Hepatic Haemangioma
Hepatic haemangioma is the most common benign liver tumor, with a prevalence ranging from 3% to 20%.1, 19, 20 These lesions are typically solitary and small, but can grow up to 20 cm in diameter.1, 20 Most patients remain asymptomatic, and haemangiomas are often discovered incidentally during imaging for other reasons. They are more frequently diagnosed in women aged 30 to 50, with a female to male ratio of 3:1.20 The exact cause of haemangiomas is not fully understood, but they are considered congenital, with some evidence suggesting hormonal influence.21 Histologically, they consist of large, blood-filled channels lined by mature endothelial cells within a fibrous stroma. Haemangiomas are benign and generally do not cause problems, although they may enlarge slightly during pregnancy or with estrogen therapy.1, 21 Spontaneous bleeding is extremely rare and should prompt consideration of alternative diagnoses in the Liver Mass Differential Diagnosis. Giant haemangiomas can become symptomatic due to infarction or thrombosis. In rare instances, a haemangioma can lead to Kasabach-Merritt syndrome, characterized by thrombocytopenia, consumptive coagulopathy, and microangiopathic hemolytic anaemia.
Ultrasound typically reveals a well-defined, hyperechoic lesion. Contrast-enhanced ultrasound demonstrates initial peripheral globular-nodular enhancement followed by centripetal fill-in, a key feature in the liver mass differential diagnosis.6 MRI is highly accurate in diagnosing haemangiomas, boasting 100% sensitivity and 95% specificity. The classic MRI appearance includes a hypointense lesion on T1-weighted sequences and a strongly hyperintense lesion on T2-weighted sequences. After contrast administration, a characteristic globular peripheral enhancement with progressive centripetal filling is observed (figure 1).22, 23 Management is usually symptomatic and conservative.1, 20
Figure 1.
MRI showing focal nodular hyperplasia (FNH) of the liver. (A) Axial T2-weighted image revealing a large liver mass in the left lobe, isointense to the liver parenchyma, with a hyperintense central scar, important for liver mass differential diagnosis. (B) Unenhanced axial T1-weighted image showing the mass as slightly hypointense and the central scar more hypointense, crucial details for liver mass differential diagnosis. (C) Gadolinium-enhanced T1-weighted image during the arterial phase showing intense and homogeneous enhancement of the mass, except for the central scar, aiding in liver mass differential diagnosis.
Focal Nodular Hyperplasia
Focal nodular hyperplasia (FNH) is the second most common benign liver tumor, with an estimated prevalence of 2.5% in the general population. It is predominantly diagnosed in women, with peak incidence between 30 and 50 years of age, making it an important consideration in liver mass differential diagnosis for this demographic. In most cases, FNH is solitary and smaller than 5 cm, but it can be larger and multiple in about 20% of cases. Usually, it is an incidental finding in asymptomatic individuals. FNH is believed to be a hyperplastic response to an abnormal artery within the liver.1, 24 Histologically, it consists of normal hepatocytes arranged in nodules with prominent bile ductular reaction at the nodule margins.24 A characteristic central fibrotic scar containing a feeding artery is often present24 and is a key feature used in CT or MRI diagnosis, particularly when biopsy is avoided. At a molecular level, FNH is polyclonal and shows activation of the β-catenin pathway without β-catenin mutation. Consequently, glutamine synthetase staining, a target gene of β-catenin, is frequently positive in typical FNH, a useful marker in liver mass differential diagnosis.24
On MRI, FNH typically appears hypointense or isointense on T1-weighted images and slightly hyperintense or isointense on T2-weighted images. The central scar is hypointense on T1-weighted images and hyperintense on T2-weighted images. Following contrast administration, FNH demonstrates intense, homogeneous enhancement in the arterial phase, sparing the central scar. In the portal venous and delayed phases, it becomes isointense to the liver parenchyma, with enhancement of the central scar25 (figure 1). Clinically, FNH has a benign course without severe complications. Therefore, treatment is generally not recommended, emphasizing the importance of accurate liver mass differential diagnosis to avoid unnecessary interventions.1, 24
Hepatocellular Adenoma
Hepatocellular adenoma (HCA) is a rare liver tumor (prevalence 0.001%) more commonly found in young women. Its occurrence is associated with oral contraceptive use, anabolic steroid treatment, and glycogen storage diseases types I and III, making patient history crucial in liver mass differential diagnosis. In most cases, HCA presents as a solitary lesion, but multiple adenomas can occur in 10%–20% of patients, raising suspicion for hepatic adenomatosis due to genetic abnormalities.26 HCA is composed of normal hepatocytes without atypia, arranged in plates separated by dilated sinusoids, lacking portal spaces or bile ducts.1 About 25% of patients with HCA experience mild abdominal pain. The most significant complication is necrosis and bleeding, which can lead to severe hemoperitoneum. The risk of bleeding is increased with pregnancy, prolonged oral contraceptive use, larger lesions, and subcapsular location, factors that need to be considered in liver mass differential diagnosis and management.
