Periodontal disease, a widespread oral health issue, is marked by gum inflammation and the breakdown of periodontal tissues. Clinicians face significant challenges in diagnosing this condition because of its fluctuating nature, characterized by periods of worsening and improvement. Traditional diagnostic methods primarily reveal past tissue damage. Therefore, innovative diagnostic techniques are essential to identify the disease’s active phase, predict its future progression, and assess therapy response, ultimately leading to improved patient management. Saliva and Gingival Crevicular Fluid (GCF) are recognized as reliable sources for biomarkers that play a crucial role in gauging disease activity. Considering these factors, rapid chairside diagnostic tools, known as Point of Care (POC) diagnostics, have been developed to simplify periodontal disease diagnosis and enhance prognosis. This article offers a comprehensive review of the biomarkers employed in diagnosis and highlights various available point-of-care diagnostic devices.
The Imperative for Periodontal Diagnostic Innovations
Identifying patients at high risk of active periodontal disease and pinpointing active disease sites are major hurdles for clinicians. There’s a pressing need for advancements in diagnostic methods to facilitate early detection of microbial challenges, presence of disease processes, sites susceptible to future deterioration, and to evaluate patient responses to treatment.
These novel diagnostic approaches largely depend on detecting markers indicative of disease activity. The term “disease markers” encompasses three distinct categories: (1) indicators of ongoing disease activity; (2) predictors of future disease progression; and (3) predictors of disease onset at currently healthy sites. Defining “disease” itself is crucial, particularly in distinguishing gingivitis from destructive periodontitis.
Biomarkers, defined as measurable biological parameters, serve as indicators for health and physiological assessments. They provide a “signature” of health status and can be found in biological fluids like blood, urine, and increasingly, saliva. Oral fluid, often considered a mirror of periodontal health, is a valuable medium for clinically relevant information due to its periodontal disease-specific biomarkers. These biomarkers include host-derived proteins (enzymes, immunoglobulins), host cells (PMNs), bacteria and their byproducts, ions, hormones, and volatile compounds.
Over the last decade, numerous diagnostic tests utilizing physical, chemical, microbiological, and immunological techniques have emerged. Given the unpredictable disease pattern and associated risk factors, these tests should provide crucial information for initial diagnosis and long-term periodontal patient management. This diagnostic innovation is poised to be a significant asset in the clinical management of periodontal disease.
Point of Care Diagnostics: Bringing Diagnosis to the Chairside
High specificity and sensitivity are critical for diagnostic markers intended for chairside or home-use devices. The widespread adoption, simplicity, reliability, and cost-effectiveness of home pregnancy tests serve as an ideal model for periodontal diagnostics.
Emerging technologies like ‘lab-on-a-chip’ and microfluidic devices offer promising solutions for managing oral fluids like saliva and gingival crevicular fluid. These devices can determine a patient’s periodontal disease risk profile, current disease activity, and response to treatments. This approach is particularly beneficial for managing chronic infectious conditions like periodontitis, enabling monitoring of its episodic nature and aiding in clinical decision-making. While saliva, serum, and GCF are all potential vehicles for assessing periodontal disease activity, saliva and GCF stand out due to their ease of collection and richness in both local and systemic markers of periodontal disease. This makes them highly valuable for patient-specific biomarker assessment in diagnosing periodontitis and other systemic conditions.
Saliva: A Non-Invasive Diagnostic Fluid
Saliva is a readily accessible biofluid obtainable through completely non-invasive methods. Various substances can enter saliva from the bloodstream via passive diffusion, active transport, or extracellular ultrafiltration within salivary glands, or through the gingival sulcus. Consequently, most compounds present in blood are also found in saliva. Saliva is a highly useful and easily obtainable bodily fluid for monitoring both oral and systemic health.
