The Food and Drug Administration (FDA) has recently granted approval for a new application of Wegovy (semaglutide), a highly successful medication for obesity. This new approval is significant as it allows Wegovy to be used to decrease the risk of serious heart issues, such as heart attacks and strokes, in individuals with cardiovascular disease who are also overweight or obese. Wegovy belongs to the GLP-1 (glucagon-like peptide-1) agonists class, initially designed for type 2 diabetes treatment but also proven very effective in managing obesity. This FDA decision is a crucial step towards Medicare coverage for Wegovy and potentially broader insurance coverage. Currently, Medicare is legally restricted from covering weight loss drugs when prescribed solely for obesity. However, semaglutide, under the brand name Ozempic, is covered by Medicare for diabetes treatment.
How Does the FDA’s Decision Impact Medicare Coverage for Wegovy?
This FDA approval effectively paves the way for Medicare to cover Wegovy, initially approved as an anti-obesity medication. Following the FDA announcement, the Centers for Medicare & Medicaid Services (CMS) released a memo indicating that Medicare Part D plans now have the option to include Wegovy in their drug formularies. This is because Wegovy now has a medically accepted use that is not specifically excluded from Medicare coverage. As Wegovy is an injectable medication for self-administration, coverage will fall under Medicare Part D, the outpatient prescription drug benefit offered through private plans, rather than Part B, which covers drugs administered by physicians.
Who in Medicare Could Benefit from Wegovy Coverage for this New Indication?
The new approved use for Wegovy targets individuals with existing cardiovascular disease – such as a history of heart attack, stroke, or peripheral arterial disease – who are also classified as obese or overweight. KFF analysis of 2020 Medicare data estimates that approximately 7% of Medicare beneficiaries, or 3.6 million people, had both established cardiovascular disease and obesity or overweight. These individuals could potentially be eligible for Medicare coverage of Wegovy based on this new indication (Figure 1). It’s important to note that this number might be even higher with more recent data. This group of 3.6 million represents just over a quarter (26%) of the 13.7 million Medicare beneficiaries diagnosed with obesity or overweight in 2020. This suggests that the FDA’s approval could make Wegovy accessible to about 1 in 4 Medicare beneficiaries struggling with obesity or overweight.
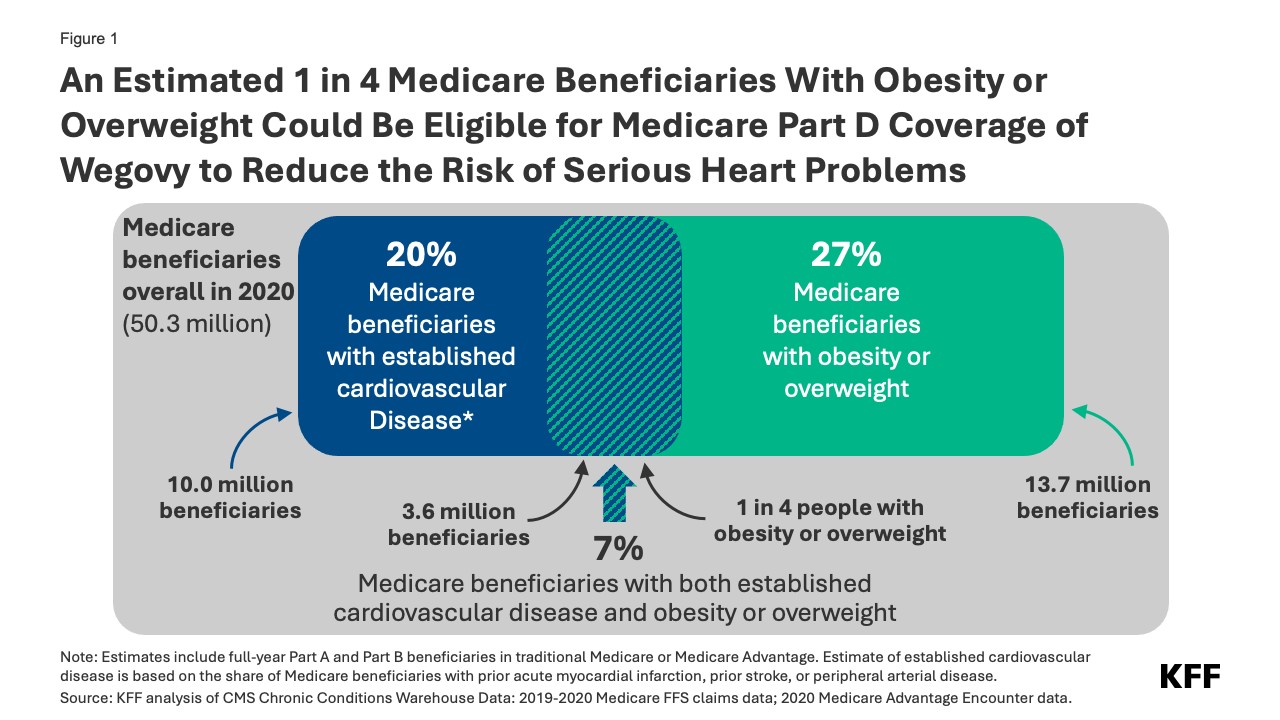 Figure 1: Estimated Medicare Beneficiaries Eligible for Wegovy Coverage
Figure 1: Estimated Medicare Beneficiaries Eligible for Wegovy Coverage
Figure 1: A chart showing that an estimated one in four Medicare beneficiaries who are obese or overweight could be eligible for Medicare Part D coverage of Wegovy to reduce the risk of serious heart problems.