Molecular analysis has revealed that HCA is a heterogeneous entity. Genotype classification has identified three main subtypes: HNF1A-mutated HCA (H-HCA) in 35% of cases, β-catenin-mutated HCA (b-HCA) in 10%, and inflammatory HCA (IHCA) in 55%. Key molecular, clinical, and radiological features are summarized in table 1, aiding in the liver mass differential diagnosis. Malignant transformation is a concern in a small percentage of patients (approximately 1%–5%), with recent studies suggesting a higher risk in β-catenin-activated HCAs.24 Unfortunately, no reliable clinical or radiological signs can predict malignant degeneration preoperatively, highlighting the challenges in liver mass differential diagnosis and risk stratification.
Table 1.
Molecular, clinical, and radiological characteristics of Hepatocellular Adenoma (HCA) subtypes, essential for liver mass differential diagnosis.
| Subtype | Mutated gene | Immuno-histochemistry | Clinical features | Radiological features |
|---|---|---|---|---|
| H-HCA (HNF1α inactivated) | TCF1 | LFABP − | – Steatotic nodules – Microadenomas – Additional nodules | – Homogeneous signal dropout on out-of-phase chemical shift images – Iso or slight hypersignal on T2W images – Moderate arterial enhancement – No persistent enhancement in venous phases |
| b-HCA (β-catenin activated) | CTNNB1 | β-catenin + | Risk of malignant transformation | – No signal dropout on out-of-phase chemical shift sequences – Strong arterial enhancement – Delayed phase: persistent enhancement versus washout |
| GS + | ||||
| IHCA* (inflammatory) | IL6ST | SAA + | – Inflammatory infiltrate | – No signal dropout on out-of-phase chemical shift sequences |
| CRP + | – Sinusoidal dilation – Obesity – Steatosis (non-tumoral liver) – Peliosis | – Marked hypersignal on T2W images (stronger in outer part of the lesions) – Strong arterial enhancement – Persistent enhancement in venous phases | ||
| Unclassified HCA | – | – | – | – |
*Around 10% of IHCA present β-catenin activation (b-IHCA).
CRP, C-reactive protein; CTNNB1, catenin β-1; GS, glutamine synthetase; HCA, hepatocellular adenoma; HNF1α, hepatocyte nuclear factor 1α; IHCA, inflammatory HCA; IL6ST, interleukin 6 signal transducer; LFABP, liver fatty acid binding protein; SAA, serum amyloid A; TCF1, transcription factor 1.
Distinguishing between HCA and FNH can be challenging, even with advanced imaging and pathological examination, which complicates the liver mass differential diagnosis. HCA can exhibit variable signal intensity based on tissue components. On T1-weighted images, lesions are often heterogeneous and may appear homogeneously or heterogeneously hyperintense on T2-weighted images. A peripheral rim, representing a fibrous capsule, may be visible. Rapid contrast uptake is seen during the arterial phase, with the lesion becoming isointense to liver tissue in delayed sequences.27 MRI with liver-specific contrast agents, such as gadoxetic acid, can aid in liver mass differential diagnosis, as FNH typically appears iso-hyperintense in the hepatobiliary phase, while HCA is usually hypointense.28 In some cases, biopsy is necessary for definitive characterization. However, even with pathological analysis, differentiating HCA from FNH may be impossible in certain cases, highlighting the inherent difficulties in liver mass differential diagnosis.
