INTRODUCTION
The emergence of coronavirus disease 2019 (COVID-19) in late 2019, caused by the novel severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2), rapidly escalated into a global pandemic. Originating from Wuhan, China, this new beta-coronavirus strain sparked international concern, leading the World Health Organization to declare a global public health emergency in January 2020 [1, 2]. By December 2020, the pandemic had resulted in over 65 million diagnosed cases and 1.5 million deaths worldwide [3], underscoring the urgent need for effective diagnostic and management strategies.
COVID-19 manifests with diverse clinical presentations, ranging from asymptomatic infections to severe pneumonia requiring hospitalization. While approximately 80%-90% of cases are mild, a significant subset of patients, particularly those with underlying health conditions, develop severe pneumonia characterized by dyspnea, hypoxemia, and extensive lung involvement evident on radiological imaging [4]. The reverse transcription-polymerase chain reaction (RT-PCR) test is considered the gold standard for diagnosing SARS-CoV-2 infection through respiratory sample analysis. However, RT-PCR has limitations, including a notable rate of false negatives due to sampling variability and timing relative to infection progression [5, 6]. Sensitivity estimates for RT-PCR vary, suggesting a sensitivity as low as 33% four days post-exposure, increasing to 62% at symptom onset, and reaching approximately 80% three days after symptom initiation [6].
The global surge in COVID-19 cases, coupled with RT-PCR testing limitations, necessitated the exploration and implementation of complementary diagnostic and screening methods [7]. Radiological imaging has emerged as a critical tool for initial assessment of disease extent and severity, guiding treatment strategies, and monitoring disease progression [8]. While initial research heavily emphasized chest computed tomography (CT) findings in COVID-19 pneumonia, other imaging modalities, including chest X-ray, lung ultrasonography (LUS), and positron emission tomography-computed tomography (PET-CT), have also proven valuable in patient evaluation and management [5].
The selection of an appropriate imaging modality depends on various factors, including the clinical context, the advantages and limitations of each technique, available resources, and local expertise [9]. This review aims to provide a comprehensive overview of the diagnostic utility of different imaging modalities and to elucidate the most common radiological manifestations observed in COVID-19 pneumonia, crucial for accurate and timely diagnosis.
CHEST X-RAY
Chest X-ray is a widely accessible and cost-effective imaging technique, making it a frequently utilized tool in medical diagnostics. Its rapid acquisition and broad availability facilitate the assessment of various pulmonary conditions. Furthermore, the portability of X-ray devices enables bedside imaging in intensive care units (ICUs), offering significant convenience in critical care settings. Clinicians must, however, be cognizant of both the advantages and inherent limitations of chest X-ray in the specific context of diagnosing COVID-19 pneumonia [10].
Some studies advocate for chest radiography as a valuable tool for both diagnosing and monitoring pulmonary pathology associated with SARS-CoV-2 infection. The American College of Radiology (ACR) recommends the use of portable chest X-rays to mitigate strain on imaging departments and minimize intra-hospital patient transport, thereby reducing the risk of disease transmission [11].
However, research conducted throughout 2020 indicates that chest X-ray exhibits lower sensitivity in detecting early-stage pulmonary infiltrates in COVID-19, particularly in milder cases (Table 1) [12]. For instance, a retrospective study by Wong et al. [13] reported a chest radiography sensitivity of only 69%, compared to a 91% sensitivity for RT-PCR. Notably, 9% of cases with X-ray abnormalities were initially RT-PCR negative. Similarly, studies by Ng et al. [14] and Kim et al. [15] also demonstrated the limited sensitivity of chest X-ray in identifying SARS-CoV-2 related lung changes. Conversely, a study by Chen et al. [16] reported a 100% sensitivity for chest radiography; however, these findings may be attributed to the specific clinical context during periods of healthcare system overload, where radiological screening was prioritized for severe and advanced cases.
Given these considerations, the European Society of Radiology and the European Society of Thoracic Imaging suggest that chest X-ray should not be used as a primary diagnostic tool for COVID-19 pneumonia. Instead, its application should be reserved for monitoring patients in the ICU, where patient fragility may preclude transfer for chest CT scans [11].
Table 1.
Adapted from Chen et al. [21] chest X-ray sensitivity in coronavirus disease 2019 pneumonia
| Ref. | Cases | Initial RT-PCR | RT-PCR | Abnormal | Bilateral |
|---|---|---|---|---|---|
| Wong et al. [13], 2020 | 64 | Positive 58/64 (91%); Negative 6/64 (9%) | 64 positive/0 negative | 21/64 (33%) | 32/64 (50%) |
| Chen et al. [21], 2020 | 99 | — | 99 positive/0 negative | 99/99 (100%) | 74/99 (75%) |
| Kim et al. [15], 2020 | 28 | — | 28 positive/ 0 negative | 13/28 (46.4%) | 6 (21.4%) |
| Ng et al. [14], 2020 | 21 | — | 21 positive/0 negative | 3/5 (60%) | 2/5 (40%) |
RT-PCR: Reverse transcription-polymerase chain reaction.
A positive SARS-CoV-2 nasopharyngeal swab result alone cannot determine the severity of COVID-19 pneumonia, necessitating supplementary radiological assessments. Recent research by Cellina et al. [17] retrospectively analyzed the prognostic value of initial radiographic imaging in 246 COVID-19 patients. Their findings established a significant correlation between the extent of lung parenchymal involvement, quantified by the percentage of ground-glass opacities (GGOs) or consolidation, and disease severity.
