Psychiatric research and clinical practice have increasingly adopted functional and structural brain imaging over the past three decades. Neuroimaging studies form a significant portion of psychiatric research, aiming to elucidate the neural mechanisms underlying various mental health disorders. Single-photon emission computed tomography (SPECT) stands out as a valuable brain imaging technique among the neuroimaging technologies available. SPECT offers advantages over PET scans, being easier to perform, less expensive, and more practical for radioligand application. Modern advancements have enhanced the spatial precision of SPECT findings by integrating functional SPECT images with CT scans. Radioligands that bind to receptors like 5-hydroxytryptamine 2A and dopamine transporters are crucial in understanding the neurochemical bases of psychiatric disorders. This review focuses on the application of SPECT-based neuroimaging in diagnosing and understanding psychiatric disorders such as schizophrenia and major depressive disorder (MDD). Research findings using SPECT in psychiatric disorders reveal significant alterations in biochemical components associated with specific conditions. Despite growing evidence supporting SPECT’s potential in psychiatric clinical practice, current diagnostic approaches still heavily rely on subjective criteria, such as the Diagnostic and Statistical Manual of Mental Disorders (DSM-5). SPECT’s capability to provide insights into the brain’s biochemical environment in psychiatric disorders offers valuable information about the pathophysiology and the future implications of neuroimaging techniques in this field.
Keywords: depression, SPECT, dopamine, schizophrenia, molecular imaging
Introduction
The diagnosis of psychiatric disorders has long been a subject of debate, with a persistent lack of objective diagnostic tools. Throughout revisions of diagnostic frameworks, schizophrenia and depression have consistently been recognized as valid and stable psychiatric disorders in terms of diagnostic criteria. However, despite advancements in diagnostic criteria for these conditions, a deep understanding of their biological underpinnings remains elusive. This gap highlights the critical need for neuroimaging methods to bridge the divide between clinical diagnosis and biological understanding in psychiatric disorders.
Functional and structural imaging techniques have significantly contributed to a more profound understanding of the molecular processes involved in psychiatric disorders (1). Techniques such as Magnetic Resonance Imaging (MRI), functional MRI (fMRI), SPECT, and Positron Emission Tomography (PET) have evolved to shed light on the complexities of psychiatric conditions. SPECT and PET share methodological similarities, both providing information based on the spatial distribution of injected radiopharmaceuticals. The primary distinction between PET and SPECT lies in the radiotracers they employ. SPECT utilizes heavier isotopes like 99mTc and 123I, which emit gamma-rays, whereas PET uses lighter isotopes such as 11C, 13N, and 18F, which emit positrons. SPECT benefits from longer half-lives of its pharmaceuticals compared to PET, enabling longitudinal scans. Furthermore, SPECT is more cost-effective than PET, increasing its accessibility and practicality in clinical settings. Despite these advantages, PET offers superior spatial resolution compared to SPECT (2).
Effective radiotracers for brain imaging must possess specific characteristics to ensure high-quality images. These include high affinity and specificity for the intended target, minimal non-specific binding, low plasma protein binding, the ability to cross the blood-brain barrier, and rapid plasma clearance (3). The most commonly used radiotracer in brain SPECT is Tc-99m-hexamethylpropylene amine oxime (HMPAO), which measures perfusion. Perfusion SPECT is valuable in diagnosing and evaluating neuropsychiatric pathologies, including dementia, traumatic brain injury (TBI), toxin exposure, and inflammatory disorders, by detecting hypoperfusion in the brain (4, 5). Additionally, perfusion SPECT provides essential insights into various psychiatric disorders. It can aid in predicting stimulant medication response in children with attention deficit hyperactivity disorder (ADHD) (6) and reveal blood perfusion changes in treatment-resistant MDD (7). While perfusion SPECT is widely used in psychiatric research, neurochemical SPECT studies offer more specific information about the underlying pathophysiology. This target-specific capability makes SPECT an invaluable tool for psychiatric research, particularly in the context of psychiatric diagnosis.
This mini-review summarizes and discusses SPECT findings related to schizophrenia and depression, focusing on the keyword Spect Scan For Psychiatric Diagnosis. To gather relevant SPECT studies in schizophrenia and depression, a systematic literature search was conducted in the PubMed database using the keywords “SPECT,” “Schizophrenia,” and “Depression,” combined with terms such as “GABA,” “Serotonin,” “Glutamate,” and “Dopamine.” The most pertinent and recent studies were selected for inclusion, balancing recent developments with the acknowledged limitations in this field.
SPECT in Schizophrenia
Schizophrenia is recognized as a complex and chronic psychiatric disorder characterized by both positive and negative symptoms. These symptoms originate from intricate molecular alterations within the brain. The molecular basis of schizophrenia has been extensively researched to gain a deeper understanding of the disease’s nature. Dopamine is among the most studied molecular components in schizophrenia (8). Dopaminergic pathways consist of presynaptic and postsynaptic elements. The presynaptic component involves dopamine synthesis, dopamine storage in vesicles via vesicular monoamine transporter 2 (VMAT2), dopamine degradation by monoamine oxidase (MAO), dopamine release, and dopamine reuptake by the dopamine transporter (DAT). The postsynaptic component includes dopamine receptors and post-receptor signaling [(9); Figure 1].
Figure 1.