Management of hepatic adenomas depends on symptoms, size, number of lesions, and the risks of bleeding, rupture, and malignant transformation. Some guidelines suggest that small lesions (29
Nodular Regenerative Hyperplasia
Nodular regenerative hyperplasia (NRH) of the liver is a benign condition characterized by the widespread regeneration of hepatocytes with minimal fibrosis, replacing normal liver tissue.30 Various factors, primarily vascular damage, have been linked to NRH development, making vascular history relevant in liver mass differential diagnosis. The pathogenesis of NRH is not fully understood, but it is hypothesized that sinusoidal portal venous hypertension, often due to thrombosis from endothelial injury, hypercoagulability, or autoimmune issues, is a key initiating event. This leads to centrolobular hepatocyte atrophy and compensatory proliferation of portal hepatocytes, forming regenerative nodules.31 Most patients with NRH are asymptomatic, although some may present with portal hypertension signs. Imaging findings in diffuse NRH can be subtle, with widespread nodularity on ultrasound. In focal NRH, CT or MRI may show multiple hypervascular masses, potentially mimicking metastases or hepatocellular carcinoma (HCC), making liver mass differential diagnosis crucial. However, stability on follow-up imaging in patients with conditions like long-standing Budd-Chiari syndrome can aid in correct diagnosis.32 Biopsy is usually required for a definitive NRH diagnosis. Microscopically, nodules are clustered around portal triads, with hypertrophied hepatocytes in the nodule center and thin, atrophic peripheral cells with sinusoidal dilatation. Prognosis is mainly determined by the development and progression of portal hypertension. Treatment for NRH focuses on managing the underlying medical condition and preventing portal hypertension complications, emphasizing the importance of accurate liver mass differential diagnosis to guide appropriate management.
Hepatic Metastasis
Liver metastases are the most common malignant liver tumors, originating from cancers in other organs, most frequently the lung, colon, stomach, pancreas, gallbladder, breast, and ovaries.33 Metastatic liver involvement typically indicates a poor prognosis. Identifying the primary tumor and obtaining biopsy confirmation are important, especially if the patient may benefit from therapies like surgery or systemic chemotherapy, underscoring the importance of liver mass differential diagnosis in cancer management.33 Fine-needle aspiration biopsy has a high diagnostic sensitivity (85%) and specificity (over 95%). Serum biomarkers can be useful in monitoring some tumors after treatment. However, in the context of a focal liver lesion without a known primary cancer, serum biomarkers lack specificity, although they can sometimes suggest the primary origin, contributing to the liver mass differential diagnosis process. On CT or MRI, liver metastases are usually hypovascular lesions, often showing peripheral rim enhancement. However, some cancers may exhibit different patterns. Arterial phase contrast uptake on CT or MRI may suggest neuroendocrine tumors, melanoma, sarcoma, hypernephroma, or thyroid cancer.9 Isotopic studies using labeled somatostatin analogues can help identify neuroendocrine tumors. Surgical resection of liver metastases can improve survival in patients with colorectal cancer,34 neuroendocrine tumors,35 and certain renal carcinomas, but its role in other cancers remains controversial, highlighting the complexity of liver mass differential diagnosis and treatment planning in metastatic disease.