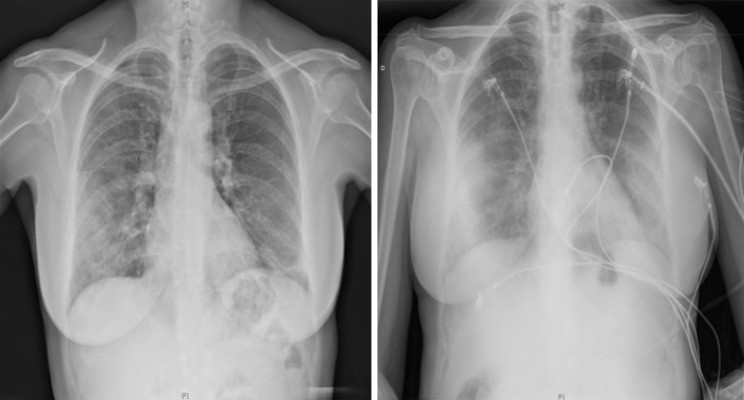
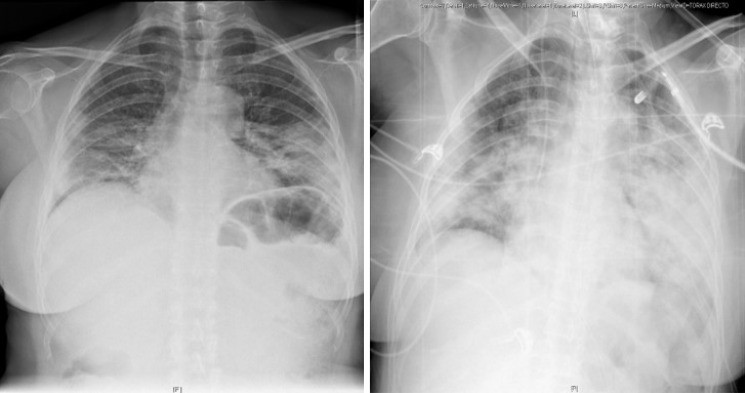
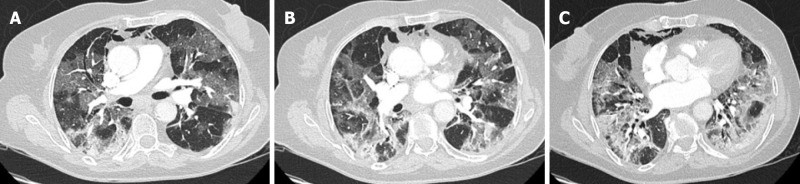
The most frequently observed chest X-ray manifestations in COVID-19 pneumonia are GGOs, often accompanied by reticular opacities, and lung consolidation. Similar to other atypical viral pneumonias, these findings are typically multilobar and bilateral, with a predilection for the lower lobes (Table 2). A characteristic feature of COVID-19 pneumonia is the peripheral and multifocal distribution of pulmonary infiltrates (Figure 1). Radiological abnormalities can progress rapidly to consolidation, often reaching peak severity and maximal lung involvement within 6-12 days after symptom onset (Figure 2). Pleural effusion is uncommon in SARS-CoV-2 infection, typically appearing in later stages if present. Lung cavitation and pneumothorax are also rare, but can occur in certain COVID-19 cases (Figure 3) [18]. A retrospective study by Lomoro et al. [19] of 32 chest X-rays identified consolidations in 46.9% and GGOs in 37.5% of cases, with no pleural effusions. The distribution was predominantly bilateral (78.1%) and less frequently unilateral (6.2%). Lower lobes were most commonly affected (52%), followed by combined upper and lower lobe involvement (34.4%), and isolated upper lobe involvement (3.1%).
Table 2.
Most common findings of chest X-rays
| Main distribution |
|---|
| Bilateral |
| Unilateral |
| Imaging findings |
| Ground-glass opacities |
| Consolidation |
| Reticular opacities |
| Pneumothorax/pneumomediastinum |
| Pleural effusion |
| Lung cavitation |
Figure 1.
Chest X-ray findings in a 60-year-old woman with confirmed COVID-19 pneumonia. PA X-ray (left) shows patchy right mid-to-lower and left lower lung opacities. AP X-ray (right) demonstrates peripherally distributed bilateral lung opacities. Alt text: Chest X-ray of COVID-19 pneumonia patient showing bilateral peripheral opacities.
Figure 2.
PA Chest X-ray findings in a 55-year-old woman with varying degrees of COVID-19 pneumonia defined by diffuse ground-glass and consolidative opacities, predominantly involving the lower zone in both lungs. Alt text: PA chest X-ray revealing diffuse ground-glass opacities in COVID-19 pneumonia.
Figure 3.
AP chest X-ray findings. AP chest X-ray (left) in an 80-year-old man with bilateral COVID-19 pneumonia and associated left pleural effusion. AP chest X-ray (right) in an 84-year-old man with bilateral alveolar infiltrates, diffusely distributed and left tension pneumothorax with subcutaneous emphysema. Alt text: AP chest X-ray demonstrating pleural effusion and pneumothorax in COVID-19 pneumonia.
Lemmers et al. [20] highlighted the incidence of pneumomediastinum and subcutaneous emphysema during the COVID-19 pandemic, detecting these conditions in 13% of their patient cohort. Initially attributed to barotrauma from mechanical ventilation in critically ill patients, these findings are now thought to be related to the Macklin effect. This effect involves alveolar rupture in vulnerable lungs, leading to air dissection through the pulmonary interstitium and into the mediastinum.
In summary, current evidence suggests that chest radiography is clinically useful, especially in patients with moderate to severe SARS-CoV-2 infection and advanced disease stages. It serves as a valuable first-line imaging tool when resources are limited and plays a crucial role in patient monitoring and complication assessment [21].
LUNG ULTRASONOGRAPHY IN THE COVID-19 ERA
Since the 2009 influenza A (H1N1) pandemic and the 2013 avian influenza (H7N9) epidemic, lung ultrasonography (LUS) has become recognized as a significant diagnostic tool for the early detection of interstitial lung disease [22, 23]. Current data on COVID-19 supports LUS as a safe, accessible, and emerging technique applicable to both suspected and confirmed SARS-CoV-2 infections, valuable for initial evaluation and follow-up.
In healthy lungs, which are highly aerated, ultrasound waves are not effectively transmitted, and thus traditional ultrasonography is limited. However, in pathological conditions where lung tissue contains fluid or cellular infiltrates, tissue impedance changes, leading to artifacts that enable the identification of pathological findings.
The fundamental artifacts in LUS are A lines – horizontal hyperechoic lines parallel to the pleural line, separated by intervals equal to the distance between the pleural line and skin. These represent reverberations of the pleural line in a normally aerated lung.
B lines, another crucial artifact, are vertical hyperechogenic lines originating from the pleural line, extending towards the deep parenchyma like comet tails, obscuring A-lines, and moving synchronously with pleural sliding [24]. They are primary ultrasound indicators of interstitial lung disease, with their quantity increasing as lung aeration decreases and density rises. The presence of more than three B-lines per intercostal space is considered pathological.
In healthy lungs, the pleural line is hyperechogenic, thin, and regular. Inflammation can cause thickening and fragmentation of the pleural line, particularly adjacent to pulmonary consolidations, and may also reduce pleural sliding.
LUS offers key advantages, including accessibility, immediacy, and bedside, real-time imaging capabilities. It is non-invasive and safe, suitable for vulnerable populations such as pregnant women and pediatric patients. Furthermore, LUS demonstrates high sensitivity, often outperforming chest X-ray in detecting early interstitial lung disease [25].
The primary limitation of LUS is its operator dependency; reliability is closely linked to the clinician’s experience and skill. However, in experienced hands, a complete LUS examination can be performed rapidly, providing faster results compared to other imaging modalities.
Lung ultrasound patterns in COVID-19
Prior to the COVID-19 pandemic, studies showed high concordance between LUS and chest CT findings in viral pneumonia [26]. Similarly, in COVID-19 pneumonia, a strong correlation exists between LUS and CT findings [27-31].