Biomarkers Present in Saliva
| Markers of Periodontal Soft Tissue Inflammation | Markers of Alveolar Bone Loss | Collagen Breakdown Products |
|---|---|---|
| Prostaglandin E2 | Alkaline phosphatase | Aspartate aminotransferase |
| β-glucuronidase | Osteoprotegerin | Alanine aminotransferase |
| IL-1β | Osteocalcin | TIMPs |
| IL-6 | Collagen telopeptidase | MMPs |
| Tumor necrosis factor-α | Pyridinoline cross-links of type I collagen | α2-macroglobulin |
| Matrix Metalloproteinase (MMP-8,9 and 13) | RANKL | |
| Osteonectin |

Whole saliva from patients with oral diseases is abundant in mediators of chronic inflammation and tissue destruction. Over 1,000 proteins have been identified as salivary biomarkers. Salivary diagnostics, particularly the detection of minute quantities of salivary components like proteins and messenger RNA (mRNA), is currently a highly promising area of dental research.
Saliva in Point of Care Diagnostics: Saliva offers numerous advantages for POC diagnostics: easy accessibility, a rich array of diagnostic biomarker molecules, non-invasive sampling, and the potential for rapid, reliable results. Saliva is also advantageous compared to blood due to easier handling (no clotting) and a lower risk of accidental infectious disease transmission during collection. However, a primary limitation of saliva is the lower concentration of informative analytes compared to serum, necessitating highly sensitive assays. Saliva composition is also influenced by its origin and various environmental and psychological factors. While qualitative analysis of salivary markers is reliable, quantifying these markers remains challenging. Furthermore, the presence of mucins and cell debris makes saliva a complex fluid to work with.
Commercially Available Point of Care Diagnostics
| Test Kits | Functions |
|---|---|
| Oral fluid nanosensor test | Detection of multiple salivary proteins and nucleic acids. |
| Electronic taste chips | Simultaneously monitor several biomarkers related to periodontal disease |
| OraQuick | Usually detects HIV 1 and HIV 2 |
| Integrated microfluidic platform for oral diagnostics | Quantification of an oral disease biomarker |
Biochemical Tests:
a. Oral Fluid Nanosensor Test (OFNASET): Developed at UCLA, this POC device is designed for electrochemical detection of multiple salivary proteins and nucleic acids, primarily for oral cancer screening. OFNASET is an ultra-sensitive microelectromechanical system capable of simultaneously and precisely detecting salivary mRNA biomarkers (SAT, ODZ, IL-8, IL-1b) and proteomic biomarkers (thioredoxin and IL-8).
b. Electronic Taste Chips (ETC): Researchers are developing a lab-on-a-chip system to differentiate between healthy and periodontally diseased individuals based on CRP levels. ETC is a microchip-based detection system for measuring analytes in solution. It utilizes a sensor array platform on microspheres within microchip chambers to perform chemical and immunological reactions, visualized by a CCD video chip. ETC offers advantages over ELISA due to porous beads allowing greater antibody capture and CRP detection at low concentrations.
c. OraQuick: This rapid POC HIV test provides results in 20 minutes, using a simple device to mix fluid with a developing solution. OraQuick® is the first FDA-approved oral swab in-home test for HIV-1 and HIV-2, demonstrating the feasibility of rapid oral fluid diagnostics.
d. Integrated Microfluidic Platform for Oral Diagnostics (IMPOD): IMPOD is a POC diagnostic test for rapid quantification of salivary biomarkers related to oral disease. It integrates sample pretreatment with electrophoretic immunoassays for quick measurement of analyte concentrations in minimally processed saliva. IMPOD can rapidly (3–10 minutes) measure MMP-8 and other biomarker concentrations in small saliva samples.
Microbiological Tests:
a. My PerioPath: This test detects periodontal disease-causing pathogens in saliva using DNA polymerase chain reaction to identify the type and concentration of bacteria present.
