Pediatric Acute Respiratory Distress Syndrome (PARDS) remains a significant challenge in critical care medicine, contributing to substantial mortality and long-term health issues in children. Recognizing the need for specialized guidelines, the Pediatric Acute Lung Injury Consensus Conference (PALICC) previously established a PARDS-specific definition and management strategies in 2015. However, the rapidly evolving landscape of critical care necessitates continuous updates to incorporate new research, technological advancements, and address previously unmet needs, especially in resource-limited settings (RLS).
This article highlights the critical updates from the Second Pediatric Acute Lung Injury Consensus Conference (PALICC-2), focusing on the refined diagnostic criteria and evidence-based management principles for PARDS. These updated guidelines aim to facilitate the implementation of best practices and guide future research in pediatric critical care.
PALICC-2 Methodology: A Robust Consensus for Pediatric ARDS
The PALICC-2 guidelines are the product of an international consensus conference, uniting 52 multidisciplinary experts in PARDS from 15 countries, alongside methodology specialists. This rigorous process ensured a comprehensive and globally relevant update to the PARDS guidelines. The methodology involved:
- Systematic Literature Reviews: Eleven subgroups conducted thorough reviews across 11 key areas related to PARDS, ensuring all relevant evidence was considered.
- GRADE Methodology: The Grading of Recommendations, Assessment, Development, and Evaluation (GRADE) approach was used to evaluate the evidence quality and formulate clinical practice recommendations.
- Consensus Voting: Recommendations and statements underwent a rigorous voting process, requiring over 80% agreement among experts to ensure strong consensus.
- Harmonization and Revision: Final recommendations were meticulously revised and harmonized to ensure clarity and consistency across all guidelines.
This meticulous methodology resulted in 146 recommendations and statements, providing a comprehensive framework for the diagnosis and management of PARDS in pediatric critical care medicine.
Key Updates in PARDS Diagnosis: A Refined Approach
The PALICC-2 guidelines introduce significant refinements to the diagnostic criteria for PARDS, enhancing precision and applicability across diverse clinical settings. Key updates include:
Revised Definition and Severity Stratification
The updated PARDS definition emphasizes the importance of oxygenation indices (OI or OSI) for invasively ventilated patients, while PaO2/FiO2 or SpO2/FiO2 ratios are recommended for non-invasively ventilated patients. A critical change is the introduction of a severity stratification to be applied at least 4 hours after initial PARDS diagnosis. This delayed stratification allows for a more accurate assessment of disease severity and guides tailored management strategies. The severity is categorized based on ventilation type (invasive vs. non-invasive) and oxygenation impairment (mild/moderate vs. severe).
Introducing “Possible PARDS”
Recognizing the increasing use of nasal respiratory support methods like High-Flow Nasal Cannula (HFNC), PALICC-2 introduces the category of “possible PARDS.” This category is crucial for children on nasal interfaces and in resource-limited settings where comprehensive diagnostic tools may be unavailable. “Possible PARDS” allows clinicians to identify and manage patients who likely have PARDS even without full imaging capabilities, ensuring timely intervention.
Diagnostic Considerations for Special Populations
The guidelines provide specific considerations for diagnosing PARDS in children with cyanotic heart disease and chronic lung disease. These populations require nuanced diagnostic approaches, considering the underlying conditions that may complicate PARDS diagnosis. The updated definition helps differentiate PARDS from acute deteriorations related to their pre-existing conditions.
