Introduction
Chronic Kidney Disease (CKD) is a widespread health issue globally, with staggering costs exceeding $1 trillion spent on end-stage renal disease (ESRD) care alone. The compelling clinical and economic reasons to develop effective health system strategies to slow down CKD progression to ESRD are undeniable. This article delves into the current gaps in our understanding: which interventions are most effective in early CKD, the ideal timing for these interventions, and the most suitable models of care to implement.
Current diagnostic tools have limitations, and improvements in clinical care hinge on the ability to distinguish between early-stage (1–3) CKD at risk of progressing to ESRD and early CKD that is unlikely to advance. Reconsidering CKD as an integral part of primary care might be beneficial. Further research is crucial to better identify risk factors for CKD progression. Systems modeling offers a valuable approach to assess the impact of different care models on CKD outcomes and associated costs. The successful experience of the US Indian Health Service highlights the potential of an integrated, system-wide approach to yield significant benefits, even within resource-constrained systems.
CKD is a prevalent condition marked by kidney damage or impaired kidney function, significantly elevating the risk of cardiovascular disease.1, 2 The current CKD classification system relies on estimated glomerular filtration rate (eGFR) and urinary albumin excretion rate (AER) (Table 1).3 Clinicians also look for signs of kidney damage, such as abnormalities in urine sediment or kidney structure, to diagnose CKD in individuals with eGFRs ≥60 mL/min/1.73 m2. Diabetes and hypertension are the primary causes, accounting for up to two-thirds of CKD cases.4 Other less common causes include glomerulonephritis, kidney stones (nephrolithiasis), and polycystic kidney disease. In some instances, progressive kidney damage leads to ESRD, requiring dialysis or kidney transplantation for survival. The pace of CKD progression varies significantly among individuals, influenced by the underlying cause and disease characteristics.5, 6
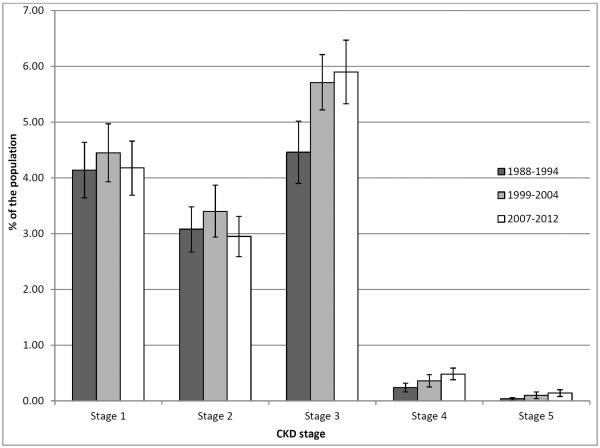
In the United States, the prevalence of CKD among non-institutionalized adults rose from 12.0% (95% CI: 10.4% – 13.5%) to 14.0% (95% CI: 12.4% – 15.7%) between 1988–94 and 1999–2004. More recent data (2007–2012) suggests a possible stabilization, with a prevalence rate of 13.7% (95% CI: 12.1% – 15.2%).7 CKD is a significant health concern in most high-income nations,1, 5 although UK data indicates a decrease from 6.7% to 6.0% between 2003 and 2009–2010.8
ESRD patients constitute a substantial portion of healthcare expenditure in many high-income countries.9 Global spending on ESRD care is estimated to exceed $1 trillion.10 Addressing the progression from early chronic kidney disease to ESRD is both clinically vital and economically sound.11–14 This article aims to highlight the knowledge gaps surrounding effective early interventions for early CKD diagnosis management and models of care, including identifying appropriate interventions, determining the optimal intervention timing, and selecting the best care models.
CKD Diagnosis
Defining early chronic kidney disease is the crucial first step in developing an effective intervention strategy. The National Kidney Foundation’s publication of the first CKD guidelines in 2002 was a pivotal moment, bringing much-needed policy attention to CKD. These guidelines, known as the KDOQI guidelines, have been adopted globally and serve as the foundation for CKD classification.