Hepatocellular Carcinoma
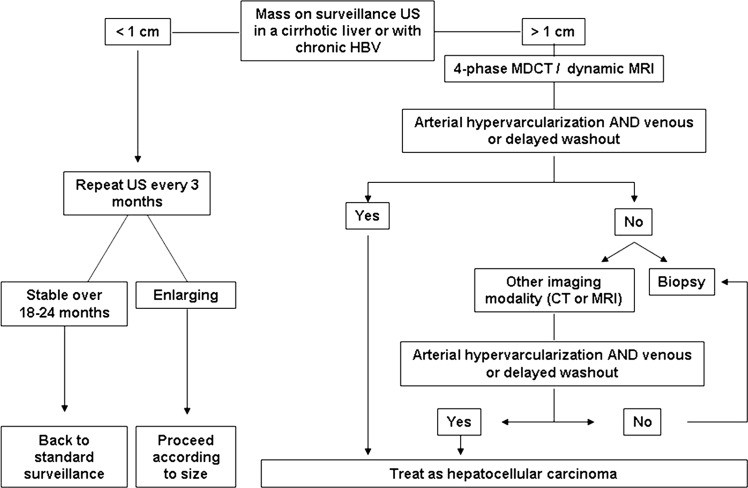
Hepatocellular carcinoma (HCC) is the sixth most common cancer worldwide and the third leading cause of cancer-related deaths.36 It is currently considered the primary cause of death in patients with cirrhosis.5 HCC typically develops in the setting of chronic liver disease, with cirrhosis being the strongest predisposing factor, making risk factors crucial in liver mass differential diagnosis. HCC incidence varies geographically, reflecting the distribution of its main causes. In Asia and sub-Saharan Africa, where incidence exceeds 20–30 cases per 100,000 population, hepatitis B virus (HBV) infection is the predominant risk factor, further increased by aflatoxin B1-contaminated food. In developed countries with lower HCC incidence, hepatitis C virus (HCV) or alcohol-related cirrhosis are more common predisposing factors.37 Surveillance programs aim to reduce HCC-related mortality and are recommended for high-risk patients, typically involving abdominal ultrasound every 6 months.3, 4, 38 When a liver nodule is detected, prompt evaluation is necessary due to the high likelihood of HCC. In cirrhotic patients, HCC diagnosis can often be established by imaging for nodules larger than 1 cm (figure 2).3 Non-invasive diagnostic criteria rely on dynamic imaging techniques to identify a specific vascular pattern: intense and homogeneous arterial phase contrast uptake followed by venous phase washout (figure 3). When this characteristic vascular pattern is absent or the nodule arises in a non-cirrhotic liver, biopsy is recommended for definitive liver mass differential diagnosis. These non-invasive criteria have been prospectively validated for high specificity, but sensitivity for nodules 39–41 However, biopsy of these nodules also carries a non-negligible false negative rate. Therefore, a negative biopsy result does not definitively exclude HCC, and repeat biopsy or close follow-up is advised.3 Nodules smaller than 1 cm are usually benign, and their small size complicates characterization; close monitoring for growth suggesting malignancy is the best approach in liver mass differential diagnosis.
Figure 2.
Diagnostic algorithm for suspected hepatocellular carcinoma (HCC) upon ultrasound detection of a liver nodule, a critical process in liver mass differential diagnosis. HBV, Hepatitis B virus; MDCT, multidetector CT; US, ultrasound. (Adapted with permission from Bruix and Sherman (3)).
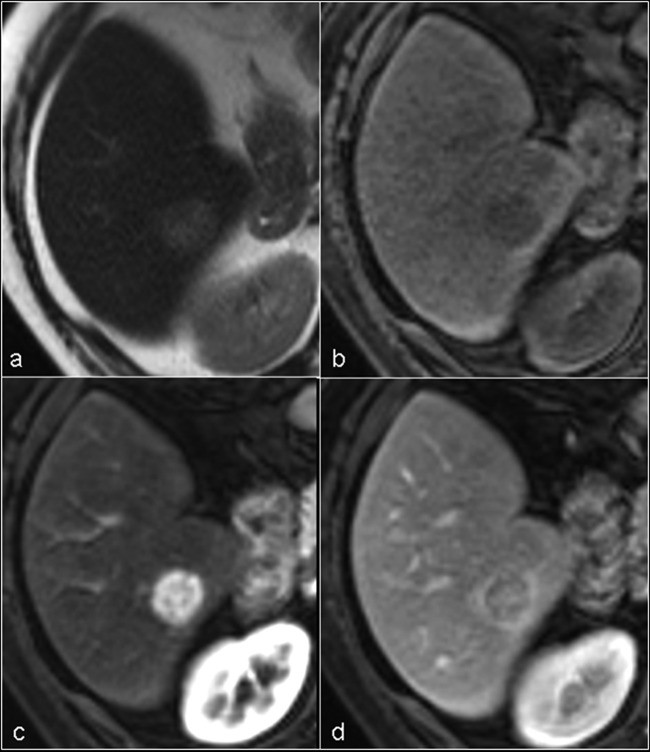
Figure 3.