Table 4. Diagnosis of Pediatric Acute Respiratory Distress Syndrome (Definition Statement 1.1; Definition Statement 1.7.1)
| Age (DS 1.1) | Exclude patients with perinatal lung disease |
|---|---|
| Timing (DS 1.2) | Within 7 d of known clinical insult |
| Origin of edema (DS 1.3) | Not fully explained by cardiac failure or fluid overload |
| Chest imaging (DS 1.3) | New opacities (unilateral or bilateral) consistent with acute pulmonary parenchymal disease and which are not due primarily to atelectasis or pleural effusiona |
| Oxygenationb (DS 1.4.1) | IMV: OI ≥ 4 or OSI ≥ 5 |
| NIVc: Pao2/Fio2 ≤ 300 or Spo2/Fio2 ≤ 250 | |
| Stratification of PARDS severity: Apply ≥ 4 hr after initial diagnosis of PARDS (DS 1.4.4) | |
| IMV-PARDS: (DS 1.4.1) | Mild/moderate: OI 4-15 or OSI 5-11 |
| NIV-PARDSc (DS 1.4.2; DS 1.4.3) | Mild/moderate NIV-PARDS: Pao2/Fio2 > 100 or Spo2/Fio2 > 150 |
| Special populations d | |
| Cyanotic heart disease (DS 1.6.1; DS 1.6.2) | Above criteria, with acute deterioration in oxygenation not explained by cardiac disease |
| Chronic lung disease (DS 1.6.3; DS 1.6.4) | Above criteria, with acute deterioration in oxygenation from baseline |
aChildren in resource-limited settings where imaging is not available who otherwise meet PARDS criteria are considered to have possible PARDS.
bOxygenation should be measured at steady state and not during transient desaturation episodes. When Spo2 is used, ensure that Spo2 is ≤ 97%. OI = mean airway pressure (MAP) (cm H2O) × Fio2/Pao2 (mm Hg). OSI = MAP (cm H2O) × Fio2/Spo2.
cDiagnosis of PARDS on NIV (NIV-PARDS) requires full facemask interface with continuous airway positive pressure/positive end-expiratory pressure ≥ 5 cm H2O.
dStratification of PARDS severity does not apply to these populations.
Table 5. Diagnosis of Possible Pediatric Acute Respiratory Distress Syndrome and At-Risk for Pediatric Acute Respiratory Distress Syndrome (Definition Statement 1.5.3; Definition Statement 1.7.2; Definition Statement 11.2)
| Age | Exclude patients with perinatal lung disease |
|---|---|
| Timing | Within 7 d of known clinical insult |
| Origin of edema | Not fully explained by cardiac failure or fluid overload |
| Chest imaging (DS 1.5.2) | New opacities (unilateral or bilateral) consistent with acute pulmonary parenchymal disease and which are not due primarily to atelectasis or effusiona |
| Oxygenationb threshold to diagnose possible PARDS for children on nasal respiratory supportc (DS 1.5.1) | |
| Nasal continuous airway positive pressure/bilevel positive airway pressure or high-flow nasal cannula (≥ 1.5 L/kg/min or ≥ 30 L/min): Pao2/Fio2 ≤ 300 or Spo2/Fio2 ≤ 250 | |
| Oxygenationb threshold to diagnose at-risk for PARDS | |
| Any interface: Oxygen supplementationd to maintain Spo2 ≥ 88% but not meeting definition for PARDS or possible PARDS | |
| Special populations | |
| Cyanotic heart disease | Above criteria, with acute deterioration in oxygenation not explained by cardiac disease |
| Chronic lung disease | Above criteria, with acute deterioration in oxygenation from baseline |
aChildren in resource-limited environments where imaging is not available who otherwise meet possible PARDS criteria are considered to have possible PARDS.
bOxygenation should be measured at steady state and not during transient desaturation episodes. When Spo2 is used, ensure that Spo2 is ≤ 97%.
cChildren on nasal noninvasive ventilation (NIV) or high-flow nasal cannula are not eligible for PARDS but are considered to have possible PARDS when this oxygenation threshold is met.
dOxygen supplementation is defined as Fio2 > 21% on invasive mechanical ventilation; or Fio2 > 21% on NIV; or “oxygen flow” from a mask or cannula that exceeds these age-specific thresholds: ≥ 2 L/min (age < 1 yr); ≥ 3 L/min (age 1–2 yr); ≥ 4 L/min (age 2–5 yr); ≥ 5 L/min (age 5–10 yr); ≥ 6 L/min (age > 10 yr). For children on a mask or cannula, oxygen flow calculated as Fio2 × flow rate (L/min) (e.g., 6 L/min flow at 0.35 Fio2 = 2.1 L/min).