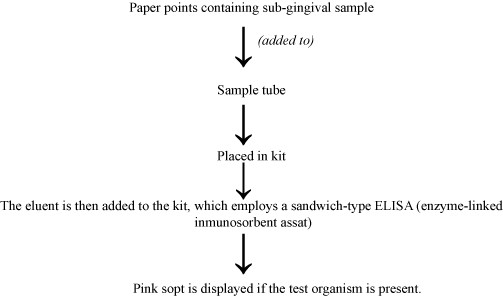
b. Omnigene: OmniGene Diagnostics offers species-specific DNA probes to identify eight key periodontal pathogens: Porphyromonas gingivalis, Prevotella intermedia, Aggregatibacter actinomycetemcomitans, Fusobacterium nucleatum, Eikenella corrodens, Campylobacter rectus, Bacteroides forsythus, and Treponema denticola. Results are quickly available and can be delivered to clinicians rapidly.
c. IAI Pado Test: This microbiological test uses RNA probes to detect microorganisms associated with periodontitis, including A. actinomycetemcomitans, P. gingivalis, T. forsythia, and T. denticola in collected samples.
Genetic Tests:
a. MyPerioID: MyPerioID assesses a patient’s genetic susceptibility to periodontal diseases using saliva samples. This test helps identify individuals at higher risk of periodontal destruction.
Gingival Crevicular Fluid (GCF): A Window into the Periodontal Pocket
GCF is a fluid derived from serum, leukocytes, periodontal cells, and oral microflora. Its composition reflects the interaction between bacterial biofilm and periodontal tissues, making it a biochemical indicator of local metabolic changes and periodontal status. Host response is crucial in periodontal disease pathogenesis, and GCF inflammatory mediator levels can assess the risk of attachment and bone loss at specific sites or the overall risk of an individual developing periodontal disease. GCF contains a broad spectrum of markers from both host and bacteria in plaque, offering numerous candidates for detecting periodontal disease activity.
Biomarkers in Gingival Crevicular Fluid
| Inflammatory and Immune Products | Bacterial Proteases | Host Derived Enzymes | Tissue Breakdown Products | Bone Specific Proteins |
|---|---|---|---|---|
| Prostaglandin E2 (PGE2) | Alkaline phosphatase | Alkaline phosphatase | Glycosaminoglycan | Pyridinium crosslink urine pyridinoline |
| Cytokines | Aminopeptidases | β-Glucuronidase | Hyaluronic acid | Pyridinium cross-link collagen peptide fragment |
| Antibacterial antibodies | Chondroitin sulphatase | Elastase | Chondroitin-4-sulfate | Tartrate-resistant acid phosphatase |
| Acute phase proteins | Collagenase | Cathepsins | Chondroitin-6-sulfate | Hydroxyproline |
| Complement | Fibrinolysin | Serine proteinase (G) | Dermatan sulfate | Galactosyl hydroxylysine |
| Vasoactive intestinal Peptide | Glucosidases | Nonspecific neutral proteinases | Hydroxyproline | Glycosaminoglycans |
| Neurokinin a | Hemolysin | Matrix metalloproteinase-l,3,8,13 | Fibronectin fragments | Osteonectin and bone phosphoprotein |
| Neopterin | Hyaluronidase | Aspartate amino transferase | Connective tissue and bone proteins | osteocalcin |
| Platelet -activating factor | Phospholipase | Myeloperoxidases | Type I collagen peptides | |
| Hydroxyproline | Lactate dehydrogenase | Polypeptide growth factor |
GCF is a valuable medium for detecting early disease onset. Over 65 GCF chemicals are considered potential prognostic markers for periodontal diseases. These biomarkers are broadly classified into inflammatory and immune products, bacterial enzymes, host-derived enzymes, tissue breakdown products, and bone-specific proteins.
GCF in Point of Care Diagnostics: GCF is frequently used for biomarker analysis due to its accessibility in the oral cavity. Biomarkers in GCF can indicate periodontal pathogens, gingival and periodontal inflammation, host immune response, and tissue destruction. However, GCF analysis can require multiple samples from individual tooth sites and extensive lab processing, making it costly and time-consuming. While GCF offers diagnostic advantages, collection and analysis procedures can be challenging for chairside use. GCF collection is technically demanding, requires specialized equipment, and is susceptible to contamination from blood, saliva, or plaque.
