IMPORTANCE
Chronic kidney disease (CKD) is a significant global health concern, ranking as the 16th leading cause of years of life lost worldwide. Effective screening, accurate diagnosis, and appropriate management by primary care clinicians are crucial in preventing the serious complications associated with CKD, including cardiovascular disease, end-stage kidney disease (ESKD), and premature death.
OBSERVATIONS
CKD is defined as persistent abnormalities in kidney structure or function, such as a glomerular filtration rate (GFR) below 60 mL/min/1.73 m2 or albuminuria of 30 mg per 24 hours or more, lasting for over 3 months. This condition affects a substantial portion of the global population, ranging from 8% to 16%. In developed nations, diabetes and hypertension are the most common culprits behind CKD. Alarmingly, less than 5% of individuals in the early stages of CKD are aware they have the disease. For those diagnosed with CKD, proper staging and risk assessment using tools that consider GFR and albuminuria are vital for guiding treatment plans, monitoring strategies, and referral decisions.
Optimal CKD management involves a multifaceted approach: reducing cardiovascular risk through interventions like statins and blood pressure control, treating albuminuria with agents like angiotensin-converting enzyme inhibitors (ACEIs) or angiotensin II receptor blockers (ARBs), avoiding nephrotoxic substances such as nonsteroidal anti-inflammatory drugs (NSAIDs), and carefully adjusting medication dosages for drugs like antibiotics and oral hypoglycemic agents. Regular monitoring for CKD-related complications, including hyperkalemia, metabolic acidosis, hyperphosphatemia, vitamin D deficiency, secondary hyperparathyroidism, and anemia, is also essential. Patients at high risk of CKD progression, indicated by an estimated GFR (eGFR) below 30 mL/min/1.73 m2, albuminuria exceeding 300 mg per 24 hours, or a rapid decline in eGFR, should be promptly referred to a nephrologist for specialized care.
CONCLUSIONS AND RELEVANCE
Timely diagnosis, accurate staging, and appropriate referral for CKD by primary care physicians are crucial steps in mitigating the global burden of this disease. By focusing on early detection and comprehensive management, primary care clinicians play a pivotal role in improving outcomes for patients with CKD worldwide.
Chronic kidney disease (CKD) is a widespread health problem, affecting 8% to 16% of people globally. Many patients and even clinicians may not recognize it early on.[1]–[4] CKD is characterized by a glomerular filtration rate (GFR) less than 60 mL/min/1.73 m2, albuminuria of 30 mg per 24 hours or more, or other signs of kidney damage (like blood or structural issues such as polycystic kidneys) that last for more than 3 months.[5] It’s more common in lower-income and middle-income countries than in wealthier ones.[6] Diabetes and hypertension are the leading causes of CKD worldwide. However, in Asia, sub-Saharan Africa, and many developing nations, other factors such as glomerulonephritis, infections, and environmental exposures (like air pollution, herbal remedies, and pesticides) are also significant contributors.[4] Genetic predisposition can also increase CKD risk. For example, sickle cell trait and specific APOL1 gene variants, more prevalent in people of African descent, can double the risk of developing CKD.[4],[7]–[10]
In the United States, GFR naturally declines by about 1 mL/min/1.73 m2 each year in the general population.[11],[12] The lifetime risk of developing a GFR below 60 mL/min/1.73 m2 is over 50%.[13] Early detection and treatment by primary care providers are vital because progressive CKD can lead to severe health issues, including end-stage kidney disease (ESKD), cardiovascular disease, and increased risk of death.[14]–[17] Current guidelines emphasize a risk-based approach to CKD evaluation and management.[5],[18]–[20] This review will discuss new risk calculators for predicting CKD progression (e.g., https://kidneyfailurerisk.com/) and focus on how primary care clinicians can diagnose, assess, and manage CKD. It will also cover when to refer patients to a nephrologist and when to consider dialysis.