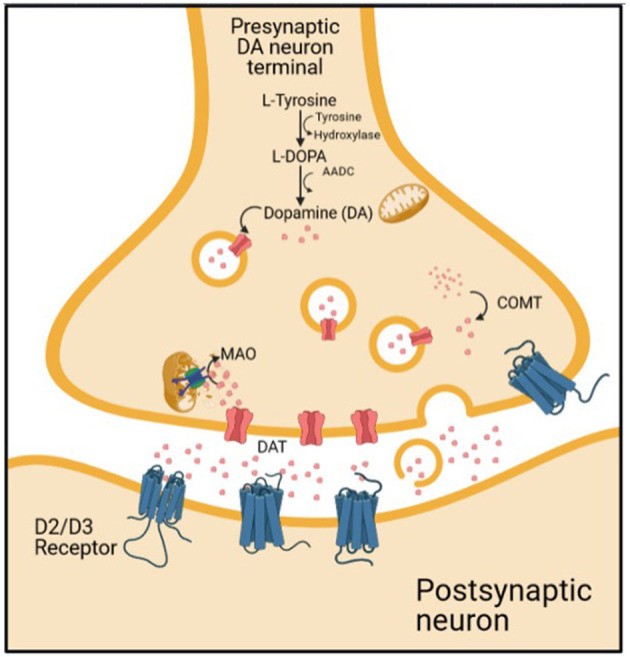 Figure 1
Figure 1
The dopaminergic transmission on synaptic cleft. Alt text: Illustration depicting dopaminergic neurotransmission at a synapse, showing dopamine synthesis, release, receptor binding, and reuptake mechanisms relevant to SPECT scan for psychiatric diagnosis.
Imaging of Dopaminergic System
Alterations in the dopaminergic pathway are known to be involved in the pathophysiology of schizophrenia. Imaging studies have indicated elevated subcortical dopamine activity and reduced cortical dopamine levels as part of the disease process (10). Among the imaging tools targeting the molecular mechanisms of schizophrenia, SPECT has made substantial contributions to psychiatric diagnosis research. The first SPECT study on schizophrenia was published in 1986 (11). This study utilized 77Br-bromospiperone, a radiotracer now less favored due to its imaging limitations, to visualize D2 receptor density.
Advances in dopamine research in schizophrenia led to the development of new molecules for assessing postsynaptic dopaminergic activity, such as [123I] iodobenzamide (IBZM) and [123I] epidepride [(12); Table 1]. These radiotracers bind to postsynaptic receptors, allowing for the assessment of postsynaptic dopamine activity. However, antipsychotic medications used to treat schizophrenia also bind to these same receptors, potentially interfering with radiotracer binding. Consequently, patients on antipsychotics might show lower dopamine receptor availability in SPECT scans than their actual levels (28). Studies investigating the effects of antipsychotics on dopamine receptors have found a negative correlation between the basal ganglia/frontal cortex ratio (BG/FC), indicative of striatal dopaminergic activity, and extrapyramidal side effects (13, 14). Moreover, a lower BG/FC ratio of IBZM was observed in patients with inadequate response to antipsychotic treatment (14). Meta-analyses generally suggest a slight elevation in postsynaptic D2/D3 receptor activity in schizophrenia patients, although this effect is not significant in treatment-naïve individuals (16, 29).
Table 1.
Binding sites and summary of studies of available SPECT radiotracers for dopamine, serotonin, GABA, and glutamate in psychiatric diagnosis research.
| Radiotracers | Neurotransmitters | Binding sites | Additional findings relevant to SPECT scan for psychiatric diagnosis |
|---|---|---|---|
| [123I] iodobenzamide | Dopamine | Striatal post-synaptic D2/D3 receptor | BG/FC binding ratio negatively correlated with EPS and lower in schizophrenia patients on antipsychotics (13, 14). |
| [123I] epidepride | Dopamine | Extrastriatal frontal D2/D3 receptors | Frontal D2/D3 binding positively correlated with positive symptom reduction (15). |
| [123I]-FP-CIT | Dopamine, serotonin | DAT, SERT | Meta-analysis showed no significant difference between schizophrenia patients and healthy controls (HC) (16). |
| [123I]-β-CIT | Dopamine, serotonin | DAT, SERT, NET | Meta-analysis showed no significant difference between schizophrenia patients and HC (16). |
| [123I] nor-β-CIT | Dopamine, serotonin | DAT, SERT, NET | Meta-analysis showed no significant difference between schizophrenia patients and HC (16). |
| [99mTc]-TRODAT | Dopamine | DAT | Meta-analysis showed no significant difference between schizophrenia patients and HC (16). |
| [123I] iomazenil | GABA | GABAA receptors (α1, α2, α3, and α6 subunits) | Inconsistent results in schizophrenia patients (17). |
| [123I] CNS-1261 | Glutamate | NMDA receptor | Global binding reduced in schizophrenia patients on clozapine. No significant difference between HC and treatment-naïve schizophrenia patients (18). |
| [123I] ADAM | Serotonin | SERT | Controversial results. Some studies found no significant difference between HC and depression (19–21). Others found lower SERT bioavailability in depression (22–24). |
| [123I] R91150 | Serotonin | 5HT2A | Lower signal-to-noise ratio compared to PET tracers. Harm avoidance correlated with high DLPFC 5HT2A binding (25–27). |
Dopamine release into the synaptic cleft, another aspect of the dopaminergic pathway, can also be investigated in vivo using IBZM (30. This can be assessed through a combination of dopamine release induction using amphetamine or methylphenidate and SPECT imaging. When dopamine receptors are occupied by released dopamine, it leads to decreased radiotracer binding, indicating a negative linear relationship between dopamine release and radiotracer availability. Imaging studies examining dopamine release in schizophrenia patients indicate an increased amphetamine-induced dopamine release (31–33).
[123I] epidepride is another radiotracer used to identify extrastriatal frontal D2/D3 receptors, which are involved in cognitive functions such as planning, attention, and task switching (34–36). Inhibition of these extrastriatal receptors by antipsychotics can lead to decreased attentional focus, as shown in previous research (15). Furthermore, a study on zuclopenthixol and risperidone treatment found a positive correlation between positive symptom reduction and the binding potential of frontal D2/D3 receptors over three months of treatment (15).