Commercially Available Kits Using GCF for Detecting Host Derived Enzymes
| Test Kits | Enzymes |
|---|---|
| Periogard | AST |
| Pocket watch | AST |
| Periocheck | Collagenase (neutral protease) |
| Prognostik (Dentsply), Biolise | Elastase (serine protease) |
| MMP dipstick method | MMP |
Biochemical Tests:
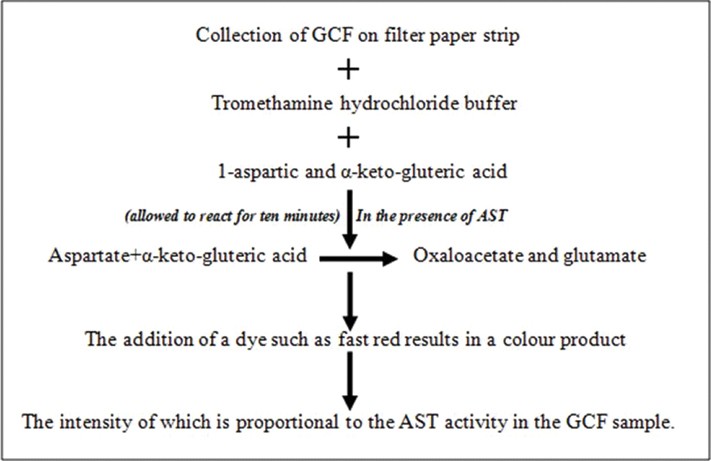
a. Periogard: Periogard detects Aspartate aminotransferase (AST), an enzyme released upon cell death. Elevated AST levels in periodontal disease indicate active disease sites. The test uses two wells for each tooth and specific chemicals.
However, Periogard is relatively complex, involving multiple steps and challenging color measurement.
b. Pocket Watch: Pocket Watch is a chairside test for analyzing AST levels. It utilizes a principle where AST catalyzes a reaction involving cysteine sulfuric acid and α-keto-gluteric acid, leading to a color change that indicates AST concentration.
c. Periocheck: Periocheck, an FDA-approved product, is a rapid chairside test for detecting neutral proteases like elastases, proteinases, and collagenase in GCF.
However, it has limitations, including difficulty sampling interproximal sites due to saliva contamination and potential inclusion of bacterial enzymes.
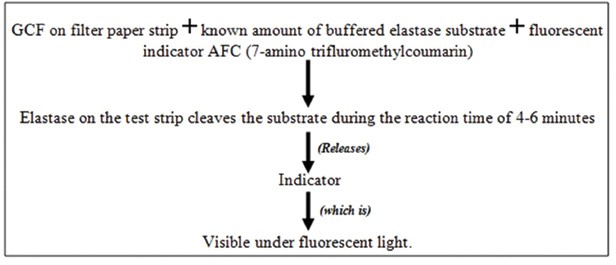
d. Prognostik: Developed in 1993, Prognostik measures elastase levels in GCF, which are elevated in active disease sites due to release from polymorphonuclear leukocyte lysosomes.
Further clinical trials are needed to confirm the correlation between GCF elastase levels and periodontal disease activity.
e. MMP Dipstick Test: MMPs, host-derived proteinases crucial in periodontitis, are the basis for POC technologies like the MMP-8 dipstick test. This immunotest uses monoclonal antibodies for MMP-8 to differentiate between healthy gingiva, gingivitis, and periodontitis sites, correlating with quantitative laboratory assays.
Microbial Test Kits: Targeting Dental Biofilm
Dental plaque is a potential medium for detecting periodontopathogens, crucial for periodontal disease diagnosis and treatment. Significant advancements have been made in methods for detecting periodontopathogens in plaque samples.
Biomarkers Present in Dental Biofilm
| Markers Present in Dental Biofilm |
|---|
| Specific (Immunoglobulins: IgA, IgG, IgM, Lysozyme, Lactoferrin, Histatin, Peroxidase) |
Other Commercially Available Kits for Detecting Bacterial Protease
| Test Kits | Bacteria and Their Products |
|---|---|
| Perioscan (BANA test) Oral B lab | Trypsin like protease |
| Evalusite (Kodak) | P. gingivalis, P. intermedia, A. actinomycetemcomitans |
| Perioscan/ Diamond probe/Probe 2000 system | For volatile sulphur compounds |
| TOPAS | Bacterial toxins and protease |
Microbiological Tests:
a. Perioscan (BANA): Perioscan detects bacterial trypsin-like proteases produced by P. gingivalis, T. denticola, T. forsythia, and some Capnocytophaga strains in dental plaque.