Methods
We performed a literature search using Medline and PubMed up to April 2019. Search terms included CKD, chronic renal failure, chronic renal insufficiency, epidemiology, incidence, prevalence, occurrence, diagnosis, assessment, identification, screening, workup, etiology, causes, management, treatment, intervention, therapy, and prevention. We limited our search to English-language studies on humans from academic journals and guidelines. The initial search yielded 998 articles, including clinical trials, meta-analyses, practice guidelines, and systematic reviews. We expanded this to include review articles, observational studies (including cross-sectional studies), and more recent publications found in the references of the initial articles. All clinical trials on CKD treatment or prevention were included, regardless of study size or patient age.
Clinical Presentation
Often, chronic kidney disease is found during routine blood and urine tests or incidentally during evaluations for other conditions. Less frequently, patients might present with symptoms like visible blood in the urine, “foamy urine” (indicating albuminuria), frequent nighttime urination, flank pain, or reduced urine output. In advanced CKD, patients may experience fatigue, poor appetite, nausea, vomiting, a metallic taste, unintentional weight loss, itching, mental status changes, shortness of breath, or swelling in the extremities.[21]
When evaluating a patient with known or suspected CKD, clinicians should ask about other symptoms that could suggest an underlying systemic illness (e.g., coughing up blood, rash, swollen lymph nodes, hearing loss, nerve damage) or urinary obstruction (e.g., difficulty starting urination, urgency, frequency, or feeling of incomplete bladder emptying).[21] Additionally, assess patients for kidney disease risk factors, such as exposure to nephrotoxins (NSAIDs, phosphate-based bowel preps, herbal remedies like those with aristolochic acid, antibiotics like gentamicin, and chemotherapy drugs), history of kidney stones or recurrent urinary tract infections, presence of other conditions (hypertension, diabetes, autoimmune diseases, chronic infections), family history of kidney disease, and known genetic risks like sickle cell trait.[9],[18],[21]–[24]
A thorough physical exam can offer more clues about the cause of CKD. This should include checking the patient’s fluid balance. Signs of dehydration might indicate poor fluid intake, vomiting, diarrhea, or excessive diuretic use. Conversely, fluid overload could be due to heart failure, liver failure, or nephrotic syndrome. Retinal exam findings like arterial-venous nicking or retinopathy suggest long-standing hypertension or diabetes. Carotid or abdominal bruits might point to renovascular disease. Flank pain or enlarged kidneys should raise suspicion of urinary obstruction, kidney stones, pyelonephritis, or polycystic kidney disease. Neuropathy can be caused by diabetes, vasculitis, or amyloidosis. Skin findings may include rashes (systemic lupus erythematosus, acute interstitial nephritis), palpable purpura (Henoch-Schonlein purpura, cryoglobulinemia, vasculitis), telangiectasias (scleroderma, Fabry disease), or extensive skin thickening (scleroderma). Patients with advanced CKD may appear pale, have skin abrasions, muscle wasting, asterixis, muscle twitching, altered mental status, and pericardial rub.[21]
CKD Definition and Staging
Chronic kidney disease is defined by kidney structure or function abnormalities lasting more than 3 months.[5],[25] This includes one or more of the following: (1) GFR less than 60 mL/min/1.73 m2; (2) albuminuria (urine albumin ≥30 mg per 24 hours or urine albumin-to-creatinine ratio [ACR] ≥30 mg/g); (3) abnormal urine sediment, histology, or imaging suggestive of kidney damage; (4) renal tubular disorders; or (5) a history of kidney transplant.[5] If the duration of kidney disease is unclear, repeat tests to differentiate CKD from acute kidney injury (kidney function change within 2–7 days) and acute kidney disease (kidney damage or decreased function present for ≤3 months).[25] Determining the cause of CKD involves considering the patient’s history, physical exam, and urine findings (Figure 1).[5],[18],[21]
Figure 1. Considerations for Diagnosis, Staging, and Referral of Patients With Chronic Kidney Disease.