Management Principles: Evidence-Based Strategies for PARDS
The PALICC-2 guidelines offer a comprehensive update on the management of PARDS, emphasizing lung-protective ventilation strategies, judicious use of ancillary treatments, and the integration of informatics and data science to improve care delivery.
Ventilatory Support: Prioritizing Lung Protection
The cornerstone of PARDS management remains lung-protective ventilation. PALICC-2 reinforces key principles:
- Tidal Volume: Using lower tidal volumes (6-8 mL/kg) is suggested over higher volumes to minimize ventilator-induced lung injury. Even lower volumes (4-6 mL/kg) may be necessary to maintain safe plateau and driving pressures.
- Plateau Pressure and Driving Pressure: Maintaining plateau pressure ≤ 28 cm H2O and driving pressure ≤ 15 cm H2O is recommended to reduce lung strain. Higher plateau pressures (≤ 32 cm H2O) may be acceptable in cases of reduced chest wall compliance.
- PEEP Titration: Positive End-Expiratory Pressure (PEEP) should be carefully titrated based on oxygenation, oxygen delivery, hemodynamics, and respiratory system compliance, following established protocols such as the ARDS Network low PEEP/FiO2 table.
- Ventilation Bundles: The use of lung-protective ventilation bundles, incorporating these strategies and staff education, is recommended to improve adherence to best practices.
While specific ventilator modes are not prioritized, the guidelines emphasize optimizing lung volume when using High-Frequency Oscillatory Ventilation (HFOV). The routine use of recruitment maneuvers is not recommended due to insufficient evidence.
Table 6. Synthesis of the Second Pediatric Acute Lung Injury Consensus Conference Clinical Recommendations and Good Practice Statements Related to the Ventilatory Support, Respiratory Monitoring, and Pulmonary Ancillary Treatment
| Topic | Recommendation | Good Practice Statement |
|---|---|---|
| Noninvasive support | ||
| Use of O2/HFNC | Worsening acute respiratory failure → time-limited trial of NIV (7.1.1) | Humidification for HFNC (7.3.3) |
| In RLS, use of HFNC/CPAP vs O2 (7.5.1) | ||
| In RLS, use of CPAP vs HFNC when available (7.5.2) | ||
| Use of NIV (CPAP or bilevel positive airway pressure) | Worsening in 0–6 hr trial → ETT (7.1.2) | Close monitoring and trained staff (7.2) |
| Humidification (7.3.3), optimal interface for synchronization (7.3.1), monitoring for complications (7.3.2) | ||
| Addition of inspiratory support if synchronized (7.3.5) | ||
| Sedation during poor tolerance of NIV (7.3.4) | ||
| Invasive ventilation | ||
| ETT | ETT: use of cuffed tubes (3.11) | |
| Maintain unobstructed airway (4.4.1) | ||
| ETT suction: nonroutine use of instilled saline (4.4.3) | ||
| MV bundle | Use of lung protective ventilation bundle (3.5) | Daily assessment for extubation readiness test and spontaneous breathing trial (6.4.1) |
| Automated monitoring of compliance with Second Pediatric Acute Lung Injury Consensus Conference lung protective strategies (10.2) | In RLS, implement locally adapted protocols (PS 11.5) | |
| Regular training and education of all staff (PS 11.6) | ||
| MV type | Cannot recommend for or against HFOV (3.8.1) | If HFOV used: lung volume optimization strategy (3.8.2) |
| Monitoring | Continuous: respiratory rate, heart rate, Spo2 (6.1.1). Intermittent: noninvasive blood pressure (6.1.1) | |
| Monitor effort of breathing (6.2.5) | ||