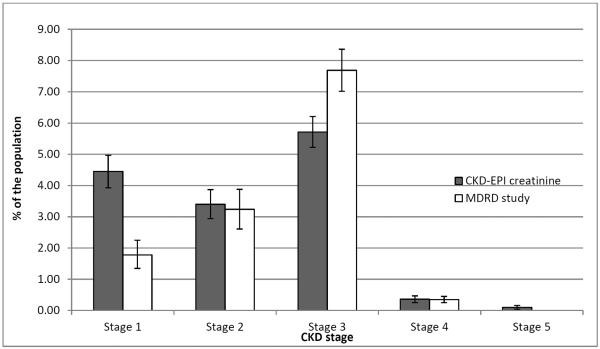
Based on current prevalence estimates,7 a significant portion of the US population – 44.6 million people, including 33.6% of those aged 60 and older – are living with CKD. Over 95% of these individuals are classified as having stages 1–3, leading to the characterization of the current situation as a “silent epidemic”10 and “the tip of the iceberg.”15 A recent study projects that a person born in the US today faces a lifetime risk of developing CKD stages 3a+, 3b+, 4+, and ESRD of 59.1%, 33.6%, 11.5%, and 3.6%, respectively.16 These prevalence rates are largely determined by eGFR readings, a proxy for kidney function, typically calculated using the CKD-EPI (CKD Epidemiology Collaboration) or MDRD Study (Modification of Diet in Renal Disease) formulas. Albuminuria levels provide additional diagnostic information.
eGFR Equations and Age-Related Decline in Renal Function
These high lifetime risks associated with CKD raise a critical question: is there a clear distinction between early chronic kidney disease and the normal age-related decline in kidney function? Natural aging processes include reductions in renal blood flow and kidney mass, along with increased glomerulosclerosis, resulting in an average eGFR decline of about 0.75 mL/min/1.73 m2 per year after age 40.17 This decline appears to be non-linear, with eGFR loss slowing down in elderly patients once eGFR drops below 45 mL/min/1.73 m2.18 Population studies reveal that the majority of individuals diagnosed with CKD are over 60, and most do not exhibit significant albuminuria.19, 20 This makes it challenging to differentiate between age-related kidney function decline and true renal disease.21 Data suggests that for a given eGFR reduction, older patients are less likely to progress to ESRD.18, 22 The impact of aging is well-recognized in other organ systems. For instance, the natural decline in forced expiratory volume with age serves as a baseline for identifying premature or accelerated loss of respiratory function.23
However, meta-analyses from the CKD Prognosis Consortium, encompassing over 1.5 million patients, have demonstrated nearly identical ESRD risks in patients both above and below 65 years of age with an eGFR of 45–59 and a similar albumin creatinine ratio (ACR).24, 25 These findings have been interpreted as arguments against age-based adjustments in CKD definitions, although the interplay between kidney function and proteinuria may indeed vary with age, potentially due to competing mortality risks. It has also been proposed that age-related eGFR changes might stem from other disease processes rather than predetermined renal decline.26, 27 The diverse interpretations of current data on eGFR loss in older adults underscore the importance of considering eGFR trends within a comprehensive clinical assessment. Regardless of whether these eGFR changes reflect intrinsic renal disease or normal aging, both CKD and aging are independently associated with increased morbidity and mortality, and their effects appear to be additive.18
Comorbidity is highly prevalent in CKD patients. In the UK, approximately 64% of CKD patients over 65 have four or more additional health conditions.28 While it’s recognized that multimorbidity increases healthcare needs, the specific risk factors for multimorbidity remain poorly understood.29 Further research is needed to determine if kidney impairment in elderly individuals is linked to or contributes to other health conditions.
eGFR estimation formulas have well-documented limitations.30, 31 These formulas were primarily developed to identify patients with eGFR ≤60 mL/min/1.73 m2 at risk of renal failure, and they lack sensitivity for stages 1 and 2.32 Therefore, eGFR estimates alone are of limited value in Early Chronic Kidney Disease Diagnosis Management And Models Of Care for early intervention. Some experts have even suggested removing the first two stages from the KDOQI guidelines,33 while others have proposed alternative classification systems.34 The MDRD Study equation tends to underestimate true GFR in individuals with normal kidney function,35–38 whereas the CKD-EPI equation tends to overestimate it in individuals with CKD or at high risk for CKD.39 Both equations provide eGFR figures within 30% of the true values, but in a significant proportion of cases (15.9% for CKD-EPI40 and 19.4% for MDRD Study41), the accuracy is even lower. Gender and ethnic differences in GFR also need to be considered.22, 33
Epidemiological studies have employed different eGFR formulas, which limits direct comparisons due to varying accuracy at higher eGFR levels. Most national studies also rely on single eGFR measurements, while a definitive CKD diagnosis requires multiple estimates over several months. Single point estimates tend to overestimate prevalence rates. Furthermore, not all studies incorporate AER when estimating prevalence.