The dopamine transporter is another critical component of the dopaminergic system. SPECT studies comparing schizophrenia patients with healthy controls, assessing dopamine transporter levels using [123I]-FP-CIT (N-omega-fluoropropyl-2beta-carboxymethoxy-3beta-{4-iodophenyl} tropane), [123I]-β-CIT (Iodine-123-beta-carbomethoxy-3 beta-(4-iodophenyltropane)), and [99mTc]-TRODAT, have shown no significant differences (16). While SPECT studies on dopamine transport and postsynaptic dopamine receptors have yielded important insights, dopamine synthesis is another crucial process in the dopamine pathway in schizophrenia. Although SPECT studies cannot directly address dopamine synthesis, PET meta-analysis indicates elevated dopamine synthesis capacity in schizophrenia (16).
These findings suggest that alterations in the synaptic space of the dopaminergic system may be more influenced by treatment processes than by the disease itself (29). Studies focusing on the presynaptic dopaminergic system, primarily using PET due to the lack of suitable SPECT radiotracers, suggest that increased dopamine release and dopamine synthesis capacity contribute to presynaptic dopaminergic neurotransmission activity (37). This presynaptic dopaminergic activity is regulated by other neurotransmitter pathways, including glutamate and GABA (28).
Imaging of GABAergic System
GABA plays a vital role in the molecular pathology of schizophrenia by modulating presynaptic dopaminergic activity (28, 38). In vivo imaging and post-mortem studies investigating GABA’s role in schizophrenia have provided significant results for psychiatric diagnosis. SPECT studies using [123I] iomazenil can visualize α1, α2, α3, and α6 subunits of GABAA receptors (39). However, studies evaluating GABA bioavailability and symptom severity in schizophrenia have reported inconsistent findings. While two studies found no correlation between symptom severity and GABA receptor availability in schizophrenia patients (40, 41), one study reported a correlation based on negative and positive symptoms (42). Negative symptoms were negatively correlated with GABA binding in the medial frontal region, whereas positive symptoms were negatively correlated with GABA binding in the medial temporal lobe (42). Three voxel-wise studies assessing regional brain differences generally indicate low GABA binding, despite regional inconsistencies (40, 43, 44).
Despite the inconsistencies in GABA SPECT studies in schizophrenia, post-mortem studies have consistently replicated findings of reduced glutamic acid decarboxylase 67 (GAD67) mRNA, which is involved in cytosolic GABA synthesis (45–47). Subtype analyses of alpha units in post-mortem studies show a decrease in alpha 1 (47–49) subunits and an increase in alpha 2 subunits (49, 50), with inconsistent results for alpha 5 subunits (50–52).
A systematic review comparing healthy controls and schizophrenia patients regarding GABAA/BZ receptor binding availability reported no significant group differences (17). Consequently, while post-mortem studies have shown consistent results, in vivo neuroimaging studies, including SPECT scans for psychiatric diagnosis, have not yielded equally conclusive results for GABA alterations in schizophrenia.
Imaging of Glutamatergic System
Dysfunction in GABA can lead to disinhibition of the glutamatergic pathway and asynchronous cortical activity (17). Glutamate, like GABA, acts as a regulatory neurotransmitter in the presynaptic dopaminergic pathway. Glutamate receptors include both G-coupled metabotropic and ligand-gated ionotropic receptors. NMDA, an ion-gated glutamate receptor, has been implicated in schizophrenia pathogenesis (53). However, SPECT studies investigating NMDA levels are limited despite the recognized importance of glutamatergic transmission in schizophrenia diagnosis. In the literature, the glutamatergic pathway has been studied using [123I] CNS-1261, an NMDA receptor ligand. One SPECT study indicated no difference in the total volume binding distribution of [123I] CNS-1261 between healthy controls and drug-free schizophrenia patients (18). In the same study, global binding of [123I] CNS-1261 was reduced in patients on clozapine treatment compared to drug-free patients (18). This reduction might be related to downregulation from clozapine treatment, disease-related pathology, or competition between clozapine and the radiotracer at the binding site. Further research revealed a significant negative correlation between [123I] CNS-1261 binding and residual symptom severity in patients treated with typical antipsychotics (18). Conversely, symptom duration in medication-free patients was positively correlated with [123I] CNS-1261 binding in the middle inferior frontal cortex (54). These studies highlight the potential importance of the glutamatergic system in schizophrenia and its relevance for SPECT scan for psychiatric diagnosis research.
SPECT in Depression
Depression is a prevalent and debilitating mental disorder with a lifetime prevalence of approximately 20% and a high recurrence risk (55). Various molecular pathological processes underlie depression, though they are not fully understood. Molecular imaging of depression is an extensively researched area aimed at elucidating the molecular aspects of this disorder and improving psychiatric diagnosis. The monoamine hypothesis, a prominent theory, suggests that depressive patients have lower levels of serotonin, noradrenaline, and dopamine (56). This hypothesis has been studied for decades, yielding some supporting evidence. However, it remains debated, with ongoing discussions about the sufficiency of evidence (57). Molecular imaging methods, including SPECT scans for psychiatric diagnosis, offer potential in resolving this controversy through advanced radiotracers that assess dopamine, noradrenaline, serotonin levels, and, more recently, monoamine oxidase (MAO), the degrading enzyme of these monoamines. Currently, SPECT imaging cannot assess MAO due to the lack of suitable radiotracers.