| Continuous monitoring of co2 during MV (6.3.3) | ||
| Calculate and monitor dead space (6.3.4) | ||
| Vt | 6–8 mL/kg (3.2) | Scale Vt and Crs to body weight (6.1.2) |
| Use of 4–6 mL/kg if needed to stay below suggested PPlat and DP (3.2) | Continuously monitor Vt (6.2.1) using compensation for circuit compliance (6.2.2) | |
| PIP and PPlat | PPlat ≤ 28 cm H2O (3.3.1) | Monitor PIP and PPlat (6.2.3) |
| PPlat ≤ 32 cm H2O if reduced chest wall compliance (3.3.1) | ||
| DP limit | DP ≤ 15 cm H2O (3.3.2) | Monitor DP (6.2.3) |
| PEEP | Titration: to O2, O2-delivery, hemodynamics, and Crs (3.4.1) | Monitor intrinsic PEEP, flow- and pressure-time curves (6.2.4) |
| Level: at or above level on Acute Respiratory Distress Syndrome Network low PEEP/Fio2 Table (3.4.2) | Titration: attend to PPlat and DP (3.4.3) | |
| Spo2 target | Mild/moderate: 92–97% strategy (3.9.1) | Avoidance of Spo2 > 97% (3.9.3) |
| Severe: accept Spo2 < 92% | Severe: when Spo2 < 92%, monitor central venous saturation (3.9.2, 3.9.4) | |
| pH/Paco2 target | Accept pH ≥ 7.2 to remain within PPlat, DP, and Vt ranges during permissive hypercapnia (3.10.1) | Adjust frequency of pH, Paco2 measurement to PARDS severity and stage and to noninvasive co2 monitoring (6.3.2) |
| No routine use of bicarbonate supplementation (3.10.2) | ||
| Ancillary treatment | ||
| Prone positioning | Cannot recommend for or against prone positioning (4.3) | |
| Recruitment maneuver | Cannot recommend for or against recruitment maneuver (3.6) | |
| Inhaled NO | Use of inhaled nitric oxide in selected populations only (4.1) | |
| Surfactant | Against routine use of surfactant (4.2) | |
| Corticosteroids | Use of corticosteroids in selected populations only (4.6) |
Monitoring: Comprehensive and Continuous Assessment
Effective monitoring is crucial for guiding PARDS management and detecting complications. PALICC-2 emphasizes:
- Continuous Monitoring: Respiratory rate, heart rate, and SpO2 should be continuously monitored in all PARDS patients.
- Ventilator Parameters: Continuous monitoring of tidal volume, and regular monitoring of plateau pressure and driving pressure are recommended for invasively ventilated patients. Compensation for circuit compliance is essential for accurate tidal volume measurement.
- CO2 Monitoring: Continuous CO2 monitoring (end-tidal or transcutaneous) is advised to assess ventilation adequacy.
- Hemodynamic Monitoring: Hemodynamic monitoring, including fluid balance assessment and echocardiography in severe cases, is crucial to evaluate cardiac function and oxygen delivery.
Ancillary Treatments: Selective and Targeted Approaches
PALICC-2 advocates for a selective, rather than routine, approach to ancillary treatments for PARDS:
- Inhaled Nitric Oxide (iNO): Routine use is not recommended. However, iNO may be considered in specific phenotypes like pulmonary hypertension or severe right ventricular dysfunction, or as a rescue therapy.
- Surfactant: Routine surfactant therapy is not recommended, although selective use in specific populations may have a role, pending further research.
- Corticosteroids: Routine use is discouraged, but corticosteroids may be considered in PARDS due to specific etiologies like COVID-19, with careful risk-benefit assessment.
- Prone Positioning: While evidence is insufficient to routinely recommend prone positioning, it may be considered in patients with refractory hypoxemia.
