Depression stands as a prevalent mental health disorder affecting millions of adults across the United States. Recognized for its significant impact on individual well-being and societal productivity, the imperative for universal depression screening and effective treatment protocols has never been more critical. Leading health organizations, such as the U.S. Preventive Services Task Force (USPSTF), advocate for routine depression screening in primary care settings, coupled with evidence-based interventions. This article delves into the crucial aspects of Depression Screening And Diagnosis, particularly within primary care, highlighting innovative approaches and their impact on patient outcomes.
The Critical Need for Depression Screening in Primary Care
Depression is not just a fleeting feeling of sadness; it is a serious medical condition. Statistics reveal that a significant portion of the adult population in the U.S. – approximately 17.3 million individuals, or 7.1% of all adults – experience at least one major depressive episode. This condition is not only widespread but also profoundly disabling. The economic burden of depression is staggering, estimated at around $210 billion annually in medical expenses and lost productivity. Beyond the financial costs, untreated depression inflicts immense emotional distress, diminishes productivity, erodes relationships, and elevates the risk of co-occurring health problems. The intricate link between mental and physical health underscores the urgency of addressing depression, especially considering its frequent presence alongside serious chronic illnesses.
Recognizing and treating depression early in primary care settings is paramount. Extensive research supports the effectiveness of depression screening and treatment within these settings, enabling timely intervention and improved patient outcomes. The USPSTF guidelines strongly recommend depression screening for all adults aged 18 and older in primary care, emphasizing the integration of screening with robust support systems and evidence-based treatment protocols. These recommendations are further reinforced by endorsements from organizations like the American Academy of Family Physicians and are increasingly incentivized by value-based payment models from the Centers for Medicare & Medicaid Services (CMS). These models, particularly within Accountable Care Organizations, link financial benefits to performance benchmarks in depression screening, follow-up care, and remission rates.
The landmark Sequenced Treatment Alternatives to Relieve Depression (STAR*D) trial, a large-scale study conducted over five years across diverse primary care and specialty sites, provided compelling evidence for the effectiveness of depression care in primary settings. This trial demonstrated that treating depression in primary care could achieve outcomes comparable to those in specialty mental health care. Despite these compelling findings and national guidelines advocating for increased screening and treatment, a significant gap persists. Alarmingly, over 50% of individuals with depression in primary care remain undiagnosed and undertreated. National data from 2005 to 2015 revealed a strikingly low depression screening rate of just 1.4%, with only 2.2% of primary care physician visits including depression screening for adult patients. This persistent gap underscores the urgent need for improved strategies and tools to enhance depression screening and diagnosis in primary care.
Tools for Effective Depression Screening and Diagnosis
Efforts to improve depression identification in primary care have increasingly focused on the use of standardized screening tools. Among these, the Patient Health Questionnaire (PHQ)-2 and PHQ-9 are the most widely used and validated instruments for adult depression screening. These tools are recognized for their clinical utility and diagnostic accuracy in primary care settings.
The PHQ-2 serves as an efficient initial screening tool. It comprises just two questions assessing the frequency of depressed mood and anhedonia (loss of interest or pleasure). Each question is scored from 0 (“not at all”) to 3 (“nearly every day”). A PHQ-2 score of 3 or higher is considered a positive screen, indicating the need for further evaluation with the more comprehensive PHQ-9. A positive PHQ-2 screen demonstrates a high degree of sensitivity (83%) and specificity (92%) in detecting major depression, making it a reliable initial filter.
The PHQ-9 expands on the PHQ-2, incorporating nine items directly aligned with the DSM-5 (Diagnostic and Statistical Manual of Mental Disorders, fifth edition) criteria for major depressive disorder. This tool delves deeper into the various symptom dimensions of depression. Scores on the PHQ-9 are used to categorize the severity of depression: scores of 5, 10, 15, and 20 correspond to mild, moderate, moderately severe, and severe depression, respectively. This detailed scoring allows clinicians to not only identify depression but also to gauge its severity, informing treatment decisions.
Meta-analyses of extensive data involving thousands of primary care patients have consistently affirmed the effectiveness, validity, and reliability of both the PHQ-2 and PHQ-9 in detecting depression in primary care settings. Furthermore, research indicates that the most successful and sustained outcomes in depression care are achieved when screening is integrated with comprehensive systems of care. These systems typically include standardized screening tools, clearly defined treatment protocols, and established referral pathways to specialty mental health services.
Measurement-Based Care (MBC) represents a comprehensive approach to depression management. MBC guidelines incorporate validated screening instruments, standardized symptom measurements, monitoring of treatment effects and patient adherence, and evidence-based decision support tools. These components empower primary care physicians in diagnosis, treatment planning, follow-up, and referral processes. Programs like VitalSign6, developed by The University of Texas Southwestern Medical Center for Depression Research and Clinical Care (CDRCC), exemplify MBC in action. VitalSign6 and similar programs leverage technology to seamlessly integrate screening and MBC into the existing primary care workflow, often through application programming interfaces that enable interoperability with electronic health records (EHRs). This integration is crucial for making depression screening and diagnosis a routine and efficient part of primary care practice.