Among these 3.6 million beneficiaries, 1.9 million also had diabetes (excluding Type 1). This subgroup may have already been eligible for Medicare coverage of GLP-1 medications as diabetes treatments before Wegovy’s new approval.
However, not all eligible individuals will necessarily choose to take Wegovy. Some may be concerned about potential side effects and adverse reactions. Cost is another significant factor. With a list price of around $1,300 per month (before rebates and discounts), Wegovy might be classified as a specialty tier drug under Part D. This could mean beneficiaries paying coinsurance might face monthly costs between $325 and $430, before reaching the annual out-of-pocket spending cap set by the Inflation Reduction Act – approximately $3,300 in 2024 for brand-name drugs, and $2,000 in 2025. Even with the cap, these costs can be prohibitive for many Medicare recipients with limited incomes. Ultimately, the actual out-of-pocket expenses will depend on the specific Part D plan and Wegovy’s placement within its formulary tiers.
Furthermore, access to Wegovy might be restricted by Part D plans through prior authorization and step therapy requirements aimed at cost management and ensuring appropriate use. These measures could limit the uptake of Wegovy, even among the intended patient population.
When Will Medicare Part D Plans Start Covering Wegovy?
Some Part D plans have already announced intentions to cover Wegovy this year, but the extent of coverage in 2024 remains uncertain. While Medicare drug plans can add new drugs to their formularies during the year to reflect new approvals, they are not obligated to cover every new medication. Part D plans must cover at least two drugs in each therapeutic category and essentially all drugs within six protected classes. However, given Wegovy’s high price and potentially large number of eligible patients, many Part D plans might hesitate to expand coverage immediately, as they cannot adjust premiums mid-year to accommodate increased costs. Therefore, broader coverage may be more likely in 2025.
What is the Potential Impact of Expanded Wegovy Coverage on Medicare Spending?
The financial impact of Wegovy coverage on Medicare will depend on several factors, including the number of Part D plans offering coverage, the extent of utilization controls like prior authorization, the number of eligible individuals who use the drug, and negotiated prices. For instance, if plans secure a 50% rebate on the $1,300 monthly list price ($15,600 annually), the net annual cost per person could be around $7,800. If 10% of the target population (approximately 360,000 people) uses Wegovy for a year, this could lead to an additional $2.8 billion in net Medicare Part D spending for this single drug in one year.
Semaglutide could be considered for Medicare drug price negotiation as early as 2025, based on Ozempic’s initial FDA approval in late 2017. For small-molecule drugs like semaglutide, at least seven years must pass from FDA approval to be eligible for negotiation. If selected next year, negotiated prices could be effective starting in 2027. This could potentially reduce both Medicare and patient out-of-pocket costs for semaglutide products, including Wegovy, Ozempic, and Rybelsus, the oral form of semaglutide for type 2 diabetes. In 2022, Medicare’s gross spending on Ozempic alone ranked it sixth among the top-selling Part D drugs, with annual gross spending of $4.6 billion, according to KFF analysis. This figure doesn’t account for rebates, which Medicare actuaries estimated to average 31.5% overall in 2022, but may be as high as 69% for Ozempic according to some estimates.
What Does This Mean for Medicare Coverage of Anti-Obesity Medications in General?