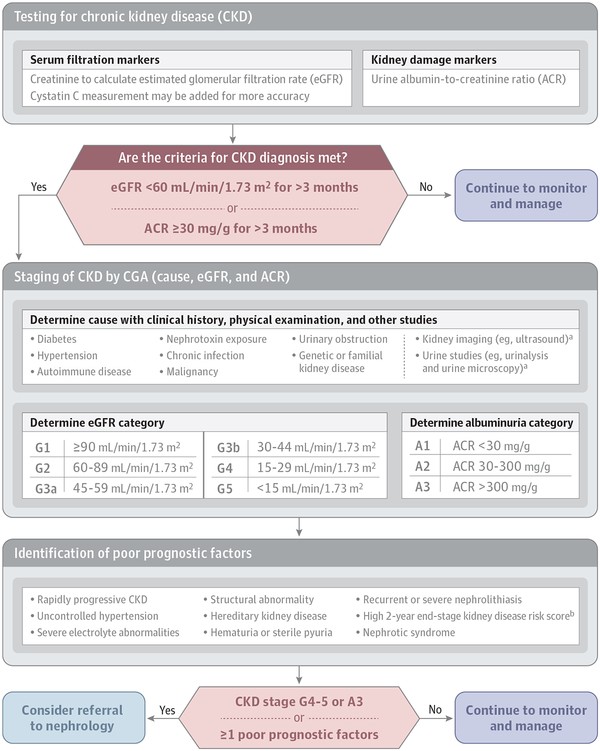 Figure 1. Diagnosis, Staging, and Referral Considerations for Chronic Kidney Disease Patients.
Figure 1. Diagnosis, Staging, and Referral Considerations for Chronic Kidney Disease Patients.
a Other imaging or urine tests may be considered.
b Risk scores are available, e.g., https://kidneyfailurerisk.com/.
Once CKD is diagnosed, the next step is staging based on GFR, albuminuria, and the underlying cause (Figure 2).[5] GFR staging is categorized as G1 (GFR ≥90 mL/min/1.73 m2), G2 (GFR 60–89 mL/min/1.73 m2), G3a (45–59 mL/min/1.73 m2), G3b (30–44 mL/min/1.73 m2), G4 (15–29 mL/min/1.73 m2), and G5 (<15 mL/min/1.73 m2).[5] While GFR can be directly measured using agents like iohexol or iothalamate,[26]–[28] estimating equations (e.g., the CKD-EPI and MDRD equations) have largely replaced direct measurement in clinical practice.[29]–[31] Labs now routinely report estimated GFR (eGFR) based on filtration markers. Creatinine, a byproduct of creatine metabolism, is the most common marker.[25] Laboratory assays for creatinine have been standardized since 2003.[32] The CKD-EPI 2009 creatinine equation is the preferred estimating equation in the US and globally, as it’s more accurate than the MDRD equation, especially for eGFR values above 60 mL/min/1.73 m2 (https://www.kidney.org/professionals/kdoqi/gfr_calculator).[29],[30] For increased accuracy, cystatin C can be combined with creatinine in the CKD-EPI 2012 creatinine-cystatin C equation.[31] Cystatin C can be particularly useful in individuals with factors affecting creatinine production or metabolism (e.g., extreme body size or muscle mass, limb amputation, high-protein diet, creatinine supplements, or drugs affecting creatinine secretion).[5],[25]
Figure 2. KDIGO 2012 Definition and Prognosis of Chronic Kidney Disease by GFR and Albuminuria Categories.