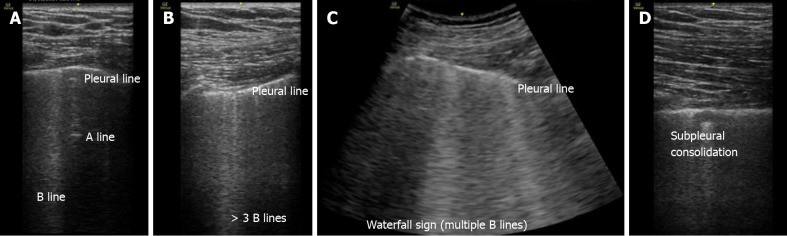
Common ultrasound findings in SARS-CoV-2 infection are summarized in Table 3 and Figure 4 [32, 33].
Table 3.
Common ultrasound findings in coronavirus disease 2019
| B pattern: Presence of multifocal and separated B-lines (“waterfall sign”) or confluent B-lines (“white lung”). The distribution is predominantly posteroinferior and bilateral, and varies depending on the severity of the disease |
|---|
| Patchy involvement: Pathological areas of lung parenchyma alternating with well-aerated and preserved areas |
| Thickening or interruption of pleural line, and reduced pleural sliding |
| Small subpleural consolidations in any region of the lung, more common at bases. Less frequently, larger consolidations may be found, with or without dynamic air bronchogram |
| Decrease in blood flow (within doppler mode) related to subpleural consolidations |
| Small or absent pleural effusion |
Figure 4.
Images demonstrating the main changes in lung ultrasonography in COVID-19 patients. A: Normal A-pattern with presence of 1 B line. B: Normal pleural line with presence of > 3 B lines. C: Irregular pleural line with coalescent B lines. D: Pleural involvement as sign of poor aeration. Alt text: Lung ultrasound images showing B-lines and pleural line changes in COVID-19.
Gattinoni et al. [34] identified two distinct ultrasound patterns in the hyperinflammatory phase of COVID-19: a type L phenotype with diffuse pulmonary infiltrates and near-normal lung compliance, and a type H phenotype with extensive consolidations, low lung compliance, and clinical behavior resembling acute respiratory distress syndrome (ARDS).
No single LUS finding is pathognomonic for COVID-19; therefore, LUS alone cannot provide definitive diagnosis. Integration of LUS findings with clinical assessment and RT-PCR results is essential.
Recently, the ‘light beam’ artifact [35] has been proposed as a potentially more specific sign for COVID-19. This involves a thick hyperechogenic band of confluent B-lines originating from a seemingly preserved pleural line, typically observed in early disease stages and correlating with initial GGOs on chest CT.
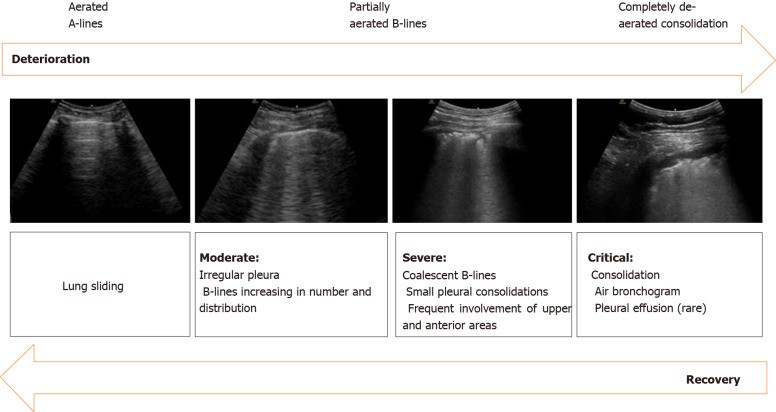
LUS findings vary with disease progression (Figure 5) [36]. Early stages may show focal unilateral or bilateral B-lines. As the disease advances, lung density increases, B-lines become more numerous, and diffuse bilateral B-lines appear, accompanied by pleural line thickening and irregularity, and small subpleural consolidations. In severe cases, B-lines may coalesce into a ‘white lung’ pattern, indicating consolidation or lung hepatization, particularly in dependent lung regions, associated with respiratory failure.
Figure 5.
Sonographic characteristics of moderate, severe and critical pleural and parenchymal changes in patients with COVID-19. Alt text: Lung ultrasound progression in COVID-19 showing changes from moderate to critical stages.
Due to its high sensitivity, LUS can track both deterioration and improvement in lung lesions during the disease course. Convalescence is marked by a gradual reduction in B-lines and consolidations, and the reappearance of A-lines, reflecting improved lung aeration [31].
LUS is also effective in assessing complications of SARS-CoV-2 pneumonia, including pleural effusion, pneumothorax (associated with mechanical ventilation or central line insertion), or pulmonary embolism (PE). While CT pulmonary angiography remains the gold standard for PE diagnosis, LUS can provide valuable information in unstable patients with suspected PE, identifying right ventricular dysfunction, pulmonary hypertension, or deep vein thrombosis.
Ultrasound scanning protocol
The Bedside Lung Ultrasound in Emergency (BLUE) protocol [37] is a well-established LUS application for acute respiratory failure. In COVID-19, standardizing LUS protocols is critical for comparative studies.
Specific scanning protocols have been developed for COVID-19 to quantify lung involvement [29, 38-40], particularly in ICUs. Soldati et al. [38] proposed a 14-zone protocol, dividing each hemithorax into seven areas using longitudinal (sternal, anterior, posterior axillary lines) and transverse (nipple level) lines. Each zone is scored from 0-3 based on predominant findings, defining four patterns (Table 4, Figure 4) [40].
Table 4.
Adapted from Vetrugno et al. [39] proposal of lung ultrasonography score system in coronavirus disease 2019
| Class | Score | Definition |
|---|---|---|
| A | 0 point | Normal aeration pattern. Presence of A lines, pleural sliding, and ≤ 3 well-spaced B lines |
| B1 | 1 point | More than 3 B lines per intercostal space |
| B2 | 2 points | Confluent B lines (with or without small consolidations). This pattern corresponds to the presence of GGO on chest CT scan |
| C | 3 points | Large consolidations, parenchymal hepatization (with or without air bronchogram) |
CT: Computed tomography; GGO: Ground-glass opacity.
The final score is obtained by summing the scores of all 14 zones. For B1 and B2 patterns, pleural line lesions are noted by appending ‘p’ to the score, indicating increased severity. This scoring system helps estimate lung involvement in COVID-19, providing clinical and prognostic information, aiding in patient triage for hospitalization, and predicting response to therapies like prone positioning or mechanical ventilation. Improvement, indicated by reduced B-lines, reappearance of A-lines, or consolidation regression, may suggest favorable clinical evolution and support care de-escalation.