Table 7. Synthesis of the Second Pediatric Acute Lung Injury Consensus Conference Clinical Recommendations and Good Practice Statements Related to Nonpulmonary Management
| Topic | Recommendation | Good Practice Statement |
|---|---|---|
| Diagnosis | ||
| Screening and monitoring | Use of electronic algorithms to help identify PARDS (10.1) | Policy statement: Healthcare organizations support for developing, implementing, and using electronic tools (10.3) |
| Risk stratification | Measure dead space to tidal volume ratio and/or end-tidal alveolar dead-space fraction (2.1), beside oxygenation-based stratification | Monitor Fio2, pulse oximeter oxygen saturation, Pao2, mean airway pressure, positive end-expiratory pressure (6.3.1) |
| Use of chest imaging (6.5.1) | ||
| Hemodynamic monitoring | Monitor to assess impact of MV on RV/LV (6.6.1) | |
| Arterial line for blood pressure and arterial blood gas in severe PARDS (6.6.4) | ||
| Perform cardiac ultrasound in severe PARDS or suspected RV/LV dysfunction (6.6.3) | ||
| ECMO | ||
| Failing response to treatment | Consider transfer to ECMO center (8.1.5) | |
| Evaluation | When lung protective strategies fail, and reversible cause. No strict criteria (8.1.1) | Structured evaluation by expert team (8.1.2) |
| Serial evaluations (8.1.3) | Education and competencies for ECMO clinicians (PS 8.2.1) | |
| Report data to Extracorporeal Life Support Organization (or equivalent) for benchmarking (PS 8.2.2) | ||
| Support type | Use of venovenous ECMO (8.1.4) | |
| Blood gas targets | Avoid hyperoxia (8.3.1a) | |
| Slow changes in Paco2 (8.3.1b) | ||
| MV | General lung protective strategy (8.3.2) | |
| Pain, Agitation, Neuromuscular Blockade, and Delirium in Critically Ill Pediatric Patients With Consideration of the ICU Environment and Early Mobility | ||
| Approach | Nonpharmacological multicomponent approaches (5.2.2; 5.7.1) | |
| Assessment | Use of scales (5.1.1) | |
| Daily assessment of activity and mobility goals (5.7.2) | ||
| Rehabilitation evaluation by 72 hr (5.7.3) | ||
| Daily assessment for delirium (5.2.1) | ||
| If treated ≥ 5 d assess for iatrogenic withdrawal syndrome (5.1.4) | ||
| Sedation | Titrate drugs for minimal, yet effective dose (5.1.2) | |
| Monitor and wean with goal-directed protocol (5.1.3) | ||
| NMBA | Use of NMBA, if protective ventilation is not achieved with sedation alone (5.3.1) | Monitor and titrate to goal-established (5.3.2) |
| Fluids | Optimize while preventing overload (5.5) | Monitor cumulative fluid balance (6.6.2) |
| Nutrition | Early start (≤ 48 hr) of enteral nutrition (5.4.1) | Nutrition plan (5.4.2) |
| Protein ≥ 1.5 g/kg/d (5.4.4) | EN monitoring with goal-directed protocol (5.4.3) | |
| Blood | No transfusion of pRBC for hemoglobin concentration ≥ 7 g/dL (5.6.2) | Use of pRBC for hemoglobin concentration < 5 g/dL (5.6.1) |
Non-Pulmonary Management: A Holistic Approach
Beyond respiratory support, PALICC-2 emphasizes a holistic approach to PARDS management, including:
- Sedation and Delirium: Implementing validated scales for pain, sedation, and delirium assessment is crucial. Goal-directed sedation protocols, minimizing sedation depth, and non-pharmacological delirium prevention strategies are recommended.
- Neuromuscular Blockade (NMB): NMB should be used judiciously, only when lung-protective ventilation cannot be achieved with sedation alone, and titrated to the desired effect.
- Nutrition: Early enteral nutrition (within 48 hours) and a protein-rich diet (≥ 1.5 g/kg/day) are recommended to support recovery.
- Fluid Management: Fluid therapy should be optimized to maintain organ perfusion while avoiding fluid overload.
- Rehabilitation: Early rehabilitation evaluation and mobilization are encouraged to mitigate long-term morbidities.