Cystatin-C-Derived eGFR Equations
Limitations of creatinine-based GFR estimates have prompted investigations into alternative molecules, including beta-trace protein42 and cystatin C.43 Cystatin C has shown promise in providing more accurate renal function estimates, particularly in elderly patients,44–46 and especially when combined with serum creatinine measurements.47 Cystatin-C-derived formulas also appear to be more reliable at higher GFRs. Current recommendations suggest using these formulas to confirm or rule out CKD in patients with mild eGFR reductions (creatinine-based) and an albumin creatinine ratio (ACR) of <30 mg/g.3, 48 This approach could lead to cost savings by reducing unnecessary healthcare utilization for misdiagnosed CKD. Although an international standard for cystatin-C measurement now exists, the assay isn’t yet widely available. Increased adoption of cystatin-C assays could improve diagnostic accuracy in early chronic kidney disease diagnosis management and models of care.
Albuminuria
While AER provides valuable diagnostic information, albuminuria tests also have limitations, including poor test-retest reliability,49, 50 further reinforcing the need to rely on multiple prognostic factors. Albuminuria readings can vary depending on assay type (monoclonal vs. polyclonal),51 sample collection method (24-hour collection vs. first morning void vs. random spot sample),52 and storage procedures.53 Moreover, standard thresholds for pathological albumin excretion are used across genders and ages, but these factors can influence urinary albumin measurement.54–56
Key Points for Clinical Practice
eGFR estimates that don’t incorporate cystatin-C data are inherently imprecise, a fact often overlooked in both research and clinical practice. Clinicians need to be acutely aware of the strengths and limitations of current diagnostic tools. Primary-care physicians (PCPs) should exercise clinical judgment when evaluating individual patients, considering eGFR trends and albuminuria levels. While these two parameters together offer valuable predictive information about ESRD risk,57, [58](#R58] clinicians should also consider the severity of comorbidities, family history, and vascular risk profiles.59, [60](#R60] Further research is essential to enhance the sensitivity and specificity of eGFR formulas and other risk prediction models for progressive CKD, especially in early chronic kidney disease diagnosis management and models of care.
How to Define and Manage Early CKD?
Given that most CKD patients do not progress to ESRD, indiscriminate treatment is neither clinically sound nor economically sustainable, especially considering the global scale of the disease. Refining clinical care requires differentiating between early chronic kidney disease at risk of progressing to ESRD and KDOQI stages 1–3 CKD that is unlikely to advance. Intervention studies would also benefit from a more selective definition of early CKD diagnosis management and models of care. For example, the benefits of dietary salt restriction remain inconclusive,61 with a post-hoc analysis of the ONTARGET and TRANSCEND studies showing no renal benefit in patients with early chronic kidney disease. 62 However, given the reported benefits of low-salt diets in advanced renal failure patients,63 considering specific patient phenotypes might be necessary.
Screening Debate
A systematic review commissioned by the US Preventive Services Task Force (USPSTF) questioned the clinical and economic value of screening both the general population and high-risk groups for early chronic kidney disease;64 this position is supported by the American College of Physicians (ACP).65 The USPSTF review found no randomized controlled trials (RCTs) on “CKD screening in adults who were asymptomatic with or without recognized risk factors for CKD incidence, progression, or complications,” and no RCTs on “monitoring adults with CKD stages 1 to 3 for worsening kidney function or damage.”
The USPSTF and ACP stance is not universally accepted. In the UK, the National Institute for Health and Care Excellence (NICE) recommends targeted CKD assessment for patients prescribed high-risk medications or with conditions linked to CKD, such as diabetes mellitus.48 The American Society of Nephrology (ASN) argues for universal CKD screening due to the asymptomatic nature of mild-to-moderate CKD and the ease of investigation. While acknowledging the lack of RCT support, the ASN proposes that early and improved blood pressure control can slow renal function decline, and CKD awareness can be crucial during hospitalizations. A recent systematic review concluded that proteinuria screening might be cost-effective in diabetic and hypertensive populations. This review also suggested that eGFR screening might be cost-effective in diabetic patients, but cost-effectiveness studies for eGFR screening in hypertensive populations were lacking.66
Evidence Gaps
These conflicting viewpoints highlight the urgent need for more RCTs on early chronic kidney disease identification, screening, monitoring, and treatment. In the interim, population health management could prioritize vascular risk factor control and focus on CKD patients at higher risk of progression to ESRD and increased healthcare costs.67, 68 Given the asymptomatic and insidious onset of CKD, research into novel biomarkers and prognostic techniques is vital for improved early CKD diagnosis management and models of care.69–71 This is an active research area, with new prediction models being continuously developed.72–76 Further validation and refinement of these models are necessary. It’s crucial to assess the generalizability of findings across diverse patient groups, as a model effective for one population in a specific region may be less accurate or even misleading when applied to others.