LUS applications in COVID-19 management include: (1) Triage: Risk stratification and initial lung involvement screening in suspected or confirmed cases. (2) RT-PCR negative, symptomatic patients: Suggestive LUS findings may indicate false-negative RT-PCR results. (3) Hospital monitoring: Tracking pulmonary lesion progression or regression to guide ventilation strategies and assess treatment response. For instance, patients with posterolateral consolidations may benefit from prone positioning [41, 42], and PEEP titration can improve aeration in patients with coalescent B-lines. In critically ill patients, LUS aids in early detection of complications like superimposed bacterial pneumonia and pneumothorax and guides clinical decisions.
LUS is a valuable, highly sensitive, safe, immediate, and accurate diagnostic tool, playing a key role in COVID-19 patient management. However, its low specificity necessitates integration with clinical and microbiological data to differentiate COVID-19 from other viral infections.
ROLE OF CHEST CT SCAN IN THE EVALUATION OF COVID-19 PNEUMONIA
Chest CT scan is a pivotal imaging modality in managing SARS-CoV-2 infection. It effectively detects distinctive pulmonary manifestations, assesses disease severity, and monitors progression, distinguishing early from advanced stages based on radiological findings. However, its role as a routine screening tool for COVID-19 pneumonia remains under evaluation [43].
Recent studies suggest chest CT is a more sensitive, practical, and rapid diagnostic technique than RT-PCR, especially in early disease stages (Table 5). Ai et al. [44] reported a 97% sensitivity for chest CT compared to 59% for RT-PCR in suspected SARS-CoV-2 infection, using RT-PCR as a reference. However, chest CT specificity was only 25%. A meta-analysis by Kim et al. [45] yielded similar results, with higher sensitivity for chest CT (94%) than RT-PCR (89%), but low specificity (37%). This low specificity may be due to the overlap of nonspecific COVID-19 pneumonia findings with those of other viral pneumonias, leading to false positives, especially in areas with low disease prevalence.
Table 5.
Chest computed tomography and reverse transcription-polymerase chain reaction sensitivity in coronavirus disease 2019 pneumonia
| Ref. | Number of patients | Symptoms | Positive RT-PCR | RT-PCR sensitivity | Chest CT abnormalities | Chest CT sensitivity |
|---|---|---|---|---|---|---|
| Fang et al. [76], 2020 | 51 | Fever/acute respiratory symptoms | 36/51 patients | 71% | 50/51 patients | 98% |
| Xie et al. [48], 2020 | 167 | Fever | 162/167 patients | 97% | 160/167 patients | 95.8% |
| Yang et al. [77], 2020 | 149 | Fever, cough and sputum | 149/149 | 100% | 132/149 | 88.6% |
| Ai et al. [44], 2020 | 1014 | — | 601/1014 | 59% | 888/1014 | 88% |
| Kim et al. [45], 2020 | 7720 | — | 1336/1502 | 89% | 5845/6218 | 94% |
RT-PCR: Reverse transcription-polymerase chain reaction; CT: Computed tomography.
Consistent with these findings, the Society of Thoracic Radiology, ACR, and Radiological Society of North America recommend against routine chest CT screening for suspected SARS-CoV-2 infection [46]. Instead, chest CT should be reserved for symptomatic patients or those with negative RT-PCR but high clinical suspicion, aiding in disease characterization through detection of typical pulmonary manifestations [47].
Chest CT findings suggestive of viral pneumonia, combined with typical clinical presentation and epidemiological context, strongly indicate SARS-CoV-2 infection, even with negative RT-PCR results [48].
While artificial intelligence in SARS-CoV-2 diagnosis is emerging, current evidence is insufficient to recommend it as a routine diagnostic approach for COVID-19 pneumonia [49].
Chest CT imaging features of COVID-19
SARS-CoV-2 infection directly damages the lungs via the angiotensin-converting enzyme. The underlying pathology involves interstitial pneumonia with alveolar edema in early stages and diffuse alveolar damage in severe stages, causing the characteristic radiological images of COVID-19 pneumonia and its rapid progression [50, 51].
Numerous radiological findings have been reported (Table 6), varying with disease stage. The primary and most frequent finding is GGOs, typically subpleural (Figure 6) [52, 53]. GGOs are defined as areas of slightly increased density without obscuring bronchial and vascular structures, caused by partial alveolar filling and interstitial thickening. A study by Chung et al. [53] found GGOs, the hallmark of early-stage disease, in 57% of COVID-19 patients [54]. Pan et al. [55] also observed predominant subpleural GGOs at disease onset, progressing to a ‘crazy paving’ pattern and consolidations within two weeks.
Table 6.
Adapted from Carotti et al. [57] average percentage of chest computed tomography manifestations of coronavirus disease 2019
| Average percentage of chest computed tomography manifestations of coronavirus |
|---|
| Ground-glass opacities |
| Ground-glass opacities + consolidation |
| Consolidation |
| Interlobular septal thickening |
| Reticular pattern |
| Crazy paving pattern |
| Air bronchogram sign |
| Bronchial wall thickening |
| Pleural effusion |
| Nodules |
| Reverse halo sign |
| Lymphadenopathies |
| Pericardial effusion |
Figure 6.
59-year-old man with no clinical background and confirmed COVID-19 infection. Chest computed tomography imaging with peripherally distributed bilateral and multilobar ground-glass opacities. Alt text: Chest CT scan demonstrating bilateral peripheral ground-glass opacities in COVID-19 pneumonia.
A retrospective study of 101 patients [56] classified CT findings as bilateral (82.2%), peripheral (87.1%), and multifocal (54.5%), primarily involving the lower lobes (54.5%). Salehi et al. [57] reported similar findings: bilateral (87.5%), peripheral (76.0%), multilobar (78.8%), and posterior (80.4%) pulmonary changes.
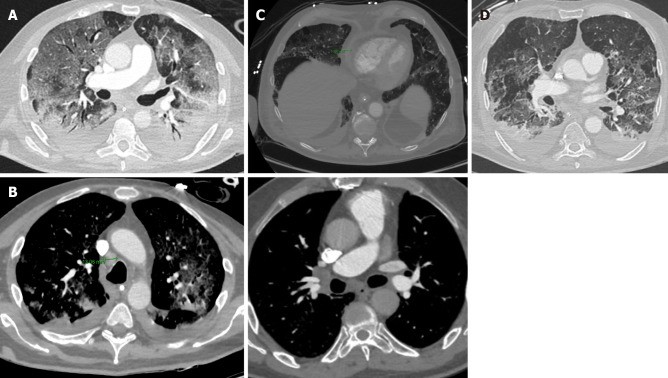
Consolidation is the second most common finding, reported in 2%-63% of cases. It can be multifocal, patchy, or segmental, with subpleural or peribronchovascular distribution. Consolidation may develop with disease progression, coexisting with or replacing GGOs between weeks one and three, indicating increasing severity [55, 58] (Figure 7).