GFR indicates glomerular filtration rate; KDIGO, Kidney Disease Improving Global Outcomes. Risk of progression includes CKD progression, defined by a decline in GFR category (with ≥25% decrease in eGFR from baseline) or sustained eGFR decline >5 mL/min/1.73 m2 per year. Green: low risk; yellow: moderately increased risk; orange: high risk; red: very high risk. Reproduced with permission from Kidney International Supplements.[5]
Albuminuria should be quantified using a urine ACR. Albuminuria stages are A1 (ACR <30 mg/g), A2 (30–300 mg/g), and A3 (>300 mg/g).[5] Guidelines recommend urine ACR over urine protein-to-creatinine ratio for CKD staging because ACR assays are more standardized and precise at lower albuminuria levels.[5],[33] First morning or 24-hour urine samples provide the most accurate measurements due to daily variability in urine albumin excretion.[5],[34],[35] However, random samples are acceptable for initial screening.[5] Urine ACR is considered a more sensitive and specific marker of glomerular disease than urine protein-to-creatinine ratio.[5] Some urine proteins, like uromodulin, are normally present and may even be protective.[36]–[38] If tubular or overflow proteinuria is suspected, urine protein electrophoresis or specific protein testing (e.g., immunoglobulin light chains, α1-microglobulin, and β2-microglobulin) can be performed.[5] Kidney ultrasound imaging to assess morphology and rule out urinary obstruction should be considered in all CKD diagnoses.[5]
Determining the cause of CKD can be complex but is generally classified by the presence of systemic disease and the location of the anatomical abnormality. Systemic diseases include diabetes, autoimmune disorders, chronic infections, malignancy, and genetic disorders affecting multiple organs. Anatomic locations include glomerular, tubulointerstitial, vascular, and cystic/congenital diseases.[5] Cause identification can significantly impact prognosis and treatment. For example, polycystic kidney disease may progress to ESKD faster and often requires evaluation for extrarenal manifestations and specific therapies like tolvaptan, a vasopressin V2 receptor antagonist that slows GFR decline.[39],[40] Patients with unexplained CKD should be referred to a nephrologist.
Screening for CKD
Since most CKD patients are asymptomatic, screening can be crucial for early detection.[18] The National Kidney Foundation offers a kidney profile test measuring serum creatinine for eGFR and urine ACR.[41] Many guidelines recommend a risk-based screening approach, particularly for those over 60 or with diabetes or hypertension.[18]–[20] Screening is also advised for individuals with clinical risk factors like autoimmune disease, obesity, kidney stones, recurrent UTIs, reduced kidney mass, exposure to certain medications (NSAIDs, lithium), and prior acute kidney injury (Box).[9],[18],[42]–[45] However, no randomized trials have yet proven that screening asymptomatic individuals for CKD improves outcomes.
Box. Clinical, Sociodemographic, and Genetic Risk Factors for Chronic Kidney Disease.
Clinical
Diabetes
Hypertension
Autoimmune diseases
Systemic infections (e.g., HIV, hepatitis B, hepatitis C)
Nephrotoxic medications (e.g., NSAIDs, herbal remedies, lithium)
Recurrent urinary tract infections
Kidney stones
Urinary tract obstruction
Malignancy
Obesity
Reduced kidney mass (e.g., nephrectomy, low birth weight)
History of acute kidney injury
Smoking
Intravenous drug use (e.g., heroin, cocaine)
Family history of kidney disease
Sociodemographic
Age >60 years
Nonwhite race
Low income
Low education
Genetic
APOL1 risk alleles
Sickle cell trait and disease
Polycystic kidney disease
Alport syndrome
Congenital anomalies of the kidney and urinary tract
Other familial causes
Other Risk Factors for CKD
Sociodemographic factors also increase CKD risk, including nonwhite race, low education, low income, and food insecurity.[18],[43],[46] African Americans and Pacific Islanders face a significantly higher risk of ESKD compared to whites.[47] This is partly due to higher rates of hypertension, diabetes, and obesity.[11] Genetic factors also play a role. Specifically, APOL1 gene risk variants can increase kidney disease risk in a recessive manner.[7],[8] Individuals with two APOL1 risk alleles (about 13% of African Americans) have a doubled risk of CKD progression and up to a 29-fold increased risk for specific CKD types (e.g., focal-segmental glomerulosclerosis and HIV-associated nephropathy) compared to those with fewer or no risk alleles.[11],[44],[45],[48],[49] Sickle cell trait (present in about 8% of African Americans) is also linked to increased kidney disease risk. Compared to noncarriers, individuals with sickle cell trait have a 1.8-fold higher odds of developing CKD, 1.3-fold higher odds of eGFR decline greater than 3 mL/min/1.73 m2, and 1.9-fold higher odds of albuminuria.[9]