MRI appearance of typical hepatocellular carcinoma (HCC), demonstrating key features for liver mass differential diagnosis. (A) Axial T2-weighted image showing a well-defined, slightly hyperintense nodule in segment VI of the right hepatic lobe. (B) Unenhanced axial T1-weighted image showing the nodule as slightly hypointense. (C) Gadolinium-enhanced T1-weighted image during the arterial phase showing intense and homogeneous contrast uptake (wash-in) in the nodule. (D) Portal phase image showing the lesion as hypointense (wash-out).
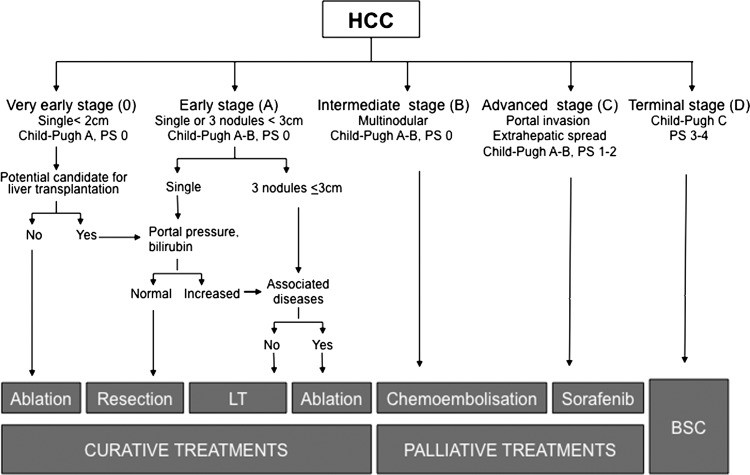
HCC prognosis depends on tumor stage, liver function, and general health.42 Several staging systems have been developed, with the Barcelona-Clinic-Liver-Cancer (BCLC) system being widely validated and recommended in the USA3 and Europe4 as it links prognosis and treatment. The BCLC system considers tumor stage (size, number of nodules, vascular invasion, extrahepatic spread), liver function (Child-Pugh score, portal hypertension), and general health (ECOG Performance Status), stratifying patients into five stages, each with specific treatment strategies (figure 4).5, 43 Very early stage (Stage 0) HCC involves patients with preserved liver function, no portal hypertension, a solitary tumor ≤2 cm, well-differentiated, and low dissemination potential. Ablation is the primary option, with resection reserved for transplant candidates or ablation failures. 5-year survival rates can exceed 80% with these curative therapies.43 Early stage (Stage A) HCC includes patients with preserved liver function, no cancer symptoms, single tumors, or up to three nodules ≤3 cm. These patients are candidates for curative treatment (resection, liver transplant, ablation), with expected 5-year survival rates of 50%–70%. Resection is best for solitary tumors without significant portal hypertension or hyperbilirubinemia.44, 45 Liver transplantation is preferred for patients with impaired liver function unsuitable for resection, particularly those meeting Milan criteria (single tumor 46 Percutaneous ablation, especially radiofrequency ablation47, 48, is considered for Stage 0 or A patients ineligible for surgery or as a bridge to transplant.3, 4 Intermediate stage (Stage B) patients are asymptomatic with large or multifocal HCC beyond curative criteria, without vascular invasion or extrahepatic spread. Transarterial chemoembolization (TACE) is the main treatment, requiring selective catheterization of the hepatic artery and injection of chemotherapy and embolic agents. TACE is well-tolerated, achieves tumor necrosis in over 50% of patients, and can lead to median survival exceeding 4 years in well-selected patients using advanced techniques.50 Yttrium-90 radioembolization is a novel approach showing promise in cohort studies51, 52, but further trials are needed. Advanced stage (Stage C) HCC involves large/multifocal tumors with vascular invasion, extrahepatic spread, or physical impairment. Untreated median survival is 4–8 months, and sorafenib is the only agent demonstrating survival benefit in randomized trials.53, 54 Terminal stage (Stage D) patients have poor physical status and/or extensive tumor burden, requiring symptomatic treatment. HCC patients with Child-Pugh grade C who are not transplant candidates fall into this category and do not benefit from antitumor treatments due to liver function impairment.5 Accurate liver mass differential diagnosis is crucial across all stages to guide appropriate management and improve patient outcomes.