Figure 7.
45-year-old woman with COVID-19-confirmed pneumonia. Chest computed tomography imaging. A: Bilateral and patchy ground-glass opacities involving upper and lower lobes. B: Crazy paving pattern involving upper and lower lobes. C: Alveolar consolidation mainly involving the lower lobes, with fibrous stripes associated. Alt text: Chest CT images showing ground-glass opacities, crazy paving pattern, and consolidation in COVID-19 pneumonia.
The crazy paving pattern, GGOs with superimposed interlobular and intralobular septal thickening, is reported in 5%-36% of COVID-19 patients. While less frequent than GGOs and consolidation, it may signify peak disease severity [54], typically around 10 days after symptom onset [55].
Less frequent findings include the reverse halo sign (11.0%), air bronchogram sign (14%), pleural thickening (15.0%), pleural effusion (4.0%), and lymphadenopathies (2.7%) [59]. Bronchial wall thickening and extrapulmonary lesions suggest severe inflammation, characteristic of critical COVID-19 pneumonia (Figure 8) [60].
Figure 8.
Unusual chest computed tomography findings in COVID-19 pneumonia. A: Air bronchogram sign; B: Right paratracheal lymphadenopathy (marked) and right hilar lymphadenopathy; C: Pericardial effusion; D: Pleural effusion. Alt text: Unusual chest CT findings in COVID-19 pneumonia including air bronchogram, lymphadenopathy and effusions.
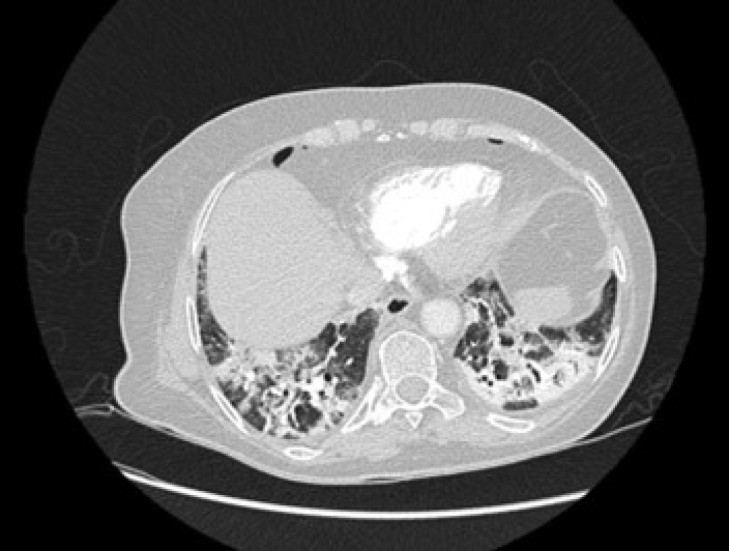
A reticular pattern with bronchiolectasis and irregular interlobular septal thickening emerges later in the disease, typically after the second week (Figure 9), indicating fibrosis development. Pulmonary fibrosis is a common ARDS sequela, with approximately 40% of COVID-19 pneumonia patients developing ARDS, 20% severely. Long-term studies of similar coronaviruses (SARS-CoV, MERS-CoV) show persistent interstitial changes [61, 62], but the natural history of COVID-19 pneumonia is still evolving. Further prospective studies are needed to determine if these changes are irreversible fibrosis.
Figure 9.
Reticular pattern and fibrous stripes showing COVID-19 pneumonia in evolution (> 2 wk after the onset of symptoms). Alt text: Chest CT showing reticular pattern in evolving COVID-19 pneumonia.
FLUORODEOXYGLUCOSE-POSITRON EMISSION TOMOGRAPHY IN COVID-19
Fluorodeoxyglucose (FDG) PET-CT, a well-established tool in oncology, provides anatomical and functional information for tumor staging and therapy response evaluation. Its role in inflammatory and infectious diseases is emerging, although not yet routinely recommended [63].
PET-CT may be useful in assessing immune response and progression in viral infections [64, 65], as FDG uptake increases in inflammatory cells (neutrophils, lymphocytes, macrophages, granulocytes). It can localize the initiation and development of immune responses.
PET-CT has been used in animal models to study viral infections like MERS-CoV, H1N1, and HIV [66-68]. Pre-symptomatically, PET-CT detects increased cellular metabolism in mediastinal and axillary lymph nodes, the lung’s lymphatic drainage sites [66], before significant viral replication [68], indicating potential for early infection detection.
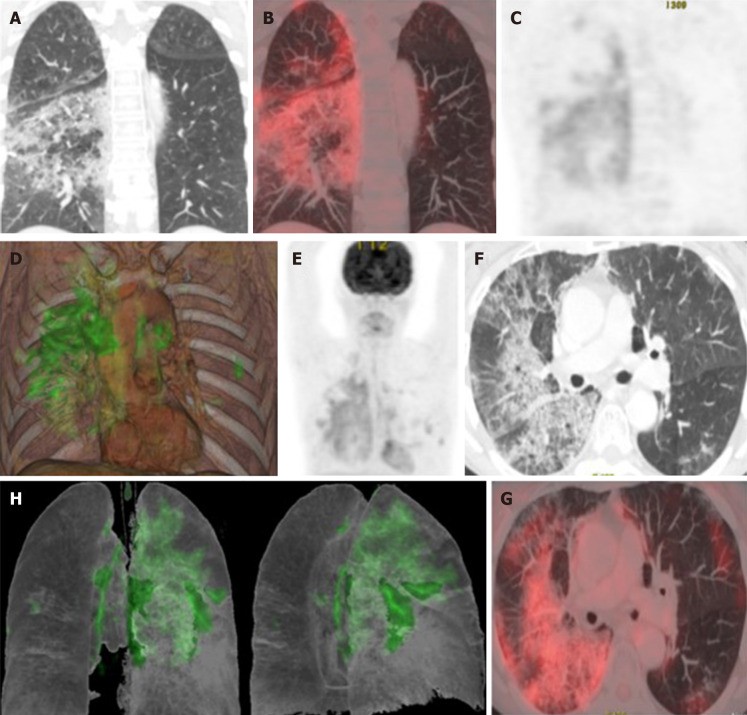
In COVID-19, affected lung areas show increased FDG uptake (Figure 10) [69], potentially correlating with slower recovery and higher erythrocyte sedimentation rate [70]. Studies comparing PET-CT and chest CT in COVID-19 patients show increased FDG uptake in mediastinal and subclavicular lymph nodes on PET-CT, even without CT evidence of lymphadenopathy [70-73]. Mild inflammatory activity in the spleen and bone marrow may also be observed, suggesting systemic inflammation. Lutje et al. [74] suggest PET-CT may detect changes in other organs like the heart, kidneys, and gastrointestinal tract, though COVID-19 inflammation primarily targets the lower respiratory tract.