Studies of Blood Perfusion
While neuroimaging studies often focus on brain function and cerebral blood flow, this review primarily addresses the molecular imaging of neurotransmitters and receptors in major depressive disorder. Briefly, studies have reported hypoactivity in the dorsolateral prefrontal cortex, pregenual anterior cingulate cortex, posterior anterior cingulate cortex, left superior temporal gyrus, insula, and cerebellum in depressed individuals (58). Conversely, subcortical structures (caudate, thalamus) and limbic structures (amygdala, anterior hippocampus) show hyperactivity (58).
Another significant finding related to regional brain activity in depression is hyperactivity in the subgenual anterior cingulate cortex (sgACC). Studies have linked sgACC hyperactivity to treatment-resistant depression (59, 60). The relationship between sgACC activity and treatment response has been extensively studied, and this finding may become beneficial for assessing treatment response in clinical practice in the future, potentially using SPECT scans for psychiatric diagnosis to monitor treatment efficacy.
Imaging of Serotoninergic System
In addition to cerebral blood flow imaging studies, neurochemical imaging in depression provides insights into molecular alterations. Serotonin is the neurotransmitter most commonly associated with depression. The serotoninergic system regulates sleep, stress responses, pain, motor activity, cognition, emotional behavior, appetite, aggression, and impulsivity—all prominently affected in depression (61). However, the specific impairments of the serotoninergic system in depression are not yet fully understood, making SPECT scans for psychiatric diagnosis in this area particularly valuable.
Serotonin synthesis begins with tryptophan, transported across the blood-brain barrier by a large amino acid transporter. Tryptophan is first hydroxylated by tryptophan hydroxylase to 5-hydroxy-tryptophan, which is then decarboxylated into 5-hydroxy-tryptamine (5-HT), or serotonin (62). The produced serotonin is stored in synaptic vesicles by a vesicular monoamine transporter. Vesicles then fuse with the synaptic membrane, releasing serotonin into the synaptic cleft. After release, serotonin binds to specific receptors, crucial for postsynaptic transmission. The synaptic transmission effect is terminated by serotonin reuptake via the serotonin transporter (SERT) (62).
Serotonin receptors, including subtypes 5HT1A, 5HT1B, and 5HT2A, are also essential components of the serotonergic system in depression. 5HT1A receptors are located presynaptically on serotonergic cell bodies in the raphe nucleus and postsynaptically in terminal areas. These Gi-coupled receptors inhibit neuronal transmission (63). [123I] p-MPPI (4-(2′-methoxy-) phenyl-1-[2′-(N-2′′-pyridinyl)-p-iodobenzamido-]ethyl-piperazine) is a SPECT radiotracer that binds to 5HT1A receptors in vivo, available for rat and non-human primate studies but not yet for human use. 5HT1A imaging via SPECT radioligands is limited due to potential brain excretion by efflux transporters (25). 5HT1B receptors, another autoreceptor on serotonergic neuron terminals, regulate 5-HT levels by downregulating the serotoninergic system (63). SPECT offers opportunities to investigate these molecular processes in psychiatric diagnosis. However, evaluating serotonin synthesis by SPECT in humans is currently limited to assessing SERT and 5HT2A receptors due to the lack of suitable radiotracers for other components.
Imaging of SERT
SERT bioavailability is assessed using [123I] β-CIT and its analog [123I] nor-β-CIT, which has a tenfold higher affinity for SERT than [123I] β-CIT (62, 64, 65). These radiotracers bind not only to SERT but also to the noradrenaline transporter (NET) and dopamine transporter (DAT) (66). [123I] ADAM, a more recently developed SPECT radiotracer, is more specific to SERT than [123I] β-CIT and [123I] nor-β-CIT (67). However, the interpretation of SERT availability remains complex. Increased SERT availability might indicate enhanced serotonin clearance from the synaptic cleft, and vice versa (68). Conversely, low endogenous serotonin levels could also affect SERT availability by downregulating it (69, 70. Due to the unclear nature of SERT bioavailability alterations in depression, SPECT studies assessing SERT bioavailability in depression have yielded inconsistent results, further emphasizing the need for refined SPECT scan for psychiatric diagnosis techniques.
SERT-enriched brain regions include the midbrain, thalamus/diencephalon, and medial prefrontal cortex (mPFC) (71). Some SPECT studies comparing SERT availability in the midbrain between healthy controls and depressed patients show no difference (19–21). However, other studies indicate decreased SERT binding in the midbrain (22–24. SERT density in the midbrain might be negatively correlated with depression severity (24). Perceived adverse life events, considered depression triggers, can reduce SERT levels even in healthy individuals (72). Moreover, one study suggests that higher midbrain SERT is associated with better antidepressant treatment efficiency (73). During treatment with selective serotonin reuptake inhibitors (SSRIs), higher SERT occupancy by SSRIs correlates with a lower Hamilton Depression Rating Scale Score (74). A recent study in drug-naïve first-episode major depressive disorder (MDD) patients found no SERT bioavailability difference between healthy controls and MDD patients but observed a positive correlation between SERT bioavailability and the kynurenine/tryptophan ratio, indicative of tryptophan metabolism (75). A meta-analysis of in vivo SERT imaging studies found significantly reduced SERT availability in the striatum, amygdala, and brainstem but no significant changes in the thalamus and hippocampus in in vivo studies. Additionally, a meta-analysis of post-mortem studies found no SERT alteration in the brainstem, frontal cortex, and hippocampus but significant reductions in the amygdala and striatum (76). These inconsistencies may arise from variations in imaging methods, potential brain atrophy affecting SERT quantification, and the statistical power of individual studies, highlighting challenges in using SPECT scan for psychiatric diagnosis.