Expanding the definition of early chronic kidney disease must consider the adverse outcomes associated with reduced eGFR.77 Two recent meta-analyses by the CKD Prognosis Consortium found that diabetic and hypertensive patients with reduced eGFR have increased risks of ESRD and death.78, 79 In both studies, ESRD risk significantly increased once eGFR fell below 50 mL/min/1.73 m2. However, the risk of death increased below an eGFR of 90 mL/min/1.73 m2. While these findings could support defining mild renal function reductions as a disease state, it’s crucial to consider the different risk thresholds for death and ESRD. These findings also raise questions about population versus individual risk, given that even patients with low ESRD risk may still face risks of other complications.80 For instance, most new diabetes diagnoses occur in these lower-risk groups.81 Therefore, screening might not provide meaningful prognostic information regarding ESRD risk at an individual level. Small eGFR reductions have been shown to improve cardiovascular risk model discrimination, and the rate of eGFR change is considered a stronger risk predictor than absolute values.82, 83
It’s plausible that what is currently classified as CKD stage 1–2 should be evaluated in relation to cardiovascular risk rather than solely renal risk. This is particularly relevant for older adults with reduced eGFR but no significant albuminuria. Both meta-analyses78, 79 demonstrated a linear association between increased albuminuria and the risk of death and ESRD, highlighting the importance of albuminuria as a marker for CKD progression in early chronic kidney disease diagnosis management and models of care.
Management of Early-Stage CKD
The evidence base supporting lifestyle modifications and preventive medications to reduce cardiovascular disease risk and potentially slow, halt, or reverse CKD progression in early stages is limited.1, 84 While generally low-quality, existing data offers some support for calorie-controlled diets,85–88 physical exercise,89–91 and smoking cessation92–94, primarily in diabetic CKD patients. Most CKD patients have comorbidities amenable to systematic prevention and early management. Many patients die from cardiovascular events or other causes before ever developing ESRD.59, 77, 95
The pathophysiology of increased vascular risk in CKD evolves throughout the disease’s natural course,96 with traditional atherosclerotic risk factors having a proportionally greater impact in early stages. In early and pre-dialysis CKD,97, [98](#R98] lipid-lowering agents reduce cardiovascular event risk, despite a weaker association between low-density lipoprotein cholesterol (LDL-C) and patient outcomes in stages 3 through 5.99 Improved awareness of CKD as a vascular risk factor would promote more timely use of these agents alongside established interventions like blood pressure control, crucial for effective early chronic kidney disease diagnosis management and models of care.100 The strongest evidence for treatment benefit in early CKD comes from RCTs of angiotensin-converting enzyme (ACE) inhibitors101 and angiotensin II-receptor blockers (ARBs).102, 103 However, these studies primarily focused on patients with proteinuric diabetic renal disease, and evidence supporting these agents for other CKD causes is less robust.104
Recent data suggests a slowing increase in ESRD incidence in some countries, including Australia, Canada, northern European nations, New Zealand, and the USA, though trends vary across subgroups (e.g., age and race).105 This stabilization may be attributed to improved cardiovascular risk management in diabetic, hypertensive, and general populations, although conclusive evidence is still needed.
Models of CKD Care
The Wagner Chronic Care Model
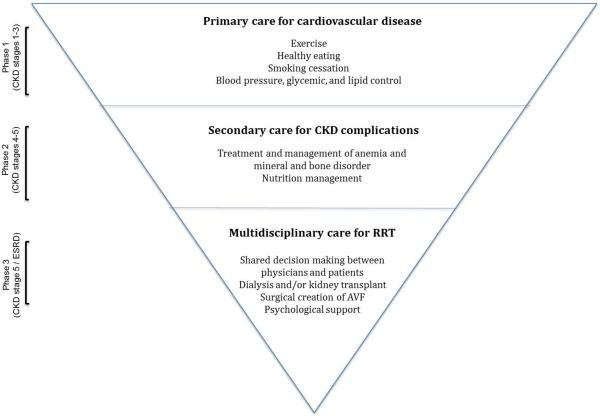
Evidence suggests a three-phase care strategy for early chronic kidney disease diagnosis management and models of care:106, 107
- Vascular risk management in primary care (early disease): Exercise, dietary changes, smoking cessation, blood pressure, glycemic and lipid control, and periodic kidney health monitoring.