Management of Patients With CKD
Reducing Risk of Cardiovascular Disease
Cardiovascular disease is significantly more common in people with CKD. In a Medicare study, 65% of older adults with CKD had cardiovascular disease, compared to 32% without CKD.[47] CKD also worsens cardiovascular outcomes. In the same study, CKD was associated with lower 2-year survival rates in people with coronary artery disease, acute myocardial infarction, heart failure, atrial fibrillation, and stroke/transient ischemic attack.[47]
Therefore, reducing cardiovascular risk is a key part of CKD management. For CKD patients aged 50 and older, low- to moderate-dose statins are recommended, regardless of LDL cholesterol levels.[50]–[52] Smoking cessation should also be strongly encouraged.[5],[53] Guidelines from JNC 8 and KDIGO recommend target blood pressures below 140/90 mm Hg for adults with CKD, based on expert opinion.[5],[54] KDIGO further suggests a lower target of below 130/80 mm Hg for adults with urine ACR of 30 mg per 24 hours or more.[5] The SPRINT trial showed that intensive blood pressure control (systolic BP <120 mm Hg) in high-cardiovascular-risk individuals without diabetes reduced major cardiovascular events by 25% and all-cause mortality by 27% compared to standard control (systolic BP <140 mm Hg).[55] The intensive treatment group had a higher risk of eGFR decline, but this might be due to hemodynamic changes rather than actual kidney damage.[55],[56] Importantly, the cardiovascular benefits of intensive blood pressure control were similar in participants with and without CKD at baseline.[57]
Management of Hypertension
Guidelines provide algorithms for hypertension treatment in CKD patients.[54],[58] Albuminuria presence and severity should be assessed. Renin-angiotensin-aldosterone system blockade with an ACEI or ARB is recommended for adults with diabetes and urine ACR ≥30 mg per 24 hours, or any adult with urine ACR ≥300 mg per 24 hours.[5],[18],[58] Combining ACEIs and ARBs is generally avoided due to risks of hyperkalemia and acute kidney injury.[5],[18],[59] Aldosterone receptor antagonists can be considered for patients with albuminuria, resistant hypertension, or heart failure with reduced ejection fraction.[58],[60]–[64]
Management of Diabetes Mellitus
Optimal diabetes management is also crucial. Good glycemic control can slow CKD progression, with a recommended HbA1c target of around 7.0%.[5],[18],[19],[65]–[67] Secondly, oral hypoglycemic agent doses may need adjustment. Drugs primarily cleared by the kidneys (e.g., glyburide) should generally be avoided. Drugs metabolized by the liver or partially excreted by kidneys (e.g., metformin, some DPP-4 and SGLT-2 inhibitors) may require dose reduction or discontinuation, especially when eGFR drops below 30 mL/min/1.73 m2.[18],[19] Thirdly, SGLT-2 inhibitors should be considered for patients with severely increased albuminuria. The CREDENCE trial showed that canagliflozin reduced the risk of renal outcomes (doubling of serum creatinine, ESKD, or renal/cardiovascular death) by 30% in type 2 diabetes patients with CKD (stage G2-G3/A3) already on ACEI or ARB therapy.[68] Previous trials also suggest cardiovascular benefits with SGLT-2 inhibitors, potentially extending to CKD patients with lower albuminuria levels.[69],[70]
Nephrotoxins
All CKD patients should avoid nephrotoxins. While a comprehensive list is extensive, some key examples include NSAIDs, which are generally not recommended in CKD, especially when patients are on ACEIs or ARBs.[5],[18] Herbal remedies are unregulated and some (e.g., those containing aristolochic acid or anthraquinones) can cause kidney damage.[22] Phosphate-based bowel preparations (oral and enema forms) can lead to acute phosphate nephropathy.[23],[24] Proton pump inhibitors (PPIs) have been linked to acute interstitial nephritis and incident CKD in studies.[71]–[73] While routine PPI discontinuation in CKD isn’t necessary, the indication for their use should be reviewed regularly.