Figure 4.
The Barcelona Clinic Liver Cancer (BCLC) staging and treatment strategy for hepatocellular carcinoma, highlighting the importance of liver mass differential diagnosis in treatment planning. LT, liver transplantation; PS, performance status. (Adapted with permission from Forner et al5 BSC, best supportive care).
Fibrolamellar Carcinoma
Fibrolamellar carcinoma is a rare variant of HCC (1%–9%), more common in Western countries and typically affecting young patients without underlying chronic liver disease. It usually presents as a large, solitary, well-defined but non-encapsulated intrahepatic mass with prominent fibrous bands connected to a fibrotic, often calcified, central scar, features important in liver mass differential diagnosis. Diagnosis is often late, when patients present with symptoms related to mass effect and constitutional syndrome. Alpha-fetoprotein levels are normal in over 90% of patients. Diagnosis and staging rely on CT and/or MRI,55 but percutaneous core biopsy may be needed for atypical imaging presentations to refine liver mass differential diagnosis. Fibrolamellar HCC tends to be slow-growing and often resectable, with a better prognosis than classic HCC.56 Aggressive surgical resection or liver transplantation is feasible in approximately 80% of cases, with a 5-year survival rate of 75%.57
Intrahepatic Cholangiocarcinoma
Intrahepatic cholangiocarcinoma (ICC) is an adenocarcinoma originating from intrahepatic biliary epithelial cells. ICC is less common than extrahepatic cholangiocarcinoma and typically presents as a focal mass-forming lesion. Risk factors for ICC include primary sclerosing cholangitis, hepatobiliary flukes, hepatolithiasis, and biliary malformations. Additionally, cirrhosis, especially secondary to chronic HCV infection, combined with alcohol use and metabolic syndrome, are recognized risk factors, potentially explaining the increasing ICC incidence in recent years, factors considered in liver mass differential diagnosis.58, 59 Dynamic imaging techniques can suggest the diagnosis. Typical radiological features include progressive contrast uptake in different phases, often with a central scar, vascular encasement, and capsule retraction. Peripheral bile duct dilatation is frequently observed.60, 61 ICC can have a vascular profile indistinguishable from HCC on contrast-enhanced ultrasound (CEUS),62, 63 limiting CEUS use for non-invasive HCC diagnosis in cirrhotic patients.3, 4 Tumors become symptomatic when large, and surgical resection is the best therapeutic option, though feasible in only a minority of patients, offering a 3-year survival rate of 40%–60%.64, 65 Liver transplantation is not recommended due to poor outcomes.59 For locally advanced or metastatic cholangiocarcinoma, systemic chemotherapy with cisplatin plus gemcitabine has shown survival benefit.66 Accurate liver mass differential diagnosis is vital to distinguish ICC from other liver malignancies and guide appropriate treatment.