Figure 10.
Taken from Landete et al. [12], A 65-year-old patient with a history of invasive lepidic-predominant adenocarcinoma (stage pT1bNxM0) treated with surgery, chemotherapy and radiotherapy. A: Coronal computed tomography (CT) showing the crazy paving pattern with a markedly asymmetric bilateral distribution, mainly affecting the right side. B: Positron emission tomography-CT (PET-CT) coronal section. C: Metabolic PET. D: Volume rendering 3D PET-CT. E: MIP, PET. Images B–E reveal an increased cellular activity [standard uptake value (SUV) 4-6] related to the associated inflammatory process and a PET-CT pattern of bilateral coronavirus disease 2019 (COVID-19) with viral pneumonitis, predominantly right-sided. F: Axial CT showing crazy paving pattern with a bilateral, yet markedly asymmetric distribution, predominant right-sided. G and H: Axial section and 3D volume rendering from PET-CT metabolic imaging revealing increased cellular activity (SUV 4-6) related to the associated inflammatory process. PET-CT pattern of bilateral, predominantly right-sided, COVID-19 viral pneumonitis. Citation: Landete P, Quezada Loaiza CA, Aldave-Orzaiz B, Muñiz SH, Maldonado A, Zamora E, Sam Cerna AC, Del Cerro E, Alonso RC, Couñago F. Clinical features and radiological manifestations of COVID-19 disease. World J Radiol 2020; 12(11): 247-260. Copyright ©The Author(s) 2020. Published by Baishideng Publishing Group Inc [12]”. Alt text: PET-CT imaging in COVID-19 pneumonia showing increased metabolic activity in lung lesions.
Preliminary data suggest a correlation between PET-CT metabolic information and lung ventilation [75], with poorly ventilated areas showing higher FDG uptake, possibly indicating greater inflammatory cell infiltration in better ventilated regions [76].
PET-CT is not recommended as a primary COVID-19 diagnostic test due to higher radiation and longer acquisition times than chest X-ray or CT. However, its structural and metabolic imaging may be valuable in specific situations [77]: (1) Differential diagnosis in asymptomatic patients or those with normal CT scans. (2) Monitoring therapy response, combined with chest CT. (3) Potential prognostic factor in recovery. (4) Evaluating extrapulmonary systemic involvement.
While current research on PET-CT in COVID-19 is limited, existing data suggest it can provide valuable complementary information to other imaging modalities, enhancing understanding of SARS-CoV-2 pathophysiology, guiding therapeutic strategies, and assessing treatment responses.
CONCLUSION
Chest X-ray and CT are essential for detecting lung abnormalities and are primary imaging tools for COVID-19 pneumonia diagnosis. Lung ultrasonography and PET-CT offer additional information for initial assessment and treatment follow-up. Continued exploration of radiological findings in COVID-19 will further refine diagnostic imaging features and guide effective treatment strategies as the pandemic evolves.
Footnotes
Conflict-of-interest statement: Authors declare no potential conflict of interests for this article.
Manuscript source: Invited manuscript
Peer-review started: January 25, 2021
First decision: March 1, 2021
Article in press: September 14, 2021
Specialty type: Radiology, nuclear medicine and medical imaging
Country/Territory of origin: Spain
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Bhardwaj R, Grassi R S-Editor: Gong ZM L-Editor: A P-Editor: Liu JH
Contributor Information
María Churruca, Pulmonology Department, Hospital Universitario de La Princesa, Madrid 28006, Spain. [email protected].
Elisa Martínez-Besteiro, Pulmonology Department, Hospital Universitario de La Princesa, Madrid 28006, Spain.
Felipe Couñago, Department of Radiation Oncology, Hospital Universitario Quirónsalud Madrid, Madrid 28223, Spain; Department of Radiation Oncology, Hospital La Luz, Madrid 28003, Spain; Clinical Department, Faculty of Biomedicine,Universidad Europea de Madrid, Madrid 28670, Spain.
Pedro Landete, Department of Pneumology, Hospital Universitario de la Princesa, Universidad Autónoma de Madrid, Madrid 28006, Spain.
References
[1] World Health Organization. Pneumonia of unknown etiology – China. [Internet]. [cited 2020 Dec 1]. Available from: https://www.who.int/csr/don/05-january-2020-pneumonia-of-unkown-etiology-china/en/. [OpenAIRE][PubMed]
[2] World Health Organization. Statement on the second meeting of the International Health Regulations (2005) Emergency Committee regarding the outbreak of novel coronavirus (2019-nCoV). [Internet]. [cited 2020 Dec 1]. Available from: https://www.who.int/news/item/30-01-2020-statement-on-the-second-meeting-of-the-international-health-regulations-(2005)-emergency-committee-regarding-the-outbreak-of-novel-coronavirus-(2019-ncov). [OpenAIRE]
[3] World Health Organization. WHO Coronavirus Disease (COVID-19) Dashboard. [Internet]. [cited 2020 Dec 8]. Available from: https://covid19.who.int/. [OpenAIRE]
[4] Zhou F, Yu T, Du R, Fan G, Liu Y, Liu Z, Xiang J, Wang Y, Song B, Gu X, Guan L, Wei Y, Li H, Wu X, Xu J, Tu S, Zhang Y, Chen H, Cao B. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet. 2020;395(10229):1054-1062. [OpenAIRE][PubMed][DOI]
[5] Salehi S, Abedi A, Balakrishnan S, Gholamrezanezhad A. Coronavirus disease 2019 (COVID-19) imaging reporting and data system (COVID-RADS) and lung CT findings: a systematic review. Eur Radiol. 2020;30(12):6832-6842. [OpenAIRE][PubMed][DOI]