Drug Dosing
Drug dose adjustments are frequently needed in CKD. The CKD-EPI equation for eGFR estimation is likely more accurate for drug clearance than the traditional Cockcroft-Gault equation.[74],[75] Common medications requiring dose reduction include most antibiotics, direct oral anticoagulants, gabapentin/pregabalin, oral hypoglycemic agents, insulin, chemotherapy drugs, and opiates.[5],[18] In general, medications with limited benefit should be minimized in CKD patients due to their higher risk of adverse drug events.[76]–[79] Gadolinium-based contrast agents are contraindicated in acute kidney injury, eGFR below 30 mL/min/1.73 m2, or ESKD due to the risk of nephrogenic systemic fibrosis, a debilitating fibrotic disorder.[5],[18],[80],[81] Newer macrocyclic chelate formulations (e.g., gadoteridol, gadobutrol, gadoterate) have a lower risk, but avoiding gadolinium altogether is best. If gadolinium is essential, patients must be informed of the risk, and nephrologist consultation for post-exposure hemodialysis may be considered.[5],[18],[80]–[82]
Dietary Management
Dietary management for CKD progression is debated, with mixed trial results.[83]–[85] The MDRD study found low-protein diets slowed GFR decline only initially, and very-low-protein diets showed no significant additional benefit. However, protein restriction appeared beneficial in patients with proteinuria >3 g/day.[83] Smaller trials suggest protein restriction may help prevent CKD progression or ESKD.[86]–[88] KDIGO recommends reducing protein intake to less than 0.8 g/kg per day for CKD stages G4-G5 and less than 1.3 g/kg per day for other CKD patients at progression risk, with proper nutritional education.[5] Benefits of protein restriction must be weighed against malnutrition and protein wasting risks.[5],[83],[84],[89] Lower dietary acid loads (more fruits/vegetables, less meats/eggs/cheeses) may also be protective.[90],[91] Low-sodium diets (<2 g/day) are recommended for hypertension, proteinuria, or fluid overload.[5]
Monitoring of Established CKD and Treatment of Complications
Once CKD is diagnosed, KDIGO recommends monitoring eGFR and albuminuria at least annually. High-risk patients should be monitored at least twice yearly, and very-high-risk patients at least three times yearly (Figure 2).[5] Moderate to severe CKD increases the risk of electrolyte imbalances, mineral and bone disorders, and anemia.[92] Screening and monitoring frequency for lab abnormalities depend on CKD stage and include complete blood count, basic metabolic panel, serum albumin, phosphate, parathyroid hormone, 25-hydroxyvitamin D, and lipid panel (Table).[5],[50],[93],[94]
Table. Screening, Monitoring, and Management of the Complications of Chronic Kidney Disease (CKD).