- Structured care (progressive cases): Target comorbidities like anemia, bone disease, and secondary hyperparathyroidism.
- Multi-professional, intensive care (ESRD transition): For patients transitioning to renal replacement therapy.
For all CKD patients, PCPs should check for drug interactions that could cause acute kidney injury.
Given CKD stage distribution, almost all patients would fall into the first phase. Reconceptualizing CKD as a primary care concern108 and adopting an integrated care approach to coordinate the CKD care continuum (Box 1)109 aligns with Dr. Edward H. Wagner’s Chronic Care Model. This model comprises six core components: community resources and policies, healthcare organization, self-management support, delivery system design, decision support, and clinical information systems.110–113 These practice changes are crucial as diabetes and hypertension prevalence are projected to rise,114–116 increasing the CKD burden. A primary care model shift could allow nephrologists and specialists to focus on primary kidney disease, rapidly progressing CKD, and ESRD.
PCPs should also closely monitor the renal effects of certain drugs, contrast dyes, and environmental toxins, potentially aided by IT systems. Some prescription and over-the-counter medications, like non-steroidal anti-inflammatory drugs, can trigger acute kidney injury in CKD patients, accelerating CKD progression.117–119
Numerous studies highlight primary care shortcomings in CKD management. Notably, there’s low awareness of KDOQI guidelines and diagnostic techniques,120–125 suboptimal prescribing and management for diabetic and hypertensive patients,7, 124–129 and poor recognition of known CKD risk factors.120 One study found only 35% of PCPs had “adequate knowledge” of CKD based on a 27-question survey of 470 clinicians; knowledge odds decreased by 26% per decade of PCP age increase.130 Another study found only 19% of family practitioners and 33% of general internists adhered to KDOQI guidelines for CKD laboratory and radiological evaluation.122 A US National Kidney Disease Education Program-commissioned study found about one-third and one-fourth of PCPs didn’t recognize family history and African-American race as CKD risk factors, respectively; however, diabetes and hypertension awareness as CKD predictors was high.120 These surveys120, 122, 130 had low voluntary response rates (7.6% to 32.4%), and participants may not represent the general PCP population. These findings, already indicating significant improvement potential in early chronic kidney disease diagnosis management and models of care, might overestimate actual CKD care quality.
The Involvement of Specialists in CKD Care
Timely specialist involvement is crucial to improve health outcomes for progressive CKD patients, but payers aim to avoid unnecessary referrals that strain resources. A systematic review of early versus late (or no) nephrologist referral found early referral linked to better health outcomes and potential cost-effectiveness.131 However, the authors found no RCTs on early referral strategies’ clinical effectiveness, and only two studies included pre-dialysis patients. They noted insufficient data on CKD natural history and early referral costs and effects. Long-term observational studies of early CKD patients are needed to better understand disease progression and cardiovascular event incidence in patients with and without conditions like diabetes, pre-existing cardiovascular disease, albuminuria, and proteinuria. The authors also suggested that early referral’s high costs might be unaffordable, even if cost-effective. Further research is needed to evaluate improved primary care cost-effectiveness for early chronic kidney disease patients.
PCPs should be involved in redefining CKD to facilitate a paradigm shift in early chronic kidney disease diagnosis management and models of care. In the UK, early CKD identification is financially incentivized in primary care, but some PCPs question whether CKD is a genuine disease state.132 In areas with incorrect patient identification on CKD registers, cardiovascular management is suboptimal, with poorer blood pressure and cholesterol control.133 A primary care model transition should be a universal decision, unlike current guideline development driven by secondary care providers.134
Integrated Care Pathways for CKD
A unified patient care strategy across healthcare providers and payers could improve healthcare outcomes in early chronic kidney disease diagnosis management and models of care. Evaluating CKD care models for value for money and understanding approaches for reliable service delivery to high-risk populations is crucial. CKD serves as a useful case study for implementing a public health-oriented, proactive, and patient-centered primary care model.
