs.
Accurate diagnosis is not only critical for individual patient management but also has broader implications for clinical research. Clinical trials evaluating interventions for ECOPDs must employ rigorous diagnostic criteria to ensure that enrolled patients are truly experiencing COPD exacerbations and not acute decompensations of other conditions mislabeled as ECOPDs. With the currently available clinical knowledge and diagnostic tools, achieving precise diagnoses in these complex cases is now within reach, paving the way for improved patient care and more robust clinical research in COPD management.