| Complication | Relevant Tests | Frequency of Repeat Testing | Management |
|---|---|---|---|
| Anemia | Hemoglobin | No anemia: CKD stages G1-G2: when clinically indicated; CKD stage G3: at least once per year; CKD stages G4-G5: at least twice per year. With anemia: CKD stages 3–5: at least every 3 months | Rule out other causes of anemia: iron deficiency, vitamin B12 deficiency, folate deficiency, occult bleeding. Consider iron supplementation and nephrologist referral for erythropoietin-stimulating agent therapy when hemoglobin <10 g/dL |
| Mineral and bone disorder | Serum calcium, phosphate, parathyroid hormone, 25-hydroxyvitamin D | Calcium/phosphate: CKD stage G3: every 6–12 months; CKD stage G4: every 3–6 months; CKD stage G5: every 1–3 months. Parathyroid hormone: CKD stage G3: at baseline, then as needed; CKD stage G4: every 6–12 months; CKD stage G5: every 3–6 months. Vitamin D: CKD stages 3–5: at baseline, then as needed | Consider phosphate-lowering therapy (e.g., calcium acetate, sevelamer, iron-based binders) and vitamin D supplementation |
| Hyperkalemia | Serum potassium | At baseline and as needed | Low-potassium diet, correction of hyperglycemia and acidemia, consider potassium binders |
| Metabolic acidosis | Serum bicarbonate | At baseline and as needed | Oral bicarbonate supplementation (e.g., sodium bicarbonate, baking soda, or sodium citrate/citric acid) for values persistently <22 mmol/L |
| Cardiovascular disease | Lipid panel | At baseline and as needed | Low- to moderate-dose statin therapy for patients aged ≥50 years with CKD. Statin therapy for patients aged 18–49 years with CKD and coronary artery disease, diabetes, prior ischemic stroke, or high risk of myocardial infarction or cardiovascular death |
Anemia and the Role of Erythropoietin in CKD
Anemia is a common CKD complication. In a study of 19 CKD cohorts, 41% of individuals had low hemoglobin levels.[92] Initial anemia workup should assess iron stores. Iron-deficient patients may benefit from iron supplementation. Patients with persistently low hemoglobin (<10 g/dL) despite addressing reversible causes can be referred to a nephrologist for erythropoietin-stimulating agent therapy. However, these agents carry risks of death, stroke, and venous thromboembolism, which must be weighed against potential benefits.[93]
Electrolyte, Mineral, and Bone Abnormalities in CKD
Electrolyte abnormalities occur in 3% to 11% of CKD patients.[92] Initial management involves dietary changes and supplements. Low-potassium diets are recommended for hyperkalemia, and low-phosphorus diets for hyperphosphatemia.[5],[18],[94],[95] For persistent low serum bicarbonate (<22 mmol/L), oral bicarbonate supplementation should be considered, as chronic metabolic acidosis may accelerate CKD progression.[5],[18],[96]–[99]
Mineral and bone disorders are also common. In a study of CKD patients, 58% had elevated parathyroid hormone levels.[92] While the optimal level is unclear, addressing hyperphosphatemia, hypocalcemia, and vitamin D deficiency is generally recommended, using low-phosphate diets, phosphate binders, adequate calcium intake, and vitamin D supplementation (Table).[94],[95]
Prognosis of CKD
ESKD incidence varies by risk factors and location. In North America, incidence in eGFR <60 mL/min/1.73 m2 ranged from 4.9 to 168.3 ESKD events per 1000 patient-years; in non-North American cohorts, it ranged from 1.2 to 131.3 events per 1000 patient-years.[100] Most CKD patients do not require kidney replacement therapy.[101] Online tools like the Kidney Failure Risk Equation (KFRE; https://kidneyfailurerisk.com/) predict 2-year and 5-year dialysis/transplant risk in eGFR <60 mL/min/1.73 m2 patients.[100],[102] Validated in over 700,000 individuals across 30+ countries, KFRE uses clinical and lab variables. The 4-variable equation includes age, sex, eGFR, and urine ACR. The 8-variable equation adds serum albumin, phosphate, calcium, and bicarbonate.[100],[102] KFRE implementation has been tested in healthcare systems; referrals based on a 5-year KFRE >3% reduced wait times,[103] and a 2-year KFRE >10% guided referrals to multidisciplinary CKD clinics.[104] A trial is ongoing to evaluate if a KFRE risk-based approach improves CKD management.[105] For eGFR <30mL/min/1.73m2, the CKD G4+ risk calculator (https://www.kdigo.org/equation/) can provide additional cardiovascular and mortality risk information.[106],[107] Risk prognostication can help identify high-risk patients and reassure those with mild CKD like stage G3a A1.
