Case Presentation
A 37-year-old woman visits urgent care complaining of right leg swelling and pain that has persisted for three days. She reports no history of trauma preceding the onset of symptoms. She denies any similar episodes in the past. Despite using ibuprofen, applying warm compresses, and massaging the affected area, her symptoms have not improved. She experiences pain when walking. Her medical history is notable for hypertension, managed with lisinopril. She denies tobacco or illicit drug use and consumes alcohol only on holidays. She maintains a physically active lifestyle.
Physical examination reveals warmth and erythema in her right calf. Palpation of the posterior calf elicits pain. Nonpitting edema is observed, and the right calf circumference is 1 cm greater than the left. The remainder of her physical exam is unremarkable. Her vital signs are within normal limits. She exhibits discomfort while ambulating but is not in acute distress.
Introduction to Deep Vein Thrombosis (DVT) in Urgent Care
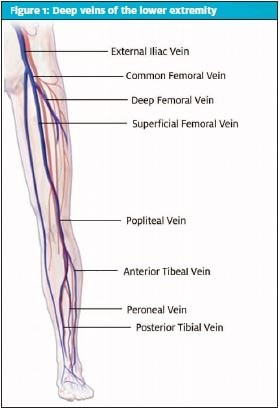
Deep vein thrombosis (DVT), a condition characterized by blood clot formation in deep veins, most commonly in the lower extremities, is a significant vascular concern frequently encountered in urgent care settings. [Figure 1] illustrates the potential locations of DVT within the venous system. The American Heart Association identifies DVT as the third most prevalent vascular diagnosis, following myocardial infarction and stroke, affecting an estimated 300,000 to 600,000 individuals in the United States annually. While upper extremity DVT can occur, the majority involve the deep veins of the legs, categorized as proximal or distal to the knee based on anatomical location. A critical complication of DVT is pulmonary embolism (PE), which arises when a thrombus detaches and travels through the venous system to the pulmonary arteries. The risk of DVT significantly escalates with age, increasing by 60% in individuals over 70. Alarmingly, approximately half of untreated proximal DVT cases progress to symptomatic PE within three months, with 25% of these resulting in sudden death. Venous ultrasound is the gold standard for DVT diagnosis. However, access to this diagnostic modality is often limited in urgent care and primary care environments. Consequently, diagnosis heavily depends on patient history, physical examination, and risk stratification tools to guide timely and appropriate testing. The Well’s score for DVT (Table 1) is a crucial risk assessment tool in this context. Distinguishing between provoked and unprovoked DVT, and first-time versus recurrent DVT is essential as these factors significantly influence management strategies.
DVT Risk Factors: Identifying Patients at Risk in Urgent Care
Numerous risk factors, both modifiable and nonmodifiable, contribute to the development of DVT. Age and genetics play significant roles, while acquired conditions and lifestyle choices also substantially impact DVT risk. Understanding these factors is vital for effective risk assessment in the urgent care setting.
Nonmodifiable Risk Factors
Age is a primary nonmodifiable risk factor. In women, the highest DVT risk occurs before age 50, while in men, it is more prevalent after 65. Genetic predisposition also plays a crucial role. Inherited thrombophilias, such as Factor V Leiden mutation, prothrombin gene polymorphism, protein C and S deficiency, non-O blood groups, and methylenetetrahydrofolate reductase (MTHFR) gene polymorphism, increase DVT susceptibility. The risk is highest in individuals homozygous for these conditions. Protein C and S deficiencies are particularly potent genetic risk factors, present in roughly 15% of DVT patients under 45, and can elevate DVT risk tenfold or more. Factor V Leiden mutation, the most common inherited thrombophilia, is associated with a lifetime DVT incidence of approximately 6.3%. Thrombophilia should be suspected in patients with a history of recurrent DVT or miscarriages, and a family history of thrombophilia in first-degree relatives is an important consideration during history taking.
Acquired and Persistent Risk Factors
Certain persistent medical conditions significantly elevate DVT risk. Congestive heart failure (CHF) in patients under 40 increases DVT risk several-fold compared to older individuals. Reduced cardiac output and patient immobility in CHF contribute to venous stasis, a major DVT risk factor. The severity of heart failure directly correlates with DVT incidence. Advanced cancer significantly increases DVT risk, with an overall seven-fold increase compared to the general population. Specific cancers, including non-Hodgkin lymphoma, lung, ovarian, brain, pancreatic, and gastrointestinal cancers, have higher DVT association rates. Chemotherapeutic agents like 5-fluorouracil, tamoxifen, and cisplatin can also increase DVT risk by influencing hypercoagulability, endothelial injury, or blood flow. Other acquired risk factors include acute infections, chronic inflammatory diseases (e.g., systemic lupus erythematosus, rheumatoid arthritis), chronic obstructive pulmonary disease, chronic kidney disease, recent stroke (with a 15% increased DVT incidence within three months post-stroke), recent major surgery (especially orthopedic), and trauma, such as spinal cord injuries. Estrogen levels also play a dose-dependent role in DVT risk, particularly in women on hormone replacement therapy or combination oral contraceptives. Pregnancy and the postpartum period increase DVT risk by approximately 1.4%. This hormonal influence is also believed to contribute to the higher DVT incidence in premenopausal women (under 50).
Modifiable Risk Factors
Modifiable lifestyle factors significantly contribute to DVT risk. Obesity, particularly increased waist circumference, and cigarette smoking are major modifiable risk factors. Obese individuals are twice as likely to develop DVT. Childhood obesity can also lead to persistent increased DVT risk into adulthood, even if BMI normalizes. Metabolic disorders like diabetes, immobilization, dehydration, and long-term steroid use are also modifiable risk factors. Addressing these modifiable factors is crucial for DVT prevention strategies in urgent care and beyond.
Diagnosis of DVT in Urgent Care: Utilizing the Wells’ Score and D-dimer
Diagnosing DVT in urgent care requires a systematic approach, combining clinical assessment with appropriate diagnostic testing. While classic DVT presentation includes unilateral leg pain, swelling, and redness, symptoms are often nonspecific, necessitating objective tests to confirm the diagnosis.
D-dimer Testing in Urgent Care
D-dimer is a widely used and readily available screening test for DVT and PE in urgent care settings. It measures a fibrin clot degradation product, elevated in both acute and chronic thrombosis. While highly sensitive, D-dimer’s low specificity, leading to frequent false positives, limits its use as a standalone diagnostic test. Conditions such as recent surgery, trauma, pregnancy, older age, and cancer can cause elevated D-dimer levels. To enhance diagnostic accuracy, pretest probability assessment, such as the Wells’ score, is often combined with D-dimer testing. The advent of point-of-care, high-sensitivity D-dimer tests holds significant promise for improving DVT diagnosis efficiency in urgent care, allowing for rapid results and quicker clinical decision-making.
Interpretation of the Wells’ Score for DVT
The Wells’ DVT risk score (Table 1) is a validated clinical decision tool used to estimate pretest probability of DVT. A score of 0 indicates a 5% DVT risk, 1-2 represents a 17% risk, and 3 or more signifies a 17-53% risk. Even the lowest Wells’ score (-2) carries a 5% DVT risk, highlighting that the Wells’ score alone cannot definitively exclude DVT. Current diagnostic guidelines recommend a combined approach: patients with a Wells’ score of 1 or less (low risk) should undergo D-dimer testing. A negative D-dimer can reliably exclude DVT in these low-risk patients. A positive D-dimer, however, necessitates confirmatory diagnostic imaging. Patients with a high-risk Wells’ score of 2 or more may proceed directly to diagnostic imaging, bypassing D-dimer testing.
Age-Adjusted D-dimer Levels
D-dimer levels naturally increase with age, further reducing specificity in older patients. An age-adjusted D-dimer threshold, calculated as age multiplied by 10 ng/mL for patients over 50, has been proposed to improve specificity and reduce false positives in this population. This age-adjusted strategy has demonstrated improved specificity and a reduction in false positive results, making D-dimer testing more reliable in older adults.
Venous Compression Ultrasound
In outpatient settings, venous compression ultrasound is the preferred diagnostic imaging modality for DVT. Alternative imaging options like venography, CT venography, and MR venography exist but are less desirable due to higher costs, radiation exposure (CT), contrast media reactions, or invasive nature (venography). However, in urgent care settings where immediate ultrasound access may be limited, the availability of point-of-care D-dimer testing becomes even more crucial for efficient triage and risk stratification.
Differential Diagnosis of Leg Swelling in Urgent Care
When evaluating patients presenting with leg swelling in urgent care, it’s crucial to consider differential diagnoses beyond DVT. Several conditions can mimic DVT symptoms, requiring careful clinical assessment and targeted investigations.
- Distal DVT: Ultrasound may detect DVT in calf veins. Anticoagulation is debated in these cases unless risk factors for extension (unprovoked DVT, prior VTE) are present, or clot propagation is observed on serial ultrasound (2-3 weeks post-initial diagnosis).
- Severe Calf Muscle Pull/Trauma: History of injury is typical, and ultrasound may show signs of bleeding, hematoma, or bruising at the ankle.
- Superficial Thrombophlebitis: Presents with tender, hard, red, swollen superficial veins. May coexist with DVT and warrants venous ultrasound evaluation.
- Cellulitis: Causes warmth, swelling, and redness, similar to DVT. Both conditions can coexist, necessitating ultrasound for differentiation.
- Lymphedema: A cause of chronic edema. Acute swelling or pain warrants ultrasound to rule out DVT, as distinguishing lymphedema from DVT-related edema can be challenging clinically.
- Popliteal (Baker’s) Cyst: Characterized by posterior knee pain, stiffness, and a palpable mass behind the knee. Ultrasound is often used to confirm Baker’s cyst and exclude DVT, though less urgently.
- Interstitial Edema: Lower extremity edema common in heart failure, liver disease, or medication side effects (e.g., dihydropyridine calcium channel blockers). Usually bilateral, but can be asymmetric with underlying venous pathology. Inflammation signs are typically absent. Low Wells’ score and negative D-dimer often rule out DVT.
Treatment of DVT in Urgent Care: Acute and Chronic Management
Anticoagulation is the cornerstone of DVT management, aiming to prevent clot progression, recurrence, and alleviate acute symptoms. DVT management is divided into acute (first 3-6 months) and chronic phases. Most DVT cases can be managed outpatient, except for severe cases like proximal clots (common femoral/iliac veins), phlegmasia/limb ischemia, significant comorbidities (end-stage renal disease), and high bleeding risk.
Acute DVT Treatment Options
Several anticoagulation options exist for acute DVT, including vitamin K antagonists (warfarin), direct oral anticoagulants (DOACs), low-molecular-weight heparin (LMWH) (Table 2), and unfractionated heparin. Optimal choice depends on patient comorbidities, renal function, and practical considerations (dosing frequency, route, cost). DOACs are often preferred for initial DVT treatment and may or may not require preceding parenteral anticoagulation (LMWH). If warfarin is chosen, initial parenteral anticoagulation is needed for at least 5 days until INR is therapeutic (≥2.0 on two occasions 24 hours apart). Parenteral bridging options include LMWH or unfractionated heparin (UFH). Guidelines generally favor DOACs over warfarin for most non-cancer-related DVTs due to lower major and fatal bleeding rates, more predictable pharmacokinetics, rapid onset of action, and avoidance of frequent INR monitoring.
Special Clinical Scenarios and Anticoagulation
Specific clinical scenarios may warrant particular anticoagulant choices (Table 3). For instance, warfarin is preferred over rivaroxaban in high-risk antiphospholipid syndrome patients due to lower thromboembolic event rates. LMWH is the standard treatment for cancer-associated DVT, showing lower major bleeding rates from gastrointestinal events compared to DOACs. However, in non-GI cancers, DOACs are acceptable alternatives to LMWH, with comparable bleeding risks and potentially lower recurrence rates. Acute proximal DVTs typically require 3-6 months of anticoagulation. Isolated distal calf DVTs can be managed with shorter 4-6 week anticoagulation or serial compression ultrasonography without anticoagulation, monitoring for clot propagation.
Chronic DVT Treatment and Recurrence Risk
Extending anticoagulation beyond the initial 3-6 months depends on recurrence risk stratification. High-risk patients (>3% recurrence risk) include those with active cancer, autoimmune disease, or antiphospholipid syndrome. Annual DVT risk assessment and anticoagulation need are recommended for these patients. While provoked vs. unprovoked DVT classification is less emphasized in determining treatment duration due to overlapping predisposing factors, provoked DVT in the context of transient risk factors like surgery or trauma generally does not warrant prolonged anticoagulation. Postthrombotic syndrome (PTS) or venous insufficiency occurs in 25-50% of patients within 3-6 months post-DVT. Prolonged anticoagulation is not typically warranted for provoked DVTs related to trauma, minor surgery (anesthesia >30 minutes), or acute illness with immobility >3 days.
Case Conclusion: Point-of-Care D-dimer Impact in Urgent Care
Applying the Wells’ score to the presented case, the patient receives 1 point for pain along the deep venous system, classifying her as moderate risk (17% pretest probability). Following current guidelines, a high-sensitivity D-dimer test is indicated. In this case, a negative high-sensitivity D-dimer test was obtained in urgent care. In moderate-risk patients, a negative point-of-care high-sensitivity D-dimer effectively rules out DVT with a 96.1% negative predictive value, obviating the need for further testing. The patient is safely discharged home with primary care follow-up. However, the reality is that many urgent care facilities lack on-site rapid D-dimer testing. The availability of point-of-care high-sensitivity D-dimer testing in urgent care settings would significantly improve patient care by reducing delays in diagnosis, minimizing diagnostic uncertainty, and decreasing associated healthcare costs. This advancement would empower urgent care providers to efficiently and accurately manage suspected DVT cases, ultimately enhancing patient outcomes.
References
- What is venous thromboembolism? https://www.heart.org/en/health-topics/venous-thromboembolism/what-is-venous-thromboembolism-vte. Published March 1, 2017. Accessed July 23, 2021.
- Phillippe, HM. Overview of Venous Thromboembolism. American Journal of Managed Care. https://www.ajmc.com/view/overview-of-venous-thromboembolism. Published December 27, 2017. Accessed July 23, 2021.
- Chopard R, Albertsen IE, Piazza G. Diagnosis and Treatment of Lower Extremity Venous Thromboembolism: A Review. JAMA. 2020;324(17):1765–1776. doi:10.1001/jama.2020.17272
- Phillippe, Haley M. Overview of Venous Thromboembolism. Am J Manag Care. 2017;23: S376-S382
- Horner, Dan. New NICE Guidelines on diagnosis and management of venous thromboembolism. St. Emlyn’s, May 2020, https://stemlynsblog.org/nice-guidelines-vte/
- Cohoon KP, Heit JA. Inherited and Secondary Thrombophilia. Circulation. 2014;129:254–257. https://doi.org/10.1161/CIRCULATIONAHA.113.001943
- Wells PS, Hirsh J, Anderson DR, et al. Accuracy of clinical assessment of deep-vein thrombosis. Lancet. 1995;345(8961):1326-30.
- Wells PS, Anderson DR, Bormanis J, et al. Value of assessment of pretest probability of deep-vein thrombosis in clinical management. Lancet. 1997;350(9094):1795-8.
- Wells PS, Anderson DR, Rodger M, Forgie M, Kearon C, Dreyer J, Kovacs G, Mitchell M, Lewandowski B, Kovacs MJ. Evaluation of D-dimer in the diagnosis of suspected deep-vein thrombosis. N Engl J Med. 2003 Sep 25;349(13):1227-35.
- Wilbur J, Shian B. Deep venous thrombosis and pulmonary embolism: current therapy. Am Fam Physician. 2017;95(5):295-302.
Author affiliations: John DesMarais, MD is program director, Adena Family Medicine Residency. Samidha Dutta, DO, PGY-3 is a third-year resident in theAdena Family Medicine Residency. The authors have no relevant financial relationships with any commercial interests