USA
Despite US healthcare system fragmentation, positive CKD trends are observed in some public (Indian Health Service135, 136 and Veterans Health Administration137) and private healthcare organizations (Kaiser Permanente138, 139). The long-term patient focus of the Indian Health Service and Veterans Health Administration encourages preventive care. In the Indian Health Service, widespread ACE inhibitor use in the early 1990s was a key intervention. Today, about 80% of hypertensive patients with diabetes in this population receive ACE inhibitors. CKD and ESRD rate growth is slowing faster among these patients than in the general population.135, 136 The Indian Health Service experience shows that a system-wide approach, even in an underfunded system, can yield significant benefits in early chronic kidney disease diagnosis management and models of care.
Kaiser Permanente of Southern California, a vertically-integrated health maintenance organization, deviates from KDOQI guidelines.139 It uses a composite risk assessment to target patients at risk of worsening conditions and IT systems to automatically recommend treatment options based on patient data. Kaiser Permanente Hawaii has also implemented risk stratification-based CKD care, showing a statistically significant reduction in disease progression.138
UK
The UK is seeing increasing integration between primary and secondary care in early chronic kidney disease diagnosis management and models of care. A 2003 study, pre-automated eGFR reporting, reviewed electronic primary care records of over 130,000 patients and found high undiagnosed CKD rates.140 While automated eGFR reporting has improved CKD recognition in primary care and increased referrals, this study also suggests alerting PCPs to missed preventive prescribing opportunities.
A similar CKD management model to Kaiser Permanente’s138 was phased in between 2003 and 2006 in England’s West Midlands region. Early results show improved patient outcomes, including a reduction in population-adjusted renal replacement therapy incidence.141 Another study evaluated health outcomes of CKD stages 4–5 patients in primary care with a disease management program similar to a secondary care multi-disciplinary clinic. The program improved health outcomes and reduced eGFR loss over nine months.142 Intensive, target-driven disease management programs have yielded positive diabetes care outcomes,143 and similar approaches have potential for CKD care, particularly in early chronic kidney disease diagnosis management and models of care.
Other International Experiences
Other countries, including Australia and Canada, are also transitioning to primary care models for CKD. Similar to the Indian Health Service, Australia has long used community-based CKD care for the Tiwi Islands aboriginal community. The Tiwi Islands model focuses on blood pressure and lipid control and health education to improve cardiovascular health and preserve renal function. Published data suggests this strategy has significantly improved cardiovascular and renal health outcomes in this population.144 These international experiences suggest a holistic care model can be successfully applied across diverse healthcare systems for effective early chronic kidney disease diagnosis management and models of care.
Systems Modeling
As mentioned, physicians should consider population-based estimating equation limitations and evaluate individual patient characteristics when predicting risk and developing treatment plans for early chronic kidney disease diagnosis management and models of care. Structured, early CKD intervention programs can form the basis for personalized care. Given early CKD patient heterogeneity and variable risks across populations, clarity on program scope and impact is needed. Healthcare providers, payers, and the public need to know the cost-effectiveness of investing in CKD intervention programs.
Systems modeling is a cost-effective method to study different interventions’ effects on chronic disease outcomes and costs.145–147 Software can design comprehensive care models in patient populations and observe diabetes, hypertension, CKD, and ESRD burdens over time. Systems modeling allows sensitivity analysis to test model assumption uncertainty, indicating result reliability and highlighting areas for further research in early chronic kidney disease diagnosis management and models of care.
Alongside systems modeling, long-term prospective cohort studies are needed to understand different intervention strategy clinical and economic value from payer or societal perspectives. Ideally, these studies should incorporate RCTs of CKD treatments. Economic analyses should also accompany outcome evaluations. Notably, the Nord-Trøndelag Health Study (HUNT) in Norway,148 Kaiser Permanente,139 English West Midlands141 studies, and most CKD quality improvement studies don’t consider costs. Integrating health-economic evaluations into these population-based studies would generate useful evidence to guide care approaches. Relevant stakeholders should also establish registries to monitor long-term health and cost effects of different care pathways and care initiation timing for early chronic kidney disease diagnosis management and models of care.