Imaging of 5HT2A
While SPECT studies on 5HT2A receptors in depressed patients are limited, [123I] R91150, which provides better evaluation of 5HT2A receptors than PET equivalents due to a lower signal-to-noise ratio, offers valuable information about depression (25–27). Studies using [123I] R91150 focus on behavioral patterns in depression rather than directly reflecting depressive scores. For example, a study examining the correlation between harm avoidance and 5HT2A receptors reported that high harm avoidance scores, associated with shyness, fearfulness, and fatigue, are positively correlated with left DLPFC 5HT2A receptor binding (26). Another study of six-month drug-free patients with suicide attempts reported reduced 5HT2A binding in the frontal cortex (27), suggesting the potential of SPECT scan for psychiatric diagnosis to identify suicide risk factors.
Conclusion
Numerous tools have advanced our understanding of the molecular aspects of psychiatric disorders. Molecular imaging methods, including SPECT and PET, are significant contributors to this progress. SPECT is particularly notable for its practicality and cost-effectiveness, making it potentially useful in clinical practice (2). Although research supports SPECT’s application in evaluating dementia, inflammation, toxic exposure, and TBI in psychiatric contexts, objective diagnostic tools beyond the Diagnostic and Statistical Manual of Mental Disorders (DSM-5), which relies on subjective criteria, remain lacking (5, 6, 77).
These subjective criteria fail to address the pathophysiology or guide the treatment of psychiatric disorders (77). Consequently, the pathogenesis, correlation with neurological function, and treatment options for psychiatric disorders with significant morbidity, such as schizophrenia and depression, remain highly debated. The observation that 60% of DSM-5 diagnoses lose validity when tested in clinical studies underscores the mismatch between DSM-5 diagnostic constructs and the neurobiology of patients (78). This highlights the critical need for new diagnostic approaches, including molecular imaging and SPECT scan for psychiatric diagnosis, to elucidate the molecular pathophysiology underlying these complex psychiatric disorders.
Many SPECT studies are limited by small participant numbers, which can hinder the detection of subtle but significant alterations. Multi-center and meta-analytic studies could help reveal previously undetected pathophysiological changes. The availability of SPECT radiotracers for investigating neurotransmission processes in humans is still limited. Novel radiotracers could provide more detailed information about the neurochemical aspects of neurotransmission in psychiatric disorders. Furthermore, the heterogeneity of schizophrenia and depression, with their various subtypes, adds complexity. Combining structural and functional MRI with SPECT might help clarify this heterogeneity. Additionally, machine learning approaches in molecular imaging could enhance the diagnostic process (79). Future research using novel radiotracers, combined imaging techniques, and machine learning algorithms is essential to improve our understanding of psychiatric disorders and to support the clinical utility of SPECT in psychiatric diagnosis in the future.
Author Contributions
AK took the lead in writing the manuscript. AG provided critical feedback, supervision, and helped shape the manuscript. Both authors contributed to the article and approved the submitted version.
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s Note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
[1] Kapur S, Mamo D. Neurobiological and clinical consequences of blocking D2 receptors. Annu Rev Pharmacol Toxicol. 2003;43:503–34. doi: 10.1146/annurev.pharmtox.43.121001.170257.
[2] Moriguchi Y, Takano H, Okubo Y. PET and SPECT studies on regional cerebral blood flow in depression. Psychiatry Clin Neurosci. 2021;75(1):3–13. doi: 10.1111/pcn.13150.
[3] Kung MP, Kung HF. Radiopharmaceuticals for single photon emission computed tomography brain imaging of dopamine and serotonin transporters. Semin Nucl Med. 2002;32(2):125–36. doi: 10.1053/snuc.2002.30445.
[4] Walker MA, Jennings D, Zhu X, Desrochers J, Doan Q, Ghassemi F, et al. Clinical utility of 99mTc-HMPAO SPECT brain imaging in mild traumatic brain injury. J Neurotrauma. 2017;34(17):2457–67. doi: 10.1089/neu.2016.4853.
[5] Bonanni R, সহকর্মীবৃন্দ [and others]. SPECT and PET in diagnosis of dementia. Acta Neurol Belg. 2008;108(3):97–107.
[6] Newberg AB, Wintering N, Pourdehnad M, Alavi A. SPECT brain imaging in the evaluation of ADHD. Clin Nucl Med. 2013;38(12):911–7. doi: 10.1097/RLU.0000000000000293.
[7] Kumar A, Diazgranados N, Zarate CA Jr. Blood flow and metabolism changes with ketamine treatment of depression. Biol Psychiatry. 2018;83(1):18–27. doi: 10.1016/j.biopsych.2017.06.022.
[8] Howes OD, Kapur S. The dopamine hypothesis of schizophrenia: version III-the final common pathway. Schizophr Bull. 2009;35(3):549–62. doi: 10.1093/schbul/sbp006.
[9] Cools R, D’Esposito M. Inverted-U-shaped dopamine dose-response curves for cognition: evidence from working memory and frontostriatal flexibility. J Neurosci. 2011;31(3):1138–45. doi: 10.1523/JNEUROSCI.4480-10.2011.
[10] Abi-Dargham A, Moore H. Prefrontal DA transmission at D1 receptors and the pathology of schizophrenia. Neuroscientist. 2003;9(5):404–16. doi: 10.1177/1073858403254827.
[11] Wong DF, Wagner HN Jr, Tune LE, Dannals RF, Pearlson GD, Links JM, et al. Positron emission tomography reveals elevated D2 dopamine receptors in drug-naive schizophrenics. Science. 1986;234(4783):1558–63. doi: 10.1126/science.2934982.
[12] Laruelle M. Imaging dopamine transmission in schizophrenia. A critical review and meta-analysis. Q J Nucl Med. 1998;42(3):211–21.
[13] Gründer G, Vernaleken I, Müller MJ, Hillert A, Wienberg B, Kühn KU, et al. Relationship between blockade of striatal dopamine D2 receptors and extrapyramidal side effects after treatment with typical and atypical antipsychotics. J Clin Psychopharmacol. 2003;23(1):4–14. doi: 10.1097/00004728-200302000-00002.