Table 8. Synthesis of the Second Pediatric Acute Lung Injury Consensus Conference Good Practice Statements Related to Follow-Up After Pediatric Acute Respiratory Distress Syndrome
| Assessment | Good Practice Statement |
|---|---|
| Initial approach | Primary care screening by 3 mo for post-PICU morbidities (9.1.1) |
| Stepwise addition of management, re-evaluations, referral to a specialist (9.1.2) | |
| Assessment of health-related quality of life, physical, neurocognitive, emotional, family, and social function | Evaluation within 3 mo of PICU discharge (9.3.1) |
| Additional pre-school (4–6 yr) assessment if pediatric acute respiratory distress syndrome during infancy (9.3.2) | |
| Referral for specialist help when deficits identified (9.3.3) | |
| Post-extracorporeal membrane oxygenation, short- and long-term neurodevelopment and physical function (8.5) | |
| Pulmonary assessment | Screen by 3 mo post-PICU discharge for pulmonary function abnormalities (9.2.1) |
| With spirometry in patients of sufficient age and capabilities (9.2.2) | |
| Referral to pediatrician or pediatric pulmonologist when pulmonary function deficits identified (9.2.3) |
Extracorporeal Support: ECMO Considerations
Extracorporeal Membrane Oxygenation (ECMO) remains a critical rescue therapy for severe PARDS. PALICC-2 provides guidance on ECMO use:
- Evaluation for ECMO: Patients with potentially reversible severe PARDS, failing lung-protective ventilation, should be evaluated for ECMO by an expert team. Serial evaluations are recommended to guide ECMO decisions.
- VV-ECMO Preference: Venovenous (VV) ECMO is preferred over venoarterial (VA) ECMO in PARDS patients with adequate cardiac function.
- ECMO Center Transfer: Transfer to an ECMO center should be considered for patients who are not stabilizing with conventional therapies.
- Management during ECMO: Lung-protective ventilation principles should continue during ECMO support. Hyperoxia and rapid PaCO2 changes should be avoided.
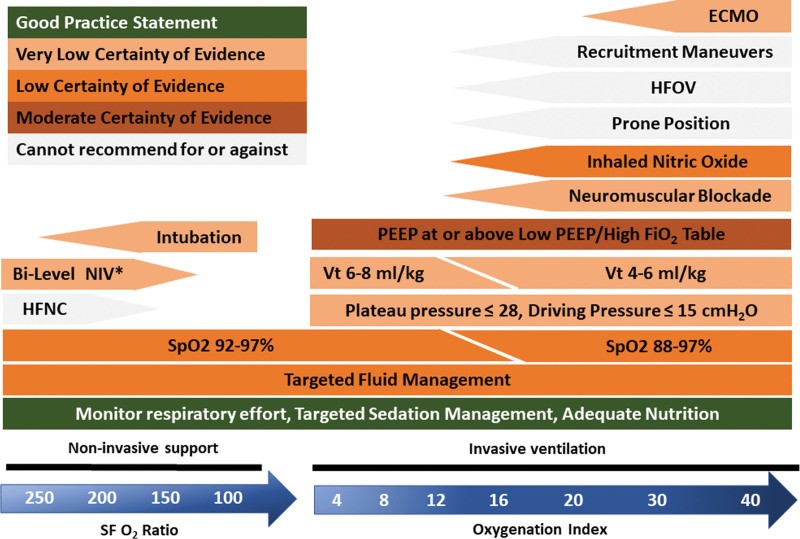
Figure 1.
Schematic summary of key therapies or management strategies in pediatric acute respiratory distress syndrome based on the Second Pediatric Acute Lung Injury Consensus Conference recommendations and statements. *Continuous positive airway pressure if unable to tolerate bilevel noninvasive ventilation (NIV). ECMO = extracorporeal membrane oxygenation, HFNC = high-flow nasal cannula, HFOV = high-frequency oscillatory ventilation, PEEP = positive end-expiratory pressure, SFo2 ratio = oxygen saturation/Fio2 ratio, Spo2 = pulse oximeter oxygen saturation, Vt = tidal volume.
Clinical Informatics and Resource-Limited Settings: Bridging Gaps
PALICC-2 addresses crucial aspects of implementation, particularly in clinical informatics and resource-limited settings:
- Clinical Informatics: The guidelines recommend implementing electronic tools for automatic PARDS screening and monitoring compliance with lung-protective ventilation strategies. Healthcare organizations are encouraged to support the development and use of these tools.
- Resource-Limited Settings (RLS): PALICC-2 provides specific adaptations for RLS, acknowledging limitations in resources and expertise. Recommendations emphasize the use of CPAP and HFNC over standard oxygen therapy when available. “Possible PARDS” definition is particularly relevant in RLS where imaging may be limited. Locally adapted protocols and regular staff training are crucial in these settings.