Currently, Medicare law still excludes coverage for GLP-1 medications used solely for obesity. However, the FDA’s recent decision marks a turning point for broader Medicare coverage of GLP-1s. Wegovy can now be prescribed to reduce heart attack and stroke risks in individuals with cardiovascular disease and obesity or overweight, broadening its use beyond just an anti-obesity drug. Further pathways for Medicare coverage might emerge if these drugs gain FDA approval for other health conditions. For example, Eli Lilly recently announced positive clinical trial results for its GLP-1 drug, Zepbound (tirzepatide), showing benefits in reducing sleep apnea severity in overweight and obese individuals. Lilly intends to seek FDA approval for this use, which, if granted, would make it the first pharmaceutical treatment for sleep apnea on the market.
Increased access to GLP-1s for Medicare beneficiaries with obesity or overweight, based on these expanded approvals, could potentially lower the estimated cost of proposed legislation to remove the legal prohibition on Medicare coverage for anti-obesity drugs. The Congressional Budget Office (CBO) would likely factor in the costs of coverage for these other approved uses when estimating the financial impact of such legislation. This means the additional cost of changing the law to allow Medicare coverage for anti-obesity drugs could be less than previously anticipated. Ultimately, the extent of Medicare Part D coverage for GLP-1s could significantly impact individuals with obesity and Medicare expenditure.
Juliette Cubanski, Tricia Neuman, and Nolan Sroczynski are with KFF. Anthony Damico is an independent consultant.
This work was supported in part by Arnold Ventures. KFF maintains full editorial control over all of its policy analysis, polling, and journalism activities.
| ###### Methods |
|---|
| The estimate of Medicare beneficiaries who could be eligible for Medicare coverage of Wegovy for cardiovascular disease is based on individual-level claims and encounter data for beneficiaries in traditional Medicare and Medicare Advantage from the Chronic Conditions Data Warehouse (CCW). For beneficiaries in traditional Medicare, coding of individual-level fee-for-service (FFS) claims data matched the following chronic condition flags in the 2020 Medicare Beneficiary Summary File 30 CCW Chronic Conditions and Other Chronic or Potentially Disabling Conditions segments: AMI_EVER, STROKE_TIA_EVER, and OBESITY. In addition to obesity, beneficiaries were coded with overweight if the following ICD-10 codes were identified in the claims with the same requirements as the CCW OBESITY flag: E66.3, Z68.25, Z68.26, Z68.27, Z68.28. Z68.29. To identify beneficiaries with peripheral arterial disease (PAD), we used ICD-9 diagnosis codes for PAD identified by either Hirsh et al (August 2008) or Jaff et al (July 2010) in their analyses of peripheral arterial disease among Medicare beneficiaries; these studies are two of three references cited by CCW in the Other Chronic Conditions Algorithms Reference List for peripheral vascular disease. We used the ICD10Data website to convert the ICD-9 codes used in the Hirsch and Jaff studies to corresponding ICD-10 codes for our analysis based on the 2020 data (ICD-9 codes were replaced by ICD-10 codes in 2015). Beneficiaries who were coded with obesity or overweight and either a prior heart attack (AMI_EVER), prior stroke (STROKE_TIA_EVER), or peripheral arterial disease were coded as being eligible for the new use of Wegovy. Among this group, beneficiaries who were flagged as having diabetes (not including Type 1 Diabetes Mellitus) based on ICD-10 codes and using the same requirements as the CCW DIABETES flag, were identified as being eligible for GLP-1s approved for use as diabetes treatments. For Medicare Advantage enrollees, the ICD-10 codes for the CCW-developed algorithms for AMI, stroke, obesity, and diabetes (not including Type 1), plus ICD-10 codes specified above for overweight and peripheral arterial disease, were used to identify whether enrollees were eligible for the new use of Wegovy, based on 2020 encounter data and utilizing a within-year lookback period for all conditions (rather than ever, or in some cases a 2-year lookback that is used for traditional Medicare enrollees). Earlier years of data to enable a longer lookback period were not available for this analysis. Among the factors contributing to imprecision in the overall estimate: – Medicare Advantage encounter data can be incomplete, which means the estimate may be too low if data are lacking on enrollees who would meet the clinical criteria for use. – Medicare Advantage plans have an incentive to code medical conditions in such a way that makes enrollees appear sicker than they would if they were in traditional Medicare, which means the estimate may be too high. – Using within-year lookback in the encounter data for Medicare Advantage enrollees means the estimate may be lower than if the same typically longer lookback period for traditional Medicare beneficiaries was used. – Medicare Part D plans may use more or less stringent criteria than the diagnosis coding criteria used in this analysis to determine whether individual enrollees are eligible for coverage of Wegovy. It is not possible to measure the degree of uncertainty associated with these different factors. |