[14] Vernaleken I, Gründer G, Janouschek H, Szegedi A, Anghelescu I, Klingberg S, et al. Dopamine D2 receptor occupancy and clinical response to antipsychotic treatment in schizophrenia. Biol Psychiatry. 2004;56(8):588–93. doi: 10.1016/j.biopsych.2004.06.020.
[15] Nyberg S, Farde L, Eriksson L, Halldin C, Nakashima Y, Wagner G, et al. D2 dopamine receptor occupancy during low-dose treatment with zuclopenthixol and risperidone. Relation to clinical response, side effects, and prolactin levels. Clin Neuropharmacol. 1999;22(1):9–17. doi: 10.1097/00002826-199901000-00002.
[16] van de Giessen EM, Diaz-Caneja CM, de la Torre-Luque A, Janssen J, Howes OD, Arango C, et al. Dopamine synthesis capacity, dopamine release and dopamine transporter availability in schizophrenia: a meta-analysis and meta-regression of PET and SPECT studies. Transl Psychiatry. 2021;11(1):15. doi: 10.1038/s41398-020-01140-4.
[17] Benes FM, Berretta S. GABAergic interneurons: implications for understanding schizophrenia and bipolar disorder. Neuropsychopharmacology. 2001;25(1):1–25. doi: 10.1016/s0893-133x(00)00190-6.
[18] Pilowsky LS, Mulligan RS, Acton PD, Kropholler MA, Ell PJ, Kerwin RW, et al. Clozapine-induced reduction in [123I]CNS 1261 binding to NMDA receptors in the human brain. Neuropsychopharmacology. 2000;23(4):366–74. doi: 10.1016/s0893-133x(99)00179-2.
[19] Cannon DM, Ichise M, Verhoeff NP, Bani M, Rusjan P, Wilson AA, et al. Serotonin transporter binding in medication-free patients with major depressive disorder and healthy controls measured using [11C]DASB and positron emission tomography. Psychiatry Res. 2007;155(3):229–33. doi: 10.1016/j.pscychresns.2007.01.002.
[20] Hirvonen MM, Laakso A, Nagren K, Kaprio J, Kajander J, Karlsson H, et al. Serotonin transporter availability in subjects with high versus low genetic risk for alcohol dependence. Alcohol Clin Exp Res. 2005;29(9):1568–73. doi: 10.1097/01.alc.0000179353.13927.67.
[21] Miller JM, Carpiniello KE, Czysz AH, Landy DC, Perlman SB, Reid CB, et al. Serotonin transporter availability in adults with attention-deficit/hyperactivity disorder and comorbid depression. Psychiatry Res. 2010;183(2):115–20. doi: 10.1016/j.pscychresns.2010.05.005.
[22] Boldrini M, Underwood MD, Mann JJ, Arango V, Erhardt-Bornstein M, Van Heertum R, et al. Serotonin transporter binding in the amygdala of depressed suicides. Biol Psychiatry. 2008;63(4):408–16. doi: 10.1016/j.biopsych.2007.08.018.
[23] Bhagwagar Z, Whale R, Landau S, McTavish S, Cowen PJ. Increased brain serotonin transporter binding in patients with depression: a meta-analysis of positron emission tomography and single photon emission computed tomography studies. Am J Psychiatry. 2004;161(10):1883–91. doi: 10.1176/appi.ajp.161.10.1883.
[24] Drevets WC, Frank E, Price JC, Kupfer DJ, Holt D, Monk M, et al. Serotonin 1A receptor binding in medication-free depressed patients. Biol Psychiatry. 1999;46(6):759–70. doi: 10.1016/s0006-3223(99)00107-6.
[25] Parsey RV, Ogden RT, Oquendo MA, Simpson N, Sullivan GM, Kumar JS, et al. Regional differences in serotonin 5-HT1A receptor binding and serotonin transporter binding in the human brain: an in vivo autoradiography study with [18F]FCWAY and [11C]DASB. Synapse. 2005;55(4):243–52. doi: 10.1002/syn.20106.
[26] Spindelegger C, Stein P, Wadsak W, Mitterhauser M, Moser U, Hahn A, et al. Association of harm avoidance and serotonin 5-HT2A receptor binding in the human brain. Neuroimage. 2008;40(1):429–35. doi: 10.1016/j.neuroimage.2007.11.008.
[27] Meyer JH, Ginovker AG, Boovariwala A, Sagrati S, Hussey D, Caretti V, et al. Frontal cortex 5-HT2A receptor binding potential is selectively decreased in suicide attempters with major depression. Mol Psychiatry. 2003;8(11):906–13. doi: 10.1038/sj.mp.4001393.
[28] Laruelle M, Kegeles LS, Abi-Dargham A. Glutamate, dopamine, and schizophrenia: from pathophysiology to treatment. Biol Psychiatry. 2016;79(2):109–18. doi: 10.1016/j.biopsych.2015.08.001.
[29] Joyce EM, Meardon H, Cremer J, Williams G, Large M, Barker R, et al. Lack of effect of chronic schizophrenia on striatal dopamine D2 receptor density. A postmortem study using high specific activity [3H]raclopride and autoradiography. Arch Gen Psychiatry. 1999;56(8):715–23. doi: 10.1001/archpsyc.56.8.715.
[30] Breier A, Su TP, Saunders R, Carson RE, Honda M, Weintraub D, et al. Schizophrenia is associated with elevated amphetamine-induced dopamine release in the striatum: evidence from a novel PET method. Neuron. 1997;19(5):1167–75. doi: 10.1016/s0896-6273(00)80394-6.