Angiosarcoma
Angiosarcoma is the most common primary sarcoma of the liver.67 Peak incidence is in the sixth and seventh decades of life, more frequently in men (male:female ratio 3:1). It originates from endothelial cells of the sinusoidal lining. Tumor cells infiltrate sinusoids, hepatic and portal veins, eventually replacing liver parenchyma. Angiosarcoma has been linked to Thorotrast, vinyl chloride, and arsenic exposure, making occupational and environmental history relevant in liver mass differential diagnosis.67 Symptoms may mimic chronic liver disease, but in 15% of patients, diagnosis occurs due to acute hemoperitoneum from tumor rupture. Dynamic CT or MRI shows gradual contrast enhancement and homogeneity in the late phase. MRI may show hypointensity on T1-weighted imaging and hyperintensity on T2-weighted imaging, potentially resembling haemangioma, emphasizing the need for careful liver mass differential diagnosis.9 Liver biopsy confirms the diagnosis. Diagnosis is usually made at an advanced stage when surgery is not feasible, and prognosis is poor, with expected survival less than 6 months.67
Hepatic Epithelioid Hemangioendothelioma
Hepatic epithelioid hemangioendothelioma (EHE) is a rare, low-grade malignant vascular tumor that can arise in the liver, lung, soft tissue, or bone in adults. EHE is more common in women. It originates from endothelial cells and histologically consists of small tumor cell groups within a distinctive sclerotic stroma. Unlike haemangiosarcoma, hepatic acinar landmarks are preserved.68 The pathogenesis is unknown; symptoms are non-specific, and the clinical course is unpredictable, ranging from stable for years to aggressive progression, posing challenges for liver mass differential diagnosis and prognosis. Imaging studies show multiple, peripheral, nodular lesions and large masses, often mimicking metastatic disease.68 Diagnosis requires pathological assessment with positive immunostaining for factor VIII-related antigen, CD31, and CD34. EHE treatment is not standardized. Liver transplantation is debated due to documented spontaneous long-term survivals, high extrahepatic disease incidence (up to 45%), lack of predictive criteria, and high recurrence rates in allografts (up to 33%).69 Transplantation is generally delayed until clear tumor progression is evident.70 Accurate liver mass differential diagnosis is crucial for appropriate management of EHE.
Summary
Accurate characterization of liver masses is a significant diagnostic challenge for clinicians. Diagnosing a focal liver lesion relies on clinical context and imaging findings, with biopsy needed in some cases for definitive liver mass differential diagnosis. For cystic lesions, ultrasound is often sufficient for simple cyst diagnosis. If lesions are atypical, clinical features, hydatid and amoebic serology, and CT or MRI help differentiate simple cysts, polycystic liver/kidney disease, hydatid cysts, pyogenic abscesses, and amoebic abscesses. Differentiating cystoadenoma from cystoadenocarcinoma is difficult and may require pathological assessment if suspected. In contrast, solid liver lesions present a broad differential diagnosis, ranging from benign asymptomatic lesions to malignant neoplasms. Clinical history is highly informative. In healthy patients, haemangioma is the most common lesion, easily diagnosed by ultrasound and MRI. In young women using oral contraceptives, FNH and HCA must be considered in the liver mass differential diagnosis due to differing prognoses and treatments. If lesion nature remains unclear after imaging and biopsy, surgical resection may be recommended. In patients with chronic liver disease, HCC is the most frequent diagnosis. A well-established diagnostic algorithm, summarized in figure 2, exists for HCC. Diagnosis in cirrhotics often relies on dynamic imaging showing a specific vascular pattern. However, other malignancies like ICC must be excluded in liver mass differential diagnosis, as treatment and prognosis differ from HCC. Finally, a solid lesion in a patient with known cancer is often liver metastasis. Imaging and tumor markers are usually diagnostic in this context, but biopsy may be needed in doubtful cases. Therefore, a systematic approach to liver mass differential diagnosis, integrating clinical information, imaging, and pathology, is essential for optimal patient care.
References
1
3
4
5
6
9
19
20
21
22
23
24
25
26
27
28
30
31
32
33
34
35
36
37
38
42
43
44
45
46
47
48
49
50
51
52
53
54
55
56
57
58
59
60
61
62
63
64
65
66
67
68
69
70