Limitations include inability to detect pathogens with non-trypsin-like enzymes and differentiate specific bacteria among those producing trypsin-like enzymes.
b. Evalusite: Evalusite, a membrane-based enzyme immunoassay, detects three periodontopathogens (Aa, Pg, Pi).
Subjective color assessment and the assumption that only these three bacteria cause disease are major drawbacks.
c. Perio 2000: Perio 2000 system digitally measures Volatile Sulphide Compounds (VSCs), produced by microorganisms like P. gingivalis, P. intermedia, and T. forsythia from serum protein degradation. VSC levels indicate subgingival microbial load and periodontal disease severity.
d. Toxicity Prescreening Assay (TOPAS): TOPAS indirectly detects bacterial toxins and proteins in crevicular fluid, indicating gingival infection. It assesses metabolic activity of pathogens to differentiate between active and inactive periodontal disease based on color intensity changes.
Genetic Tests:
a. Periodontitis Susceptibility Trait Test (PST): PST identifies genetic predisposition to periodontitis by detecting polymorphism in the IL-1 gene, specifically at positions -889 and +3953, which are associated with periodontal disease.
Advantages of Point of Care Diagnostics
POC testing eliminates the need for blood draws and reduces costs associated with sample shipping and centralized lab processing. This significantly reduces turnaround time and enhances care quality by enabling immediate treatment initiation. Periodontal POC devices require less training and resources than traditional tests, proving more efficient for rapid, large-scale screening. Population screening effectively identifies at-risk groups and improves treatment access.
Disadvantages of Point of Care Diagnostics
While promising, POC diagnostics in periodontal care face obstacles in clinical settings. These new diagnostics need validation and benchmarking against established methods like alveolar bone level and clinical attachment level assessments. Acceptance by dentists and clinicians is crucial but may be challenging. Cost-effectiveness is another important consideration. Clinicians need comprehensive knowledge of diagnosis, disease risk, and prevention before integrating POC diagnostics into routine practice.
Summarization of Oral Tests
| S. No. | Oral Fluid | Test | Kit |
|---|---|---|---|
| 1 | Saliva | Biochemical test | a) Oral fluid nanosensor test b) Electronic taste chip c) OraQuick d) Integrated microfluidic platform for oral Diagnostics |
| Microbiological test | a) My PerioPath b) Omnigene c) IAI pado test | ||
| Genetic test | a) MyperioID | ||
| 2. | Gingival crevicular fluid | Biochemical test | a) Perriogard b) Pocket watch c) Periocheck d) Prognostik e) MMP dipstick test |
| 3. | Plaque | Microbiological test | a) Perioscan (BANA) b) Evalusite c) Perio 2000 d) TOPAS Genetic test kits |
| 4. | Periodontitis susceptibility trait test |
Advances in Point of Care Diagnostics: Lab-On-Chip Devices
Lab-on-a-chip technology represents the next generation of POC diagnostics. These devices integrate and automate complex lab procedures onto a computer chip-sized device. This technology aims to measure multiple biomarkers in small saliva samples, promising enhanced diagnostic capabilities.
Conclusion: Point of Care Diagnostics – A Paradigm Shift in Periodontal Care
Accurate initial diagnosis is fundamental to successful periodontal treatment. While current methods are adequate, assessing “active disease sites” is increasingly important. Commercially available chairside test kits utilizing host and bacterial markers now enable site-specific monitoring. Extensive research is exploring oral fluids as diagnostic media across various medical fields. Despite existing challenges, saliva and GCF-based oral fluid diagnostics hold significant promise for periodontal disease diagnosis and treatment outcome prediction, heralding a new era in periodontal care.
Financial or Other Competing Interests
None.