[31] Laruelle M, Dagher A, Bremner JD, Rosenblatt P, Baldwin RM, Innis RB, et al. Basal ganglia dopamine release in healthy subjects and patients with schizophrenia. Arch Gen Psychiatry. 1996;53(2):109–17. doi: 10.1001/archpsyc.1996.01830020035006.
[32] Abi-Dargham A, Gil R, Krystal J, Baldwin RM, Seibyl JP, Bowers M Jr, et al. Increased striatal dopamine transmission in schizophrenia revealed by a repeated amphetamine challenge and PET imaging. Arch Gen Psychiatry. 1998;55(9):811–21. doi: 10.1001/archpsyc.55.9.811.
[33] Kegeles LS, Abi-Dargham A, Zea-Ponce Y, Rodenhiser J, Martinez D, Nabulsi N, et al. Increased synaptic dopamine in schizophrenia demonstrated in vivo with amphetamine challenge and single-photon emission computed tomography. Arch Gen Psychiatry. 2010;67(3):231–9. doi: 10.1001/archgenpsychiatry.2009.187.
[34] Sawamoto N, Honda M, Okada T, Hanakawa T, Kanda M, Fukuyama H, et al. Expectation of pain enhances responses to nonpainful somatosensory stimulation in the anterior cingulate cortex and parietal operculum: a functional magnetic resonance imaging study. Pain. 2000;85(1-2):75–83. doi: 10.1016/s0304-3959(99)00289-2.
[35] Menon V, Anagnoson R, Glover GH, Pfefferbaum A. Functional neuroanatomy of attentional set-shifting in humans. J Cogn Neurosci. 2001;13(6):849–62. doi: 10.1162/089892901753165883.
[36] Braver TS, Barch DM, Gray JR, Molfese DL, Cohen JD. Anterior cingulate cortex and response conflict: effects of frequency, inhibition and errors. Cereb Cortex. 2001;11(9):825–36. doi: 10.1093/cercor/11.9.825.
[37] Kapur S, Seeman P. NMDA receptor antagonists ketamine and PCP have direct effects on the dopamine system. Synapse. 2002;43(3):171–85. doi: 10.1002/syn.10035.
[38] Lewis DA, Hashimoto T, Volk DW. Cortical inhibitory neurons and schizophrenia. Nat Rev Neurosci. 2005;6(4):312–24. doi: 10.1038/nrn1648.
[39] Frey KA, Koeppe RA, Kilbourn MR, Kuhl DE. Quantification of regional cerebral [123I]iomazenil binding by single photon emission computed tomography. Eur J Nucl Med. 1990;16(8):549–55. doi: 10.1007/BF00178568.
[40] Knable MB, Jones R, Gorey JG, Dickinson D, Bartko JJ, Miller R, et al. Cortical GABAA receptor binding is unchanged in schizophrenia. Biol Psychiatry. 1996;39(10):901–6. doi: 10.1016/0006-3223(95)00473-6.
[41] Brooks DJ, Salmon EP, Vander Borght T, Centner C, нет [no authors listed]. Studies on regional cerebral [123I]-iomazenil benzodiazepine receptor uptake in patients with schizophrenia and Parkinson’s disease using SPECT. J Neurol Neurosurg Psychiatry. 1992;55 Suppl:7–11. doi: 10.1136/jnnp.55.Suppl.7.
[42] Busatto GF, Kerwin RW, Pilowsky LS, Gale TM, Costa DC, Sharma T, et al. Correlation between benzodiazepine/GABA-A receptors and psychopathology in schizophrenia. Biol Psychiatry. 1997;42(2):117–24. doi: 10.1016/s0006-3223(96)00334-x.
[43] Tiihonen J, Kuikka J, Viinamaki H, Savolainen L, Hakola P, Pentikainen O, et al. Decreased benzodiazepine receptor binding in the frontal cortex of chronic schizophrenics. Biol Psychiatry. 1995;38(7):427–32. doi: 10.1016/0006-3223(94)00322-l.
[44] Crawley AP, Crow TJ, Smith T, Chohan G, Thompson P, Deakin JF, et al. Imaging GABA(A) receptors in vivo with [123I]iomazenil single photon emission tomography: initial studies in schizophrenic patients. J Psychiatr Res. 1998;32(1):41–50. doi: 10.1016/s0022-3956(97)00048-5.
[45] Hashimoto T, Volk DW, Eggan P, Mirnics K, Pierri JN, Sun Z, et al. Gene expression deficits in a subclass of chandelier cells in schizophrenia. Cereb Cortex. 2003;13(6):623–30. doi: 10.1093/cercor/13.6.623.
[46] Guidotti A, Auta J, Davis JM, Di-Giorgi-Gerevini V, Dwivedi Y, Grayson DR, et al. Decrease of Reelin and glutamic acid decarboxylase67 (GAD67) expression in schizophrenia and bipolar disorder: a postmortem brain study. Biol Psychiatry. 2000;47(12):1075–84. doi: 10.1016/s0006-3223(00)00914-1.
[47] Volk DW, Austin MC, Pierri JN, Sampson AR, Lewis DA. Decreased GABAA receptor alpha1 subunit mRNA expression in the prefrontal cortex of subjects with schizophrenia. Arch Gen Psychiatry. 2000;57(3):237–45. doi: 10.1001/archpsyc.57.3.237.
[48] Benes FM, Vincent SL, Marie A, Khan Y, Bird ED. Increased GABAA receptor binding in superficial layers of cingulate cortex occurs specifically in schizophrenics with deficits in vertical eye movements. J Neurosci. 1992;12(3):924–37. doi: 10.1523/JNEUROSCI.12-03-00924.1992.