Conclusion
There’s insufficient evidence on early CKD intervention clinical and economic benefits, especially compared to diseases like diabetes and stroke. RCTs on early intervention strategy clinical and cost outcomes are lacking, and comprehensive patient-level data is not readily accessible in early chronic kidney disease diagnosis management and models of care. However, early CKD diagnosis, treatment, and management could alter the disease’s natural history and generate substantial cost savings. A better understanding of different CKD care approach merits and demerits, and where evidence is lacking, is essential to improve health outcomes and minimize expenditure.
CKD, like many chronic conditions, is essentially a broad indicator of overall health. Patients with mildly or moderately reduced eGFRs often have comorbidities more relevant to their current and future well-being than a CKD diagnosis. These comorbidities should remain the treatment and management focus, as few CKD-diagnosed patients develop ESRD.
Physician difficulty in identifying progressive CKD patients has weakened early intervention effectiveness in early chronic kidney disease diagnosis management and models of care. While new biomarkers may improve CKD detection and prognosis, a single solution to fully address the CKD burden is unlikely. Promoting a unified care strategy across healthcare providers and payers is crucial. This holds true for CKD, a complex disease involving many providers over a patient’s lifetime, but also for other chronic illnesses.
Box 1. Prevention Strategies for Early-Stage CKD.
Primary and secondary
- Clarification of CKD staging and prognostication to improve PCP engagement
- Patient and clinician education to link public health programs to kidney health
Target population
- Development of high-quality evidence to guide screening programs for CKD and RCTs
- Patients at risk of AKI or with previous AKI
- Continued assessment of high-risk populations
Impact assessment
- Assessment of patient awareness of disease and personal clinical data
- Proportion of patients experiencing cardiovascular events, preventable renal function loss, or requiring secondary care referral
Abbreviations: AKI, acute kidney injury; CKD, chronic kidney disease; PCP, primary care physician; RCT, randomized controlled trial.
Box 2. Service Delivery for CKD.
Primary care
- Patient assessment by eGFR trend and/or trajectory reporting
- Classification of CKD based on risk for progression
- Identification of CKD as an indicator for elevated cardiovascular risk, with early modification of traditional risk factors
- Patient advocacy and self-management during early-stage CKD
- Referral to secondary care for specialist treatment of CKD complications
Secondary care
- Multidisciplinary management of disease complications
- Ongoing support for patient self-management programs
- Integration with other secondary care services to manage comorbidity burden
- Personalized treatment goals considering quality of life
- Integration into primary care to support periodic monitoring of stable patients by PCPs
- Structured follow-up for patients experiencing AKI, with data collection to describe long-term effects on GFR trajectory
Abbreviations: AKI, acute kidney injury; CKD, chronic kidney disease; eGFR, estimated glomerular filtration rate; PCP, primary care physician.
Box 3. Workforce, ICT, and Other Strategies to Improve Care for Early-Stage CKD.
Workforce
Essentials and supportive
- Motivated and educated workforce and patient population
- Easy access to laboratory monitoring
- Specialist nursing staff to support patient understanding of the disease
- Financially viable secondary care renal services for a potentially smaller but very ill patient population
PCP-specialist interface
- Multi-specialty clinics in primary care to support PCP education and patient care
- Defined referral and discharge criteria for secondary care
Role of ICT and decision-support systems
- Integration of primary and secondary care records
- Accessible results reported to patients in any location
- Automated analysis of eGFR and/or proteinuria trends
- Incorporation of validated predictive models for ESRD into laboratory reports
- Electronic prescribing linked to biochemical results
Health-economic impact and health-system financing
- Economic analyses built into all studies of CKD screening and treatment
- Financial incentives balanced towards preventing ESRD progression
- Establishment of CKD registries for health-economic analyses
Leadership, governance, and role of national and international organizations
- International, evidence-based accordance between national and international bodies regarding CKD screening and treatment
- Increased data sharing between health systems of epidemiologic CKD trends
- Strong patient representation in all organizations
Abbreviations: CKD, chronic kidney disease; eGFR, estimated glomerular filtration rate; ESRD, end-stage renal disease; ICT, information and communications technology; PCP, primary care physician.
Figure 2.
Figure 4.
Table 1.
Definition and classification of CKD
Table 2.