Implementing VitalSign6: A Quality Improvement Project
To address the persistent gap in depression screening and management, a quality improvement project was undertaken to implement VitalSign6, an evidence-based MBC program, within the Department of Family and Community Medicine at the University of Texas Southwestern Medical Center (UTSW). The project aimed to improve the identification and management of depression among adult patients aged 18 and older. The specific goals included:
- Screening at least 75% of unique patients for depression using the PHQ-2.
- Screening at least 85% of patients who screened positive on the PHQ-2 with the PHQ-9.
- Administering MBC to at least 75% of patients diagnosed with depression and scheduling follow-up appointments within 4 to 6 weeks.
- Monitoring patient responses, treatment effects, and remission using the VitalSign6 MBC system.
- Assessing the satisfaction of physicians and staff with VitalSign6 and its integration with the EHR.
These targets were adapted from the VitalSign6 Primary Care First Model for depression screening and MBC, reflecting evidence-based benchmarks for effective implementation.
The project employed a pre-post intervention design to evaluate the effectiveness of VitalSign6 in improving screening rates, clinical outcomes, and provider/staff satisfaction. The study was conducted at the UTSW Department of Family and Community Medicine, and it received approval from the Institutional Review Board, with a waiver for individual patient consent. The clinic setting included a residency program and served a diverse patient population, with a monthly volume of approximately 585 visits. The project was initiated in February 2019, with initial screening conducted through May 31, and patients diagnosed with depression and receiving MBC were followed for 14 weeks thereafter.
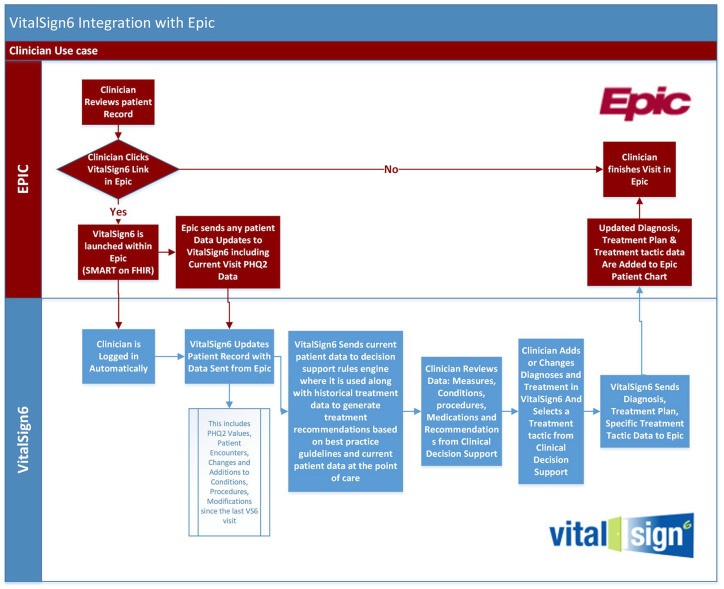
VitalSign6, the core innovation, is a web-based MBC program designed to provide primary care providers with tools for standardized assessment of symptom severity, antidepressant side effects, adherence, and algorithm-based medication selection with decision support. This system enables providers to tailor treatment plans to individual patient needs, selecting appropriate medications, dosages, referrals, and follow-up strategies in alignment with evidence-based guidelines.
The VitalSign6 software utilizes a user-friendly application accessible on iPads, available in both English and Spanish. Patients complete self-reported depression questionnaires during check-in, seamlessly integrated into the clinic’s existing workflow. Providers can then review results in real-time, document diagnoses, and initiate personalized treatment planning with clinical decision support. For patients screening negative, VitalSign6 automatically triggers rescreening in one year, ensuring ongoing monitoring.
A key element of the project was addressing the challenge of EHR interoperability. To facilitate seamless integration within the clinic’s existing system, a UTSW technology team developed an application programming interface using Fast Healthcare Interoperability Resources (FHIR) and Substitutable Medical Apps Reusable Technology (SMART). This enabled real-time integration of VitalSign6 directly into the Epic EHR database. This integration allowed physicians to launch VitalSign6 contextually within Epic, review patient measures and clinical decision support data, and seamlessly transfer treatment decisions back into the patient’s EHR chart. This technological advancement was crucial for minimizing workflow disruption and maximizing the utility of VitalSign6.
Prior to implementation, comprehensive training sessions were conducted for 25 physicians and residents, and 20 clinic staff. These two-hour in-person sessions covered depression education, screening protocols, symptom measurement, diagnosis, best treatment practices, medication management, and referral procedures. VitalSign6 expert trainers remained on-site for two weeks following the training to provide immediate support and address any implementation challenges.
Results and Outcomes: Impact of VitalSign6
The implementation of VitalSign6 demonstrated significant improvements in depression screening and management within the primary care clinic. During the study period, 1200 unique adult patients were seen, with a diverse demographic profile. Remarkably, 95.4% of these patients (n=1145) were screened for depression using the PHQ-2, exceeding the project’s initial aim of 75%. Of those screened, 18.2% (n=208) screened positive on the PHQ-2. Among patients with positive PHQ-2 screens, 89.4% (n=186) proceeded to PHQ-9 screening. Technical issues affected data recording for a small percentage (10.5%) of positive PHQ-2 screens and associated PHQ-9 assessments.