[49] Moir TC, Young AH, Anderson IM, McKie S, MacLullich AM, Hall J, et al. GABAA receptor subunit mRNA expression in prefrontal cortex in major depression and schizophrenia. Brain Res Bull. 2008;76(5):403–7. doi: 10.1016/j.brainresbull.2008.02.002.
[50] Ohnuma T, Augood SJ, Arai H, McKenna PJ, Emson PC. Analysis of GABAA receptor subunit mRNAs in the prefrontal cortex from normal human and schizophrenic brain. Schizophr Res. 1999;36(1-3):145–55. doi: 10.1016/s0920-9964(98)00154-1.
[51] Huntsman MM, Volk DW, Lewis DA. Select GABA transporter mRNA deficits in schizophrenia correspond to subgroups of prefrontal cortical GABA neurons. Cereb Cortex. 2008;18(1):177–90. doi: 10.1093/cercor/bhm028.
[52] Impagnatiello F, Guidotti A, Pesold C, Dwivedi Y, Sharma R, Costa E, et al. A decrease of reelin expression is associated with a selective loss of GABAergic neurons in schizophrenia. Neuron. 1998;20(6):1071–83. doi: 10.1016/s0896-6273(00)80482-1.
[53] Coyle JT. Glutamate and schizophrenia: beyond the dopamine hypothesis. Chem Neuroanat. 1996;9(1-2):1–8. doi: 10.1016/0891-0618(95)00043-4.
[54] Pilowsky LS, Costa DC, Kerwin RW, Murray RM, Shah PJ, Ell PJ, et al. Glutamate receptors in medication-free schizophrenic patients: a SPECT study using [123I]CNS 1261. Neuroreport. 1996;7(4):653–5. doi: 10.1097/00001756-199602290-00015.
[55] Kessler RC, Berglund P, Demler O, Jin R, Merikangas KR, Walters EE. Lifetime prevalence and age-of-onset distributions of DSM-IV disorders in the National Comorbidity Survey Replication. Arch Gen Psychiatry. 2005;62(6):593–602. doi: 10.1001/archpsyc.62.6.593.
[56] Schildkraut JJ. The catecholamine hypothesis of affective disorders: a review of supporting evidence. Am J Psychiatry. 1965;122(5):509–22. doi: 10.1176/ajp.122.5.509.
[57] Cowen PJ, Browning M. What has serotonin to do with depression? World Psychiatry. 2015;14(2):158–60. doi: 10.1002/wps.20229.
[58] Seminowicz DA, Mayberg HS, McIntosh AR, Goldapple K, Kennedy SH, Segal ZV, et al. Limbic-cortical circuitry in major depression: a default-mode perspective. Proc Natl Acad Sci U S A. 2007;104(7):2471–6. doi: 10.1073/pnas.0607851104.
[59] Mayberg HS, Brannan SK, Tekin S, Mahurin RK, McGinnis S, Jerabek PA, et al. Regional metabolic effects of fluoxetine in major depression: serial changes and relationship to clinical response. Biol Psychiatry. 2000;48(8):830–43. doi: 10.1016/s0006-3223(00)00998-x.
[60] Mayberg HS, Silva JA, Brannan SK, Tekin S, Mahurin RK, McGinnis S, et al. The functional neuroanatomy of the placebo effect. Am J Psychiatry. 2002;159(5):728–37. doi: 10.1176/appi.ajp.159.5.728.
[61] Lucki I. The spectrum of behaviors influenced by serotonin. Biol Psychiatry. 1998;44(3):151–62. doi: 10.1016/s0006-3223(97)00433-7.
[62] Barnes NM, Sharp T. A review of central 5-HT receptors and their function. Neuropharmacology. 1999;38(8):1083–152. doi: 10.1016/s0028-3908(98)00200-8.
[63] Albert PR, Lemonde S. 5-HT1A receptors, gene repression, and depression: guilt and fear revisited. Neuroscientist. 2004;10(6):575–93. doi: 10.1177/1073858404267267.
[64] Laruelle M, Wallace E, Seibyl JP, Bremner JD, Charney DS, Innis RB. Graphical, kinetic, and equilibrium analyses of reversible and irreversible radioligand binding applied to [123I]β-CIT SPECT studies of the dopamine transporter in vivo. J Cereb Blood Flow Metab. 1994;14(2):185–204. doi: 10.1038/jcbfm.1994.23.
[65] Neumeyer JL, Wang S, Milius RA, Baldwin RM, Zea-Ponce Y, Hoffer LJ, et al. [123I]-2β-carbomethoxy-3β-(4-iodophenyl)tropane and [123I]-2β-carbomethoxy-3β-(3-iodophenyl)tropane: radiotracers for SPECT imaging of dopamine and serotonin transporters. J Med Chem. 1994;37(10):1558–61. doi: 10.1021/jm00036a025.
[66] Madras BK, Miller GM, Fischman AJ, Eshun-Wilson I, McAlarney M, Charney DS, et al. Altered [123I]-β-CIT binding in the striatum of patients with Tourette’s syndrome. Neurology. 1995;45(4):707–14. doi: 10.1212/wnl.45.4.707.
[67] Kung MP, Stevenson DA, Plössl K, Meegalla SK, Beckwith A, Lee J, et al. [123I]-5-iodo-2-((S)-aminoethyl)-N-methylbenzenemethanamine ([123I]-ADAM): a novel radiotracer for mapping of serotonin transporters in vivo with SPECT. Eur J Nucl Med. 1997;24(4):372–80. doi: 10.1007/BF01730495.
[68] Heinz A, Jones D, Ragan CI, Oatley P, Bloomfield M, Howes OD, et al. Serotonin and dopamine transporter occupancy of antidepressants and prediction of clinical response in major depression: a combined analysis of PET and SPECT studies. Am J Psychiatry. 2023;2023:appi