Table 9. Synthesis of the Second Pediatric Acute Lung Injury Consensus Conference Research Recommendations
| Activity | Research Neededa |
|---|---|
| Screening, diagnosis, and monitoring | |
| Diagnosis | Adult vs pediatric age-group comparison in pediatric acute respiratory distress syndrome progression and outcome (2.2) |
| Trajectory of illness using standardized minimal datasets and operational definitions (2.4.1) | |
| Diagnostic utility of Second Pediatric Acute Lung Injury Consensus Conference definition in RLS (11.3) | |
| Collaborative research networks to include RLS sites (11.4) | |
| Biomarker | Biomarker and genetic insight into pathophysiology (2.3.1) |
| Molecular phenotype and endotype studies (2.3.2) | |
| Data science | Collaborative data science (10.4) |
| Development of generalizable electronic tools (10.5) | |
| MV and cardiovascular system monitoring | |
| Endotracheal tube | Standardize spontaneous breathing trial and extubation readiness test in studies (6.4.2) |
| MV bundle | Closed vs open suctioning (4.4.2); methods and populations for airway clearance (4.5) |
| MV type | Studies of ventilator modes (3.1), studies of high-flow nasal cannula (7.4), studies needed inclusive of RLS (11.8) |
| Vt | Use of protocols and definitions in studies measuring Vt, peak inspiratory pressure, plateau pressure, positive end-expiratory pressure, mean airway pressure (2.4.2); use of mechanical power calculations (3.7); use of monitoring loops, compliance, resistance, strain-stress, etc. (6.2.6) |
| Hemodynamic | Studies of devices, approach, and tests of hemodynamics (6.6.5); studies of routine CT, lung ultrasound, electrical impedance tomography (6.5.2) |
| Other lung | |
| Other drugs | Studies of many agents (4.7; 11.9) |
| ECMO | |
| ECMO type | Studies of extracorporeal co2 removal (8.4) |
| Pain, Agitation, Neuromuscular Blockade, and Delirium in Critically Ill Pediatric Patients With Consideration of the ICU Environment and Early Mobility | |
| Assessment | Medications for prevention and/or treatment of delirium (5.2.3) |
| General approach | |
| Fluids | Validated measure of nonpulmonary organ system dysfunction (2.4.3) |
| Nutrition | Reporting of nutrition strategy in research studies (5.4.5) |
| Blood | Studies of packed RBC and alternatives (5.6.3) |
| Post-PICU follow-up | |
| Initial | Assessment of pre-PICU baseline state if follow-up is anticipated (9.4.1); practices to optimize follow-up (9.4.5) |
| Assessment | Postdischarge endpoints (9.4.2); relationship between short- and longer-term outcomes (9.4.3); factors affecting trajectory of recovery (9.4.4) |
aThe premise of research needed statements (which is not referenced in the Table) is that everything in the Research column needs a dimension that is relevant to RLS, as well as collaboration between the spectrum of resourced settings.
Conclusion: Charting the Future of Pediatric Critical Care in PARDS
The PALICC-2 guidelines represent a significant advancement in the principles of diagnosis and management of PARDS. By incorporating new evidence, refining diagnostic criteria, and addressing challenges in diverse settings, these guidelines offer a robust framework for improving outcomes in pediatric critical care medicine. While acknowledging the limitations of current evidence, PALICC-2 highlights critical areas for future research, urging collaborative efforts to strengthen the evidence base and further refine clinical practice in the management of PARDS. These guidelines serve as an invaluable resource for healthcare professionals seeking to implement best practices in the care of children with PARDS, ultimately aiming to reduce mortality and improve long-term outcomes in this vulnerable population.
For a comprehensive understanding of the evidence and rationale behind each recommendation, readers are encouraged to consult the full PALICC-2 guideline documents and supplementary materials. These resources provide detailed insights into the critical care medicine principles that underpin these updated recommendations for PARDS diagnosis and management.