Furthermore, a significant number of patients (18.6%, n=214) who initially screened negative on the PHQ-2 but had a documented history of depression or antidepressant medication in their EHR also underwent PHQ-9 screening, triggered by a Best Practice Advisor (BPA) alert within the system. Additionally, physicians proactively requested PHQ-9 screening for another 7.1% (n=81) of patients with negative PHQ-2 scores and no prior history of depression, highlighting the increased awareness and proactive approach to depression assessment.
In total, 481 patients were screened with the PHQ-9. Of these, 62.8% (n=302) screened positive (score >4), and 89.4% of those who screened positive (n=270) were subsequently diagnosed with a depressive disorder. Among those diagnosed, the vast majority (87.4%, n=236) initiated MBC with personalized treatment plans, while 10% (n=27) were referred to specialty care, and a small percentage (2.6%, n=7) declined treatment.
Follow-up care was a crucial component of the project. Patients diagnosed with depression and started on personalized treatment plans were scheduled for follow-up visits within 4 to 6 weeks. Among the 236 patients receiving MBC, the majority (59.7%, n=141) received pharmacological treatment, while 8.1% (n=19) received non-pharmacological treatment, and 31.3% (n=74) were recommended for symptomatic monitoring and rescreening. Patients were followed for a total of 14 weeks to assess treatment progress and outcomes.
Analysis of PHQ-9 scores revealed a statistically significant decrease in self-reported depression symptoms from baseline to follow-up. Among 65 patients with complete data, the mean PHQ-9 score decreased from 14.89 at baseline to 9.58 at follow-up. Notably, 23.1% (n=15/65) of these patients achieved remission, defined as a PHQ-9 score <5, within the 14-week follow-up period.
Physician and staff satisfaction surveys provided valuable qualitative feedback. While physicians reported mixed experiences, with some indicating a decrease in their perceived importance of depression screening and MBC post-implementation (potentially due to workflow disruptions), staff reported a significant increase in their comfort level discussing mental health with patients. Open-ended survey questions highlighted workflow slowdown and technical issues with EHR interoperability as key challenges associated with VitalSign6 implementation. However, both physicians and staff acknowledged the valuable aspects of VitalSign6, including improved mental health awareness, enhanced depression screening, and the potential of EHR integration.
Discussion and Conclusion: Advancing Depression Care in Primary Settings
The findings of this quality improvement project underscore the effectiveness of implementing a measurement-based care program like VitalSign6 to enhance depression screening and management in primary care. The project demonstrated a notable improvement in patient remission rates, increasing from a baseline of 0% to 23.1% post-intervention. This improvement is attributed to the integrated approach of screening, diagnosis, and treatment guided by MBC principles and evidence-based clinical decision support tools, all facilitated by EHR interoperability. The results suggest that EHR systems equipped with CDS capabilities can be a powerful mechanism for increasing depression screening and improving remission rates in primary care settings.
The project successfully improved depression screening rates to over 95%, significantly exceeding the initial target. The two-step screening process using PHQ-2 and PHQ-9, combined with real-time EHR integration, proved to be efficient and effective. Physicians and staff acknowledged the positive impact of VitalSign6 on raising mental health awareness and improving screening practices within the clinic.
The study’s findings support the concept of viewing depression care as a triage issue rather than solely a mental health access issue. VitalSign6 equipped primary care providers with the necessary tools and CDS to manage the majority (87.4%) of diagnosed patients within the primary care setting, with only a small proportion requiring referral to specialty care. This approach highlights the potential of primary care to effectively address a significant portion of depression cases, particularly when supported by integrated MBC programs and technological tools.
Despite the successes, the project also encountered challenges. Workflow disruptions and technical issues related to EHR interoperability were identified as areas for improvement. Physician feedback emphasized the need for streamlining workflow and enhancing the seamlessness of data exchange between VitalSign6 and the EHR. However, significant progress was observed in staff comfort levels regarding mental health discussions with patients, likely attributable to the comprehensive training and on-site support provided during the project.
Limitations of this project include its single-site nature within an academic medical center residency program, which may limit generalizability. The project primarily focused on PHQ-2 and PHQ-9 measures and did not comprehensively analyze other features of VitalSign6, such as anxiety assessment or treatment side effect monitoring. Furthermore, a high patient attrition rate (70.2% not returning for follow-up) necessitates further investigation into strategies to improve patient retention and engagement in depression care.
In conclusion, the implementation of VitalSign6 in a primary care setting demonstrates its effectiveness as a valuable tool for improving depression identification, treatment, and referral management. While further refinement is needed to optimize workflow and address technical challenges, the project’s results contribute to the growing body of evidence supporting MBC programs like VitalSign6 as effective adjuncts in enhancing depression care within primary care. Future efforts should focus on developing integrated, multidisciplinary care team models, streamlining workflows, and establishing sustainable policies to further improve depression care delivery in primary settings.