Referral to a Nephrologist and Timing of Kidney Replacement Therapy
KDIGO guidelines recommend nephrologist referral for CKD when eGFR falls below 30 mL/min/1.73 m2 (stage G4) and/or urine ACR exceeds 300 mg per 24 hours (stage A3).[5] Albuminuria >2200 mg per 24 hours warrants expedited nephrologist evaluation for possible nephrotic syndrome. Other referral indications include unexplained >20 red blood cells per high-power field, red blood cell casts, uncontrolled hypertension despite ≥4 medications, persistent hypo/hyperkalemia, anemia requiring erythropoietin, recurrent kidney stones, hereditary kidney disease, acute kidney injury, and rapid CKD progression (eGFR decrease ≥25% or sustained decline >5 mL/min/1.73 m2).[5] Even small serum creatinine changes (e.g., 0.7 to 1.2 mg/dL) in non-CKD individuals indicate significant eGFR decline, requiring primary care clinicians to identify reversible causes. Kidney biopsy indications include unexplained persistent/increasing albuminuria, cellular casts or dysmorphic red blood cells, and unexplained/rapid GFR decline.[5] Specific thresholds vary. Polycystic kidney disease, certain glomerulonephritis types, and nephrotic-range albuminuria carry a particularly high ESKD progression risk.[5],[39],[102]
Nephrology referral is crucial for kidney replacement therapy planning and transplant evaluation. Dialysis initiation is based on symptoms, not solely GFR level.[108] Urgent indications include encephalopathy, pericarditis, and pleuritis due to severe uremia.[109] Otherwise, dialysis is considered for uremic symptoms (nausea, vomiting, poor appetite, metallic taste, pericardial rub/effusion, asterixis, altered mental status), electrolyte imbalances (hyperkalemia, metabolic acidosis), or fluid overload refractory to medical management.[5],[18],[109] A shared decision-making approach is best. Patients should be educated about treatment options and actively involved in decisions. Early education should cover CKD complications and kidney replacement therapy modalities. Kidney transplantation is the optimal ESKD therapy, with living donor transplants pre- or shortly post-dialysis initiation having the best outcomes.[110],[111] Early referral (eGFR <30 mL/min/1.73 m2 and elevated 2-year ESKD risk) for transplant evaluation is important.[112],[113] ESKD alternative therapies include in-center hemodialysis, home hemodialysis, peritoneal dialysis, or conservative care without dialysis.[107] Patient preference should guide dialysis modality choice, although peritoneal dialysis may be unsuitable for those with abdominal scarring or unstable housing.[107],[109] Patients planning hemodialysis with rapid eGFR decline should be referred for arteriovenous fistula placement when eGFR is 15-20 mL/min/1.73 m2, per KDOQI guidelines.[114] Dialysis initiation in older patients with poor functional status has been linked to accelerated functional decline and high short-term mortality.[115],[116] Patient preferences for conservative medical management should be discussed and respected.
Conclusions
Chronic kidney disease affects a significant portion of the global population and is a leading cause of death. Optimal CKD management involves cardiovascular risk reduction, albuminuria treatment, nephrotoxin avoidance, and drug dose adjustments. Monitoring for CKD complications like hyperkalemia, metabolic acidosis, anemia, and other metabolic abnormalities is also essential. Primary care clinicians play a vital role in reducing the global CKD burden through early diagnosis, proper staging, and appropriate referral.
Funding/Support:
Dr. Chen was supported by a Clinician Scientist Career Development Award from Johns Hopkins University and is supported by a George M. O’Brien Center for Kidney Research Pilot and Feasibility Grant from Yale University and award K08DK117068 from the National Institutes of Health/NIDDK. Dr. Grams is supported by NIDDK grants DK1008803, DK100446, and DK115534.
Role of the Funder/Sponsor: The supporting institutions had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; or decision to submit the manuscript for publication.
Footnotes
Conflict of Interest Disclosures: Dr. Chen reported receipt of grants from the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) and Yale University. Dr. Grams reported receipt of grants from the NIDDK and the National Kidney Foundation and travel support from Dialysis Clinics Inc. for an invited speakership at a directors’ meeting in May 2019. No other disclosures were reported.
Submissions: We encourage authors to submit papers for consideration as a Review. Please contact Edward Livingston, MD, at Edward. [email protected] or Mary McGrae McDermott, MD, at [email protected].