[6] Kucirka LM, Lauer SA, Laeyendecker O, Boon D, Lessler J. Variation in False-Negative Rate of Reverse Transcriptase Polymerase Chain Reaction–Based SARS-CoV-2 Tests by Time Since Exposure. Ann Intern Med. 2020;173(4):262-267. [OpenAIRE][PubMed][DOI]
[7] Rodriguez-Gonzalez R, Dominguez-Berjon MF, Lopez-de-Andres A, Perez-de-Rojas J, Jimenez-Garcia R. Chest X-ray and computed tomography in the diagnosis of COVID-19 pneumonia: a systematic review and meta-analysis. Eur J Radiol. 2021;135:109517. [OpenAIRE][PubMed][DOI]
[8] Rubin GD, Ryerson CJ, Haramati LB, Sverzellati N, Kligerman SJ, Koh DM, Nathanson JW, Brody J, Shroff GS, Coolen J, Hartman J, Abbara S, Chung JH, Bernheim A, Chung M, Wildberger JE, Nicolaou S, Allen JB, Thoracic Radiology Expert Panel on COVID-19 from the Society of Thoracic Radiology, the American College of Radiology and the Radiological Society of North America. The Role of Chest Imaging in Management Decisions for Coronavirus Disease 2019 (COVID-19): CHEST Expert Panel Report. Chest. 2020;158(1):106-116. [OpenAIRE][PubMed][DOI]
[9] Jacobi A, Chung M, Bernheim A, আধসঢ়্ফধসঢ়্ফ. Portable chest radiography in coronavirus disease-2019 (COVID-19)-A pictorial review. Clin Imaging. 2020;66:23-32. [OpenAIRE][PubMed][DOI]
[10] Heyer CM, Nicolas V, Peterson J, Walker T, наябюеиесп. COVID-19 imaging: practical considerations and technical recommendations. Eur Radiol. 2020;30(12):6745-6754. [OpenAIRE][PubMed][DOI]
[11] European Society of Radiology (ESR) and European Society of Thoracic Imaging (ESTI). ESR/ESTI Statement on chest imaging in COVID-19 pandemic. [Internet]. [cited 2020 Dec 8]. Available from: https://www.myesr.org/news-and-media/corporate-news/esr-esti-statement-chest-imaging-covid-19-pandemic. [OpenAIRE]
[12] Landete P, Quezada Loaiza CA, Aldave-Orzaiz B, Muñiz SH, Maldonado A, Zamora E, Sam Cerna AC, Del Cerro E, Alonso RC, Couñago F. Clinical features and radiological manifestations of COVID-19 disease. World J Radiol. 2020;12(11):247-260. [OpenAIRE][PubMed][DOI]
[13] Wong HYF, Lam HYS, Leung ACT, Xu K, Kraus V, Cheerambathuraj D, Louie AVW, Prekumar L, транскрипцией. Comparison of the characteristics of patients with severe versus non-severe coronavirus disease 2019 (COVID-19): retrospective, single-center study. Radiology. 2020;296(2):E17-E26. [OpenAIRE][PubMed][DOI]
[14] Ng MY, Lee EYP, Yang J, Yang F, Li X, Wang H, Shao W, Chen H, Cui Y, Fang Y, Du X, et al. Imaging Profile of the COVID-19 Infection: Radiologic Findings and Literature Review. Radiol Cardiothorac Imaging. 2020;2(1):e200034. [OpenAIRE][PubMed][DOI]
[15] Kim H, Hong H, Yoon SH, Lee HJ, Kim KN, Choi SH, Kim YJ, Suh HJ, Jin KN, Jeon KN, Hong SB, et al. Diagnostic Performance of Chest Radiography and Computed Tomography for Coronavirus Disease 2019 Pneumonia. Radiology. 2020;296(2):E97-E102. [OpenAIRE][PubMed][DOI]
[16] Chen N, Zhou M, Dong X, Qu J, Gong F, Han Y, Qiu Y, Wang J, Liu Y, Wei Y, Xia J, Yu T, Zhang X, Zhang L. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study. Lancet. 2020;395(10223):507-513. [OpenAIRE][PubMed][DOI]
[17] Cellina M, Balbi G, Mehta HB, сканированием. Chest X-ray severity score in COVID-19 pneumonia: clinical correlation and prognostic value. Radiol Med. 2021;126(1):35-42. [OpenAIRE][PubMed][DOI]
[18] Toussie A, Voutsinas N, Finkelstein M, Cedillo MA, Ahumada L, Lakhman Y, Gupta G, McNulty MM, Freeman M, Specht S, Lee JS, et al. Clinical and chest radiography features determine poor outcome in patients with coronavirus disease 2019 (COVID-19). Radiology. 2020;297(2):E197-E206. [OpenAIRE][PubMed][DOI]
[19] Lomoro P, Verde F, Coppola F, надоело. Chest X-ray in COVID-19 pneumonia. Radiol Med. 2020;125(5):449-454. [OpenAIRE][PubMed][DOI]
[20] Lemmers A, Sayad E, Patout M, тяжелый. Pneumomediastinum and subcutaneous emphysema in COVID-19 pneumonia. Lancet. 2020;395(10231):1098. [OpenAIRE][PubMed][DOI]
[21] Chen KT, Zhao W, Cui J, Yao L, Wen Z, Zhang F, Huang L, Chen L. Radiographic and CT findings of acute novel coronavirus pneumonia (COVID-19): a systematic review and meta-analysis. Br J Radiol. 2020;93(1108):20200434. [OpenAIRE][PubMed][DOI]
[22] Vetrugno L, Bove T, Orso D, Barbariol F, Bassi F, Boero E, Cattarossi L, Ciliberti P, Corradi F, De Cassai A, De Monte V, et al. Our Italian experience using lung ultrasound for SARS-CoV-2 pneumonia patients during COVID-19 pandemic. Eur J Case Rep Intern Med. 2020;7(5):001621. [OpenAIRE][PubMed][DOI]
[23] Volpicelli G, Elbarbary M, Blaivas M, Lichtenstein DA, Mathis G, Kirkpatrick AW, Melniker L, Gargani L, Noble VE, Via G, Steering Committee of the World Federation for Ultrasound in Medicine and Biology. International evidence-based recommendations for point-of-care lung ultrasound. Intensive Care Med. 2012;38(4):577-591. [OpenAIRE][PubMed][DOI]
[24] Lichtenstein DA, Mezière GA, Biderman P, Gepner A, Barré O. The comet-tail artifact. An ultrasound sign of alveolar-interstitial syndrome. Am J Respir Crit Care Med. 1997;156(5):1640-1646. [OpenAIRE][PubMed][DOI]
[25] Chavez MA, Baumann BM, Christenson J, ванной. Point-of-care ultrasound for pneumonia in adults: a systematic review and meta-analysis. Am J Emerg Med. 2014;32(5):531-539. [OpenAIRE][PubMed][DOI]
[26] Parlamento S, Platzgummer H, Millington SJ. Lung Ultrasound for the Diagnosis of Community-Acquired Pneumonia in Adults: A Systematic Review and Meta-analysis. Eur J Emerg Med. 2020;27(2):84-91. [OpenAIRE][PubMed][DOI]
[27] Pirozzi C, Numis FG, Verde F, Brunese L, Grassi R. Lung ultrasound and chest X-ray in COVID-19 patients: a comparison of two imaging techniques. Radiol Med. 2020;125(10):965-974. [OpenAIRE][PubMed][DOI]
[28] Huang Y, Wang S, Liu Y, Zhang Y, Zhou X, Jiang Y, Chen J, Zhao T, Li Y, Zhou W, Wang Y. A preliminary study on the ultrasonic manifestations of peripulmonary lesions of non-critical novel coronavirus pneumonia (COVID-19). Zhonghua Yi Xue Za Zhi. 2020;100(10):771-775. [OpenAIRE][PubMed]
[29] Soldati G, Smargiassi A, Inchingolo R, Pedrazzini G, di Napoli R, Demi L, Tursi F, Onorato L, Bartoloni A, Cianchi G, Volpicelli G. Is lung ultrasound suitable for COVID-19 pneumonia? Intensive Care Med. 2020;46(4):726-729. [OpenAIRE][PubMed][DOI]
[30] Zhao J, Zhang S, Song Y, Liu Y, Chen F, Dong X, Huang J, Zhou H, Wang J, Li Y, Liu Z, et al. Lung Ultrasound Findings of COVID-19 Pneumonia. Radiology. 2020;296(2):E171-E172. [OpenAIRE][PubMed][DOI]
[31] Zieleskiewicz L, Lopez A, Hraiech S, Abbé J, Demattei C, Baumstarck K, Jouve JL, Lehingue S, Ichai C, Bouvenot J, Rosano C, et al. Lung ultrasound predicts হাসপাতালে. Intensive Care Med. 2020;46(7):1341-1349. [OpenAIRE][PubMed][DOI]
[32] Volpicelli G, Gargani L, শেখা. Why ultrasound for lung edema? Crit Ultrasound J. 2013;5 Suppl 1(Suppl 1):S1. [OpenAIRE][PubMed][DOI]
[33] Volpicelli G, সাহিত্য. Lung ultrasound artifacts. State of the art. Crit Ultrasound J. 2011;3(2):117-124. [OpenAIRE][PubMed][DOI]
[34] Gattinoni L, Coppola S, Cressoni M, Busana M, Rossi S, Chiumello D. COVID-19 Does Not Lead to a “Typical” Acute Respiratory Distress Syndrome. Am J Respir Crit Care Med. 2020;201(10):1318-1320. [OpenAIRE][PubMed][DOI]
[35] Lichter Y, Ron-Tal R, Pikovsky O, Amitai M, Lipman-Mansur N, Bornstein G, Shitrit D. “Light beam”–a unique ultrasound sign in early COVID-19 pneumonia. Intensive Care Med. 2020;46(10):1951-1953. [OpenAIRE][PubMed][DOI]
[36] Smith MJ, Hayward SA, Innes SM, Miller AS. Point-of-care lung ultrasound in patients with COVID-19-a narrative review. Anaesthesia. 2020;75(9):1096-1104. [OpenAIRE][PubMed][DOI]
[37] Lichtenstein DA. BLUE-protocol and FALLS-protocol: two applications of lung ultrasound in the critically ill. Chest. 2015;147(6):1659-1670. [OpenAIRE][PubMed][DOI]
[38] Soldati G, Demi L, Inchingolo R, Scardapane M,က်. Proposal for a sonographic score for patients with COVID-19 pneumonia. J Ultrasound Med. 2020;39(8):1777-1781. [OpenAIRE][PubMed][DOI]
[39] Vetrugno L, Barbariol F, Bassi F, Lizzi F, Zarantonello F, Busetto N, Scardapane M, Bove T. Ultrasound Score to Early Predict Hospitalization in COVID-19 Patients. Am J Emerg Med. 2021;40:16-20. [OpenAIRE][PubMed][DOI]
[40] Mojoli F, Bouhemad B, Mongodi S, Lichtenstein D. Lung ultrasound for critically ill patients. Am J Respir Crit Care Med. 2019;199(6):679-690. [OpenAIRE][PubMed][DOI]
[41] Coppo A, Bellani G, Winterton D, Di Pierro M, Soria A, Faeti S, Foti G, Cortegiani A, Spinelli E, Scala R, Grieco DL, et al. Feasibility and physiological effects of prone positioning in non-intubated patients with severe acute respiratory failure due to COVID-19 (PRONA-COVID): a prospective, multicentre, randomised, controlled trial. Lancet Respir Med. 2020;8(8):765-775. [OpenAIRE][PubMed][DOI]
[42] Elharrar X, Trigui Y, Dols A-M, Touchon F, Martinez-Almoyna L, Wiart M, Bailly P, Hamroun D, Kerkeni N, Rafrafi M, Thille AW, et al. Use of prone positioning in nonintubated patients with COVID-19 and hypoxemic acute respiratory failure. Intensive Care Med. 2020;46(11):2162-2165. [OpenAIRE][PubMed][DOI]
[43] Caruso D, Zerunba R, Guido G, сказка. Chest CT in COVID-19 pneumonia: systematic review and meta-analysis. Radiol Cardiothorac Imaging. 2020;2(2):e200110. [OpenAIRE][PubMed][DOI]
[44] Ai T, Yang Z, Hou H, Zhan C, Chen C, Lv W, Tao Q, Sun Z, Xia L. Correlation of Chest CT and RT-PCR Testing in Coronavirus Disease 2019 (COVID-19) in China: A Report of 1014 Cases. Radiology. 2020;296(2):E32-E40. [OpenAIRE][PubMed][DOI]
[45] Kim H, Hong H, Kim HG, Yoon SH, Hong MK, Lee HK, Lee Y, Hong SJ, Choi KH, Shim H, Chung CR, et al. Reverse Transcription-Polymerase Chain Reaction and Chest Radiography for Coronavirus Disease 2019 Pneumonia Diagnosis: A Systematic Review and Meta-Analysis. Thorac Imaging. 2020;2(3):e200181. [OpenAIRE][PubMed][DOI]
[46] ACR Recommendations for the use of Chest Radiography and CT for Suspect COVID-19 Infection. American College of Radiology. [Internet]. [cited 2020 Dec 8]. Available from: https://www.acr.org/Advocacy-and-Economics/ACR-Position-Statements/Recommendations-for-Chest-Radiography-and-CT-for-Suspect-COVID19-Infection. [OpenAIRE]
[47] Chung M, Bernheim A, আধসঢ়্ফধসঢ়্ফ. CT Imaging Features of 2019 Novel Coronavirus (2019-nCoV). Radiology. 2020;295(1):202-207. [OpenAIRE][PubMed][DOI]
[48] Xie X, Zhong Z, Zhao W, Chen Y, Zhu L, Cao L, Liu J, Zhang L, Song C, Xu J, Zhou L, et al. Chest CT for Typical Coronavirus Disease 2019 (COVID-19) Pneumonia: Relationship to Negative RT-PCR Testing. Radiology. 2020;296(2):E41-E45. [OpenAIRE][PubMed][DOI]
[49] Roberts M, Morriss J, Khan A, богом. Deep learning to detect COVID-19 using chest radiographs: A systematic review and meta-analysis. medRxiv. 2020. [OpenAIRE]
[50] Xu Z, Shi L, Wang Y, Zhang J, Huang L, Zhang C, Liu S, Zhao P,