Selected observational studies of the quality of primary care for CKD patients
| Study | Design | Population | Period | Key results |
|---|---|---|---|---|
| Allen et al 2011124 | Retrospective cohort | 166 PCPs in 15 health centers in eastern Massachusetts, USA, caring for 11,774 CKD patients (eGFR between 15 and 60 mL/min/1.73 m2) | 2004–2008 | Many of the patients were not tested yearly for urine protein (70%), had BP ≥130/80 mmHg (46%), and were not receiving appropriate treatment with ACE inhibitors or ARBs (25%). More than a quarter of patients (26%) were receiving potentially-harmful medicines. |
| Boulware et al 2006121 | Cross-sectional (questionnaire) | National, random, stratified sample of 400 nephrologists and 800 PCPs (400 family practitioners and 400 general internists) in the USA; 959 eligible respondents. Responses obtained from 126 nephrologists and 178 PCPs (89 family practitioners and 89 general internists) (31.7%, 304/959) | 2004–2005 | Family practitioners (56.2%) and general internists (70.7%) were less likely to recognize CKD than nephrologists (96.0%) (P |
| Charles et al 2009122 | Cross-sectional (questionnaire) | Same population as in the study by Boulware et al (2006) | 2004–2005 | Only 48% of nephrologists, 19% of family practitioners, and 33% of general internists followed the KDOQI guidelines on the laboratory and radiological evaluation of patients with CKD (P |
| Fox et al 2006123 | Qualitative interviews | Ten PCPs from ten health care facilities affiliated with the Upstate New York Practice-based Research Network | Not stated | There was low awareness of KDOQI guidelines, and PCPs often favoured less-accurate diagnostic tests for CKD (that is, serum creatinine); there was uncertainty about the appropriate timing of referral to nephrologists. |
| Israni et al 2009130 | Cross-sectional (questionnaire) | Random sample of 1,550 US PCPs; 1,453 eligible respondents. Responses obtained from 470 PCPs (32.4%, 470/1,453) | 2007 | Only 35% of PCPs had “adequate knowledge” of CKD based on responses to 27 questions; for each 10-year increase in age of the PCP, the odds of having adequate knowledge decreased by 26%. |
| Lea et al 2006120 | Cross-sectional (survey) | PCPs in six predominantly African-American communities in the USA (number of contacted physicians not stated). Responses obtained from 464 PCPs (7.6%) | 2003 | About 34% and 22% of PCPs did not identify family history and African-American ethnicity as risk factors for CKD, respectively; there was high awareness that hypertension and T2DM are predictors of CKD. |
| Minutolo et al 2005127 | Cross-sectional | Nephrologists (tertiary care) and 39 PCPs caring for hypertensive patients with CKD (eGFR between 15 and 60 mL/min/1.73 m2) in Naples, Italy | 2003 | CKD patients cared for by PCPs had higher BP levels ([143±15] / [82±7] mmHg) than CKD patients cared for by nephrologists ([136±18] / [78±11] mmHg) (P |
| Ravera et al 2009129 | Cross-sectional | PCPs caring for 7,582 hypertensive patients with type 2 diabetes in Italy | 2005 | Only 10.4% of patients with diabetes mellitus in this population achieved BP 2, only 17% had been coded as having CKD. |
| Ravera et al 2011128 | Cross-sectional | PCPs caring for 39,525 hypertensive patients in Italy (nationally-representative sample of patients) | 2005 | Only 13.8% of patients with eGFR 2 and 72.6% of patients with eGFR 2 were coded as having CKD. Of patients with eGFR 2, only 45.4% and 13.2% achieved BP |
| Razavian et al 2012125 | Cross-sectional (survey) | Nationally-representative, cluster-stratified sample of 534 PCPs in Australia; responses obtained from 322 GPs (60.3%, 322/534) caring for 4,966 patients (age ≥55 years) with available data on kidney function | 2008 | Fewer than 18% of CKD patients (235/1,312) were correctly diagnosed with CKD. The decisions by PCPs not to prescribe BP- or lipid-lowering agents for CKD patients only adhered to guideline recommendations in 51% and 46% of cases, respectively. |


Abbreviations: ACE, angiotensin-converting enzyme; ARB, angiotensin II-receptor blocker; BP, blood pressure; CKD, chronic kidney disease; eGFR, estimated glomerular filtration rate; KDOQI, Kidney Disease Outcomes Quality Initiative; OR, odds ratio; PCP, primary care physician; T2DM, type 2 diabetes mellitus
Acknowledgments
No sources of funding were used to prepare this manuscript. The authors have no conflicts of interest that are relevant to the content of this article. We would like to thank Dr. Shari Ling and Dr. Kimberly Smith (Food and Drug Administration, Washington DC, USA) for their useful comments on